Outline
Pelvic Floor Anatomy and Function, 1026
Risk Factors and Indications for Dynamic Pelvic Imaging, 1027
Ultrasound Imaging, 1027
Endoanal Sonography for Anal Incontinence, 1027
Endoanal Sonography for Fistulas and Perianal Abscesses, 1029
Transperineal Sonography of the Anal Sphincter, 1029
Pelvic Floor Sonography, 1030
Dynamic Pelvic Floor Magnetic Resonance Imaging, 1034
Technical Considerations, 1034
Anatomy and Magnetic Resonance Imaging Reference Lines, 1036
Abnormal Findings on Magnetic Resonance Imaging, 1036
Limitations of Magnetic Resonance Imaging, 1039
Summary and Future Direction, 1040
Summary of Key Points
- •
Endoanal sonography has an important role in the assessment of the anal sphincter in patients with fecal incontinence.
- •
Sonography and magnetic resonance imaging (MRI) of the pelvic floor can be useful in evaluation of women with pelvic floor disorders and in assessment of mesh implants.
- •
There is no strong evidence for routine use of pelvic floor sonography in assessment of women with pelvic floor disorders.
- •
Fistulas and perirectal abscess can be evaluated well by either sonography or MRI prior to therapy.
- •
Urethral diverticula are well seen by either sonography or MRI, although the direct connection to the urethra is best depicted with MRI.
- •
Criteria for postintervention and postsurgical evaluation are less well defined for both sonography and MRI.
Pelvic organ prolapse (POP), fecal incontinence, and urinary incontinence in women have been largely ignored from an imaging perspective; recent innovations in both imaging and treatment options have increased attention on the evaluation and management of these conditions. Traditionally, there has been limited research focusing on imaging of female pelvic disorders other than malignancy. This may be in part due to reluctance of women to report this constellation of problems to their clinicians because of embarrassment or because they ascribe these symptoms to a “part of life” that is to be expected with aging and childbirth. However, pelvic floor dysfunction is a significant medical and quality of life issue, and pelvic floor disorders have been reported to affect one in four of all women. Although prevalence increases with age, pelvic floor disorders should not be considered a normal part of aging.
Pelvic floor disorders include POP, urinary incontinence, and anal incontinence. These conditions may manifest with a variety of urinary and defecatory symptoms as well as pain and sexual dysfunction. Urinary incontinence (UI) is defined as involuntary leakage of urine. There are two common subtypes—stress urinary incontinence and urgency urinary incontinence. Stress urinary incontinence occurs in response to increased intra-abdominal pressure that occurs during exertion, strain, coughing, or sneezing. Urgency urinary incontinence occurs at the time of sensation of urgency to urinate. The combination of urinary urgency and frequency is also referred to as overactive bladder syndrome.
Anal incontinence is the involuntary loss of feces or flatus. The underlying cause of anal incontinence is often an anal sphincter defect following vaginal delivery and less commonly direct trauma, including surgery. However, a variety of other factors can contribute. Neurogenic disorders may result in anal incontinence in the setting of an intact anal sphincter and may be caused by traction neural injury during delivery, as well as by diabetes, certain medications, radiation therapy, and aging. Fecal incontinence is six to eight times more common in women than in men, and the incidence increases with aging. The incidence is highest among multiparous older women, and it is estimated that up to 15% of multiparous women in the United States over the age of 64 years may have fecal incontinence to some degree.
Female pelvic medicine and reconstructive surgery (FPMRS) and urogynecology focus on diagnosis and treatment of pelvic floor disorders. A wide variety of treatment options is available for pelvic floor disorders, ranging from conservative measures, such as pelvic floor muscle training, physical therapy, medications, and supportive devices, to surgical intervention. The lifetime chance of having primary surgery for POP or stress urinary incontinence is estimated to be 20% by the age of 80 years. Many do not seek care, but even the few who seek help may not be effectively treated if multicompartmental disease is not fully appreciated and addressed. Unfortunately, recurrence rate is high, leading to reoperation in 30% of women. Until successful treatment is realized, the impact on a woman’s daily quality of life can be significant. Persistent symptoms such as pain or incontinence can be both debilitating as well as embarrassing and may carry significant psychosocial implications.
Despite ongoing efforts to improve understanding of pelvic floor disorders, the underlying pathophysiologic mechanism is not fully elucidated. Although clinical assessment and examination remain the mainstay of pelvic floor evaluation, imaging has emerged as a potentially useful modality. Indeed, clinicians have begun to avoid using terms such as cystocele and rectocele, recognizing that the physical examination only identifies prolapse of the vaginal walls—whether anterior and posterior—and cannot identify with certainty the pelvic organs lying behind the vaginal walls. Pelvic floor imaging, however, clarifies the specific anatomy underlying these conditions and potentially helps in understanding the physiologic and pathophysiologic changes. Assessment of anatomy and function using pelvic floor imaging is used increasingly to improve understanding of pelvic floor disorders and hence optimize treatment approaches. However, despite more than 2 decades of imaging research in pelvic floor disorders, imaging still plays a rather limited role in the evaluation of pelvic floor disorders at most institutions.
Pelvic Floor Anatomy and Function
The pelvic floor is a complex structure encompassing an interplay of smooth and striated musculature that, along with both fascia and ligaments, supports and assists in the function of the pelvic floor organs. The main muscles of the pelvic diaphragm are the levator ani muscles, comprising the pubococcygeus, puborectalis, and iliococcygeus muscles. These muscles span the space between the obturator internus muscle laterally, the pubis symphysis anteriorly, and the coccyx posteriorly. The levator hiatus is a funnel-shaped cleft in the muscles of the levator ani from which exit the urethra, the vagina, and the anal canal. The puborectalis muscle (PRM) is the most inferior (caudal) muscle of the pelvic floor. The two sides of the PRM attach to the two pubic rami anteriorly and merge posterior to the anal canal to form the anorectal angle. The superficial muscles of the pelvic floor make up the urogenital diaphragm and include the ischiocavernosus, bulbospongiosus, and transversus perinei superficialis muscles ( Fig. 35-1 ).

Fecal continence is maintained by both constriction of the anal canal as well as anorectal lift of the pelvic floor. The normal anal canal extends proximally from the puborectalis portion of the levator ani muscles to the anal verge, for a distance of approximately 4 cm. The structures surrounding the anal canal are responsible for maintaining fecal continence and include the internal anal sphincter (IAS), the external anal sphincter (EAS), and the PRM. The EAS encircles the anal canal and is a striated muscle that is under voluntary control. The superficial portion of the EAS muscle is attached posteriorly to the coccyx through the anococcygeal ligament and anteriorly inserts into the perineal body. The deep portion surrounds the anal canal with some integration with the PRM posteriorly and some fibers of this portion interdigitate with the deep transverse perineal muscles. The subcutaneous portion of the EAS is the portion that surrounds the anal canal most distally, approximately 1 cm below the termination of the IAS. The IAS is a smooth muscle, which is under autonomic control and is responsible for 80% of the resting anal sphincter tone. The IAS is created by thickening of the circular smooth muscle in the rectal wall and represents the terminal portion of the inner circular smooth muscle layer of the intestinal tract. The IAS is shorter in length than the EAS, terminating approximately 1 cm distal to the dentate line. The dentate line represents the junction of the subcutaneous and superficial components of the EAS. In addition to these two circular muscle layers, a longitudinal muscle layer extends between the EAS and IAS and serves to anchor the sphincter complex to the fascia of the levator ani as well as to the pelvic side wall.
The perineum separates the anus from the vagina. The central portion of the perineum contains the perineal body, which is the point where the EAS, the bulbospongiosus, and the superficial and deep transverse perineal muscles meet. The presence of a thick perineal body is suggestive of a normal anal sphincter.
The female urethra is a tubular conduit that passes through the retropubic space, perforating the perineal membrane to terminate at its external orifice (external urethral meatus) in the vaginal vestibule directly ventral to the vaginal opening. The normal female urethra is approximately 4 cm long. Urethral continence is maintained by the interplay of multiple structures. The urethral muscle layers are composed of both smooth muscle layers and the striated rhabdosphincter. Dorsally the rhabdosphincter is tightly connected to the ventral portion of the vagina. The proximal urethra and the bladder base are supported by the anterior vaginal wall, which creates a sling-like support that connects laterally to the levator ani muscles at the attachments of the arcus tendineus fasciae pelvis . As pelvic floor muscles play an important role in urethral closure for urinary continence, pelvic floor physical therapy targets these muscles to improve their function for management of incontinence and other pelvic floor disorders. In addition to the pelvic floor muscles, another factor important in maintaining urinary continence is the contraction of the smooth and striated muscles within the urethral wall, as well as the ligaments and the fascia that support the bladder and urethra in proper position when intra-abdominal pressure is increased. Urinary continence is preserved by support applied to the bladder neck during pelvic floor contraction, as well as constriction of the urethral lumen.
The normal function of the pelvic floor musculature aids in control of both urinary and anal function and also supports the pelvic floor contents. Pelvic floor muscles must perform both voluntary contractions as well as involuntary, or reflex, contractions preceding or at the time of increased abdominal pressure. In response to increased abdominal pressure, the superficial pelvic muscles, such as the anal and urethral sphincters, resist these pressures; and the levator ani muscles support the pelvic floor and counteract this increased pressure by contracting and creating a circular closing of the levator hiatus as well as an upward movement of the pelvic floor and perineum. The contraction of the PRM leads to reduction of its two limbs, and this change lifts the anal canal anteriorly, compressing the structures within the levator hiatus against each other as well as the back of the pubic symphysis.
Risk Factors and Indications for Dynamic Pelvic Imaging
In the surgical literature, there are well-defined risk factors associated with female pelvic floor disorders. Most studies on this topic suggest that pregnancy and childbirth are risk factors for developing pelvic floor dysfunction. Broadly, the trauma of childbirth plays a key role in the development of tears of the pelvic muscles or the EAS/IAS. Symptoms associated with these tears are worsened in the setting of other risk factors such as age, obesity, or poor stool consistency. However, not all women with muscle or sphincter tears will develop incontinence or prolapse. Risk factors for incontinence in women with sphincter tears are Caucasian race, fourth-degree tear during childbirth, increased age, and increased body mass index.
Only in the last 2 decades has cross-sectional imaging played a critical role in characterization of pelvic floor disorders. Surgeons previously based surgery on physical examination and clinical history. As an early adjunct, radiographic defecography was used to confirm abnormal movement of the pelvic structures, with a positive impact on care. More recently, MRI has been demonstrated to provide exquisite detail of the anatomy of the pelvic floor beyond previous techniques. Further advances with rapid image acquisition have allowed dynamic evaluation of pelvic floor displacement in the setting of strain or evacuation of bowel contents, similar to radiographic defecography. Even more recently, ultrasound has made inroads into pelvic floor imaging and has strengths of its own.
The numerous indications for dynamic pelvic floor imaging range from a bulging mass visualized within the vagina, sensation of pelvic pressure, urinary incontinence, voiding difficulty, anal incontinence, defecatory dysfunction, to postoperative evaluation for complications or recurrent abnormality. In the 2014 ACR Appropriateness Criteria for Pelvic Floor Dysfunction, both transperineal sonography and dynamic MRI are considered “usually appropriate” for symptoms relating to POP, urinary incontinence or dysfunction, and recurrent prolapse after surgical repair. Both “may be appropriate” for anal incontinence, whereas sonography performed with an endorectal probe or MRI with an endorectal coil are highly rated for this indication. For the evaluation of defecatory dysfunction, dynamic MRI or x-ray defecography are considered more appropriate than sonography. However, for evaluation of postoperative complications, standard sonography or MRI may be appropriate, whereas dynamic studies are usually not helpful.
The choice of imaging modality will vary by physician practice pattern and is largely dependent upon surgeon preference and available imaging expertise. Some hospitals may have these studies requested primarily by gastrointestinal surgeons, whereas patients at other sites may be evaluated more often by urogynecologists or gynecologic surgeons. Regardless of the referral source, sonography and MRI are becoming more widely used for evaluation of signs of POP and for workup of urinary and anal incontinence.
Sonography may be performed in the office of the surgeon and is a relatively inexpensive modality. It has been used to explore multiple aspects of pelvic floor dysfunction, including anal and urinary incontinence and POP. It is the best means of assessing the integrity of the IAS and EAS. Pelvic floor sonography has been shown to be a superior diagnostic tool in comparison to anal manometry, electromyogram, and conventional defecography. Sonography can also be used to provide biofeedback during pelvic floor training.
MRI may be especially useful in patients who have undergone a failed repair and are being evaluated for further therapy or when there is clinical concern for challenging anatomy or multicompartmental disease. Typically, surgeons make a plan based upon clinical experience, but clinical examination has its limitations in detection and delineation of pelvic floor disorders. MRI can directly impact care by refining and improving the surgical plan. In addition, MRI has been clearly shown to detect additional findings beyond clinical examination. Two studies support this concept. In a study reported by Kaufman and associates, MRI changed the surgeon’s plan in 41% of complex pelvic surgeries. In another study of 33 patients, the surgeon developed a plan before MRI and revisited the plan afterward. In 67%, the plan was changed based on the imaging findings. A relatively common specific benefit of MRI is the detection of unexpected multicompartmental disease, which substantially improves patient care and has been shown to reduce the rate of reoperation.
Ultrasound Imaging
Endoanal Sonography for Anal Incontinence
Endoanal sonography, like endoanal MRI, is well established in evaluation of the anal sphincter complex in women with fecal incontinence (FI). Fecal incontinence has been reported to affect 9% to 15% of the female population. Social and psychological consequences can be devastating to patients, and many do not discuss fecal incontinence with their providers. Fecal incontinence is usually multifactorial, and risk factors vary from aging, diabetes, neurologic disease, prior anal surgery, and prior obstetric anal sphincter injuries (OASIS). Endoanal sonography is best performed with two-dimensional (2D) ultrasound imaging with a 7- or 10-MHz rotating 360-degree radial array endoanal transducer. The patient is most commonly placed into the left lateral position with the knees bent upward. However, some imagers advocate using the prone or supine lithotomy position. Using a 360-degree radial array transducer, providers can identify the normal anal sphincter complex anatomy and rapidly delineate tears of the anal sphincters.
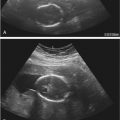
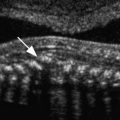
Using endoanal sonography, multiple layers or rings of differing echogenicity can be identified ( Fig. 35-2 ). The innermost ring is hyperechoic and represents the interface between the transducer and the anal mucosa and submucosal tissues. The underlying markedly hypoechoic ring represents the IAS. The next layer is the intermediate to hyperechoic longitudinal muscle, which is not always distinct on ultrasound imaging and is an extension of the outer longitudinal layer of the rectum. The outermost layer, which is generally echogenic to mixed echogenic, is the EAS. Endoanal axial imaging of the normal anal canal should include at least three characteristic levels. Images of the proximal (cranial) third of the anal canal should include the sling of the PRM, which is visualized posteriorly as a hyperechoic U-shaped structure that closely apportions the posterior portion of the EAS. At this level the EAS will not be observed anteriorly in women. However, the normal IAS will form a complete hypoechoic ring at this level ( Fig. 35-2A ). In the middle third, the complete rings of both the hypoechoic IAS and echogenic EAS will be seen ( Fig. 35-2B ). At the level of the distal anal canal, the normal EAS will extend approximately 1 cm below the IAS ( Fig. 35-2C ). The anal sphincter thickness has been shown to change with the aging process. The IAS becomes thicker with age, likely because of collagen replacement, whereas the EAS tends to become thinner as the patient ages.

Defects in the anal sphincter complex have been shown to have a strong association with fecal incontinence. Most commonly tears of the anal sphincter are combined and symmetric anterior defects in the EAS and IAS. Isolated EAS defects are also common whereas isolated IAS defects are much less frequent. IAS defects usually appear as a hyperechoic disruption of the normally hypoechoic ring of the IAS, whereas EAS tears are seen as hypoechoic, often heterogeneous defects disrupting the continuity of the relatively echogenic EAS. These sonographic changes correspond to the replacement of striated muscle with fibrosis. The extent of the tear is conventionally described as an analogy using the face of a clock, with the 12 o’clock position anterior, the 6 o’clock position posterior above the coccyx, 3 o’clock to the patient’s left, and 9 o’clock to the patient’s right ( Fig. 35-3 ). In studies in which ultrasound findings have been confirmed by intraoperative findings, the diagnostic accuracy of ultrasound imaging in detecting anal sphincter injury has been reported to have a sensitivity that well exceeds 90%. The findings of sonographic anal sphincter defects have been confirmed in multiple studies to correlate with poor sphincter function on anorectal manometry as well as with surgical and histologic findings of sphincter tears. Endoanal sonography also shows very good interobserver and intraobserver reliability. When added to immediate postpartum clinical assessment, endoanal sonography was shown to enhance recognition of occult anal sphincter tears in postpartum women with second-degree tears who did not have evidence of sphincter tear on clinical examination. Immediate repair of these occult anal sphincter tears reportedly decreased the risk of developing severe postpartum fecal incontinence based on 3 months follow-up examination.

Endoanal Sonography for Fistulas and Perianal Abscesses
Anorectal abscesses and fistulas usually result from anal gland infections secondary to obstruction from fecal impaction. Approximately 10% are reportedly due to inflammatory bowel disease or direct trauma (including anorectal surgical procedures). A perianal abscess is thought to spread in multiple directions along the path of least resistance and can lead to formation of fistulas. Anorectal fistulas are connections between the perianal skin or vagina and the lumen of the anus or the rectum, although some may be blind ending. For planning surgical intervention, the most important factors for surgeons to know are the location and the size of the abscess, the exact location and number of fistulous tracts, and the relationship of these fistulous tracts to the IAS and EAS. Parks and colleagues described four types of fistula tracts based on their relationship to the sphincteric structures: intersphincteric— passes through the IAS and then courses between the muscle layers to the perianal skin; transphincteric— passes through both the IAS and EAS and then through the ischiorectal fossa to the skin; suprasphincteric— passes above the PRM and then courses through the ischiorectal fossa to the skin; and finally extrasphincteric— direct communication between perineum and rectum, bypassing the anal canal.
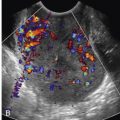
In imaging the fistula tract, the identification of the internal opening can be challenging. The internal opening can appear hypoechoic when acute inflammation is present, or hyperechoic when chronically inflamed or if it contains air. If the external opening of the fistulous tract is visible, injection of hydrogen peroxide into the fistulous opening may improve sonographic identification of the tract as a result of creation of gas bubbles within the fistula ( Figs. 35-4 and 35-5 ).


Transperineal Sonography of the Anal Sphincter
Although endoanal sonography remains a reference standard in anal sphincter assessment, transperineal sonography can be an effective but less invasive and more accessible modality for evaluation of the anal sphincter. The advantage of transperineal sonography for assessment of the anal sphincter is the possibility of dynamic assessment of the pelvic floor structures without interference of the endocavitary transducer. Transperineal assessment of the anal sphincter complex can be performed in the sagittal and transverse (axial) planes. When 3D sonography is used to image the anal sphincter complex, the transducer is oriented toward the anal canal. Postprocessing of the captured 3D volumes then allows detailed evaluation with thin-slice utility ( Fig. 35-6 ).

Using the transperineal technique, the IAS and EAS can be assessed, and the integrity of the PRM can be evaluated. With the transperineal approach, the mucosal folds of the anal canal can be visualized; they are not seen on endoanal imaging because the inserted transducer flattens the folds of the anal mucosa. The stellate-appearing normal folds of the anal mucosa on transperineal imaging have been referred to as the mucosal star . Asymmetry in these folds sometimes helps identify disease. Similar to the sonographic findings on endoanal imaging, the appearance of the transverse anal sphincter complex is different, depending on the level of imaging. Some studies suggest that anatomic assessment of the normal anal sphincter is comparable with endoanal and transperineal techniques. Although studies have suggested good agreement in detection of anal sphincter defects, others report inferior sensitivity of the transperineal approach. In the authors’ experience, endoanal sonography is superior to 2D transperineal sonography, unless 3D transperineal sonography is additionally performed.
Pelvic Floor Sonography
Over the past 2 decades, many authors have investigated the usefulness of sonographic assessment in women with POP and urinary incontinence or urinary symptoms. Despite the many reported techniques and approaches, pelvic floor sonography still plays a limited role in the investigation of pelvic floor disorders, and thus there are no standardized indications, terminology, and techniques. In this section, the most common techniques and indications will be summarized. The most commonly utilized protocols assess pelvic floor structures and pelvic floor muscle anatomy, integrity, and function as well as dynamic assessment of the pelvic floor structures with the transperineal or translabial approach. Other indications for pelvic floor sonography include assessment of urethral diverticula (sometimes with a transvaginal approach), surgically implanted mesh, and residual urine volume. Many studies have explored the use of 2D and 3D sonography for evaluation of women with pelvic floor symptoms. However, the exact role of pelvic floor sonography is still under investigation.
Women are usually scanned in the dorsal lithotomy position. However, some investigators use the left lateral decubitus position. The most common method for ultrasound evaluation of the pelvic floor is tranperineal sonography. Transperineal sonography encompasses two distinct techniques: the translabial technique, which uses a curved array transducer positioned on the labia majora or the perineum, and the transintroital technique, which uses a transvaginal transducer placed on the perineum or just within the vaginal introitus. Depending upon equipment availability, goals, and experience of the sonographer 2D, 3D, or 4D imaging is used. Imaging can be obtained under resting or dynamic conditions, during maximal strain (Valsalva maneuver), and with pelvic floor muscle contraction (Kegel exercise). Assessment can include a sagittal view of the pelvic floor viscera to localize the urethra, bladder, urethrovesical junction, vaginal walls, uterus, and anorectum, as well as volume imaging to assess pelvic floor hiatus and muscle anatomy, integrity, and function. Evaluation of the urethrovesical angle and anorectal angle can be performed under dynamic conditions.
The midsagittal view of the pelvic floor anatomy helps identify the relative location of the bladder and the urethra, the vagina, and the anorectum ( Fig. 35-7 ). From this view, the posterior urethrovesical and anorectal angles can be assessed. The posterior urethrovesical angle or urethrovesical junction is measured by creating a perpendicular line from the x-axis on the image and following this line to the margin of the bladder base. The anorectal angle is measured at the cross section of the line designating the longitudinal axis of the anal canal and the line along the posterior border of the rectal wall ( Fig. 35-8 ). These measurements can be performed at rest and with dynamic conditions of pelvic floor muscle contraction and strain. The dynamic displacement of the anorectal angle can provide visual biofeedback for PRM and levator ani activity, which can enhance proper muscle biomechanics and is readily accepted by women. In the sagittal view, the perimeter of the PRM is measured as a line that connects the posterior margin of the pubic symphysis and the anorectal angle and is used to assess the dynamic function of the PRM during contraction. Puborectalis activity can be measured as the difference between the “rest” and the “squeeze” measurement. The muscle usually contracts and moves anteriorly and cephalad during contraction.


Transperineal 3D sonography of the pelvic floor has the advantage of improved visualization of the entire pelvic floor hiatus or anal sphincter complex. The 3D images can be captured using either a curvilinear or transvaginal transducer placed on the perineum or labia. To optimize 3D imaging, the sagittal view should include the pubic symphysis and the anorectal angle because it is on this transverse plane that the pelvic floor hiatus (levator hiatus) is described ( Fig. 35-9 ).

Pelvic Floor (Levator) Hiatus and Pelvic Floor Musculature
Injury to the levator muscles can be seen either as an avulsion found at the point of muscle insertion onto the inferior pubic ramus or as a tear along muscle fibers. These injuries are possible consequences of overstretching of the levator ani muscles during the second stage of labor when the fetus travels through the birth canal. Levator muscle injuries can occur in 10% to 36% of women at the time of their first delivery. There is no agreement on classification of these muscle injuries. The most common injury related to childbirth is an avulsion injury of the insertion of the puborectalis (or pubvisceralis) muscle onto the pubic ramus ( Fig. 35-10 ).

A variety of studies have assessed pelvic floor morphologic changes related to vaginal delivery and the possible consequences of these changes on future symptoms. Dietz and Lanzarone showed that in a third of women delivering vaginally, avulsions were associated with stress urinary incontinence at 3 months postpartum. Further data from the same group suggest that levator muscle trauma is associated with prolapse of the anterior and apical compartment, but not with bladder dysfunction or urinary incontinence. They also demonstrated that a larger levator hiatus was correlated with POP. Research analyzing a large retrospective cohort suggested that levator ani avulsions are associated with prolapse in women with previous pelvic surgery. Furthermore, some studies suggest that levator avulsion is a predictor of prolapse recurrence after surgical correction of prolapse. Limited reports regarding attempts to surgically repair levator ani muscle avulsions have not been shown to reliably resolve associated pelvic floor disorders or improve symptoms. This indicates that restoration of anatomy does not always reconstitute function.
Urinary Incontinence
Pelvic floor sonography has been used to evaluate women with stress urinary incontinence to assess pelvic floor muscle contraction, urethral anatomy, and bladder neck position before and after incontinence procedures and to evaluate implanted materials used to treat stress urinary incontinence. Sonographic assessment of the anatomy and physiology of the urinary tract can provide insight into the pathophysiology of urinary incontinence. There have been many studies reported using sonography to assess women with urinary incontinence; however, there are no definitive studies on normal female sonographic anatomy of the lower urinary tract. With translabial or transperineal sonography the urethra appears in cross section as a hypoechoic structure surrounded by an echogenic layer representing the urethral muscularis and the sphincter. On sagittal view, the urethra usually courses behind the pubic symphysis. Tunn and coworkers suggested measuring the retrovesical angle with transperineal sonography. This measure evaluates urethral mobility. It is usually performed with Valsalva maneuver. In some patients with urinary incontinence, urethral funneling could be observed during Valsalva maneuver and even at rest. Significant funneling correlates with poor urethral closure pressure, suggesting urethral sphincter deficiency. Khullar and associates reported that women with urgency urinary incontinence and overactive detrusor had significantly thicker bladder walls, as did women with stress urinary incontinence.
Pelvic floor exercises are the mainstay of conservative measures for treatment of urinary incontinence. Bernstein and coworkers showed that pelvic floor muscle thickness and function could be reproducibly assessed by transperineal sonography. He also demonstrated that pelvic floor muscles were thinner in women older than 60 years and in women with stress urinary incontinence compared with continent control subjects. After pelvic floor muscle training, all groups showed increase in muscle thickness, and 60% of women with stress urinary incontinence showed subjective and objective improvement in symptoms. Dietz and coworkers used translabial sonography to provide biofeedback in teaching women to perform proper pelvic floor muscle contraction and showed that translabial sonography is a useful adjunct in pelvic floor muscle training.
Postintervention Imaging
Since the late 1990s, placement of minimally invasive slings has become a mainstay in surgical treatment of stress urinary incontinence. These slings are composed of synthetic material, most commonly prolene mesh, that is implanted around the urethra to restore anatomic support. The original retropubic placement of the slings was later modified with an option of using a transobturator placement. The sling is usually placed over the midurethra; however, position may vary. The mesh is highly echogenic and can be easily visualized with pelvic floor sonography ( Fig. 35-11 ). Using pelvic floor sonography, some researchers suggest that sling position along the length of the urethra (i.e., midurethral or in the proximal or distal third of the urethra) is not essential in restoring continence. In one study, Schuettoff and associates compared assessment of mesh slings with MRI and sonography. They suggested that sonography is most suited for assessment of suburethral and periurethral portions of the sling, whereas MRI is better for evaluating the retropubic portion of the mesh. Pelvic floor sonography can also demonstrate details of the relationship between the suburethral portion of the sling, the urethra, and the symphysis pubis.