, Gabriele Tonni2 , Elisabetta Pelo3 and Marco di Maurizio4
(1)
Prenatal Diagnostic Unit, University Hospital Meyer, Florence, Italy
(2)
Department of Obstetrics and Gynecology, Prenatal Diagnostic Service, Guastalla Civil Hospital, AUSL Reggio Emilia, Reggio Emilia, Italy
(3)
Medical Genetics, University Hospital Careggi, Florence, Italy
(4)
Diagnostic Imaging, University Hospital Meyer, Florence, Italy
18.1 Introduction
Agenesis of the ramus of the mandible (agnathia) is a rare disease that can be part of more than 100 genetic syndromes. From an embryological point of view, agenesis of the mandible is linked to defects in migration of neural crest cells that determine a developmental anomaly of the first arch, or mandibular arch, during the 4th week of embryogenesis [1]. Agnathia can be isolated or associated with other malformations, of which the most frequent is holoprosencephaly. Indeed, evidence from humans and animal models suggests that agnathia-holoprosencephaly represents a causally heterogeneous single developmental field defect [2]. Agnathia may be usually recognized by ultrasound during the second trimester of pregnancy and may be misinterpreted as micrognathia [3–5]. If micrognathia is the only ultrasound finding identified, parents should be aware of possible respiratory complications at birth, presence of facial clefting, and/or developmental delay [6]. Chromosomal abnormalities have been detected in 66 % of fetuses with micrognathia, in 77 % with macroglossia, in 48 % with cleft lip and palate, in 45 % with severe hypotelorism or cyclops, and in 32 % with nasal hypoplasia, proboscis, or single nostril [4, 7]. Teoh and Meagher [8] reported a first-trimester (13 weeks) diagnosis of micrognathia and receding chin by using transvaginal ultrasound, with later diagnostic confirmation at 19 weeks’ gestation. These findings were associated with complex cardiac defect and unilateral talipes equinovarus in a fetus with normal karyotype and with a definitive diagnosis of Pierre Robin syndrome. Sepulveda et al. [9] have demonstrated the value of the retronasal triangle view for detecting micrognathia in the first trimester. Absent visualization of the mandibular gap or failure to identify the mandible in coronal view is highly suggestive of micrognathia and should prompt a targeted ultrasound scan to assess for other anomalies. The role of the retronasal triangle view in the early prenatal diagnosis of orofacial clefts has been subsequently confirmed byTonni et al. [10].
18.2 Case Description
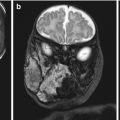
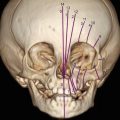
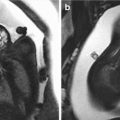
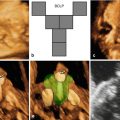
A 25-year-old primigravida underwent first-trimester combined screening for common trisomies at 12 weeks 2 days of gestation. Although the test resulted as low risk, the fetus had premaxillary protrusion and micrognathia at the ultrasound evaluation of the sagittal plane of the face (Fig. 18.1). Chorionic villus sampling was performed the same day. Quantitative fluorescence polymerase chain reaction (QF-PCR) for chromosomes 21, 18, 13 and sex chromosomes resulted as normal. Fetal karyotype in cultured villi was 46,XY. A thorough ultrasound examination, which included three-dimensional (3D) ultrasound of the fetal face and skull, was performed at 20 weeks of gestation. Application of 3D ultrasound reverse face techinque in surface maximum mode enabled clear detection of unilateral, left mandibular agenesis, micrognathia, and cleft lip (Fig. 18.2). The nose was abnormal in shape resembling a proboscis. The diagnostic clue was failure to identify the ossifying mandible in the sagittal and coronal planes from the temporo-mandibular joint to the symphysis of the chin. No other congenital malformatiosn were detected on ultrasound. Fetal magnetic resonance imaging (MRI) examination was arranged as complementary diagnostic investigation and confirmed the ultrasound diagnosis in both sagittal (Fig. 18.3) and coronal (Fig. 18.4) planes.
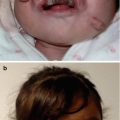
Full genetic consultation was then performed. Family history was remarkable for a first-degree cousin on the maternal side that had surgery for macroglossia due to Beckwith-Wiedemann syndrome. In addition, the father had an older brother with congenital clubfoot, a brother who died in the neonatal period due to multiple congenital malformations including cleft palate, clubfoot, and renal anomalies, and his mother had a miscarriage at 20 weeks of gestation after the ultrasound diagnosis of multiple congenital malformations in the fetus (Fig. 18.5).
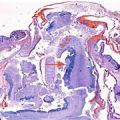
Multiplex ligation-dependent probe amplification (MLPA) was performed in a frozen sample of chorionic villi revealing a subtelomeric deletion of the short arm of chromosome 4 and a subtelomeric duplication of the long arm of chromosome 10 (Fig. 18.6). Subsequent paternal fluorescence in situ hybridization (FISH) was performed showing a balanced, cryptic translocation in the subtelomeric 4p/10q region (Fig. 18.7). The rearrangement in the father was inherited from his mother. In this case, the detected anomalies were attributed to a segregated, unbalanced, familial, cryptic translocation. Genetic counseling was conducted and the wide spectrum of ocular-auricular-vertebral anomalies involving the first and second pharyngeal arch such as Gorlin-Goldenhar, Treacher Collins and auriculo-condylar syndromes were discussed. The pathogenesis of Gorlin-Goldenhar syndrome may be associated with in utero exposure to teratogenic agents, chromosomal abnormalities (chromosomes 5, 18, 22, and X), while Treacher Collins is caused by haploinsufficiency of TOCF1 gene, the auriculocondylar syndromes (ARCND)are caused by gene mutations at different loci on chromosome 1p13 (ARCND1, GNAI3 gene),chromosome20p12 (ARCND2, PLCB4 gene) andchromosome 6p24 (ARCND3, EDN1 gene). Beckwith-Wiedemann syndrome, characterized by macroglossia, macrosomia, abdominal wall defect, and hemi-hyperplasia, and Silver-Russell syndrome (Pierre Robin sequence, cleft palate, renal anomalies, and fetal growth restriction) were also considered. The parents opted for second-trimester termination of pregnancy according to Italian legislation.
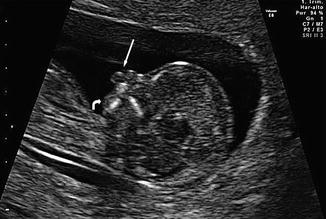





Fig. 18.1
Two-dimensional ultrasound performed at 12 weeks and 2 days of gestation. Note premaxillary protrusion (arrow) and micrognathia (curved arrow) in midsagittal plane
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree