Fig. 19.1
Renal anatomy and vascular supply. Stylized images demonstrate the relative lack of blood flow of the posterolateral aspect of the kidney, known as Brodel bloodless line of incision (a, b). This is the rational for targeting a posterolateral calyx when obtaining percutaneous access to the renal collecting system (c)
Indications
The indications for PCN generally fall into one of four major categories: relief of urinary obstruction, urinary diversion, access for endourologic procedures, and diagnostic testing. The most common indication for percutaneous nephrostomy (PCN) is relief of urinary obstruction. Common causes of obstruction are calculi and malignancies of the renal collecting system and pelvis. Iatrogenic injuries and pelvic masses are less common causes of obstruction. Mass effect on the ureters from uterine fibroids or a gravid uterus can result in symptomatic obstruction. Pelvic malignancies include cervical and endometrial cancer, bladder cancer, and prostate cancer. Less common causes are congenital abnormalities, retroperitoneal fibrosis, and lymphoma. Emergent decompression is indicated in cases of obstruction coupled with urosepsis and/or acute renal failure. Other indications for decompression include obstruction with infection, declining renal function, or intractable pain, commonly when retrograde decompression has been attempted or is not indicated. In the setting of ureteral obstruction due to malignancy, a common approach in patients with bilateral obstruction due to pelvic malignancy is to initially drain the symptomatic or most hydronephrotic collecting system and only perform secondary PCN if renal function does not improve or in the presence of infection. This common practice is due to the high failure rate of stent placement in the setting of extrinsic compression due to pelvic malignancy [5].
Consultation for PCN is common in the setting of urinary leakage to divert downstream urine extravasation and to support healing and sealing of the ureteral injury. Urine leak, either traumatic or iatrogenic, can occur anywhere in the urinary tract or at the site of an anastomosis. In the setting of known malignancy, or inflammatory fistulas, PCN is indicated to divert urine in conjunction with stent placement [3]. Urgent urinary diversion is indicated in cases of hemorrhagic cystitis, which carries a mortality of 4 %.
Access for Interventions/Endourologic Procedures
PCN access is often the initial procedure prior to therapeutic interventions including lithotripsy, stone extraction, and treatment of ureteral strictures and possible direct infusion of antifungal agents. At times, the only indication for PCN access is when retrograde stent placement is not feasible or successful [5].
Nephroscopic-guided interventions and operations may be performed in conjunction with a Urologist, include but are not limited to resection of urothelial neoplasms, endopyelotomy, and intracavitary injection of chemotherapy for transitional cell carcinoma [3].
When the indication for PCN is nephrolithotomy and lithotripsy, the procedure is commonly performed in the operating room immediately prior to lithotomy or lithotripsy [5].
Diagnostic Testing
Nephrostomy access can provide urologists a conduit for biopsy of urothelial lesions with the aid of brushes and possibly forceps. In addition, although less common, PCN access allows for the performance of functional studies such as the Whitaker test. Rarely, access may be performed for foreign body retrieval in the setting of a fractured or migrated ureteral stent.
Contraindications
There are no absolute contraindications for percutaneous nephrostomy, so in the presence of a “relative contraindication,” the risks must be carefully weighed in the setting of a critically ill patient requiring renal access or urinary diversion.
Relative Contraindications
Uncorrectable Coagulopathy
Nephrostomy catheter placement is considered a procedure with significant bleeding risk that may be difficult to detect or control. A relative contraindication is a severe coagulopathy that cannot be corrected or bleeding diathesis.
The consensus guidelines [5] for patients undergoing percutaneous nephrostomy tube placement include:
INR: Correct above 1.5 (95 % consensus).
Activated PTT: Stop or reverse heparin for values >1.5 times control.
Platelets <50,000: Transfuse.
Severe Hyperkalemia (Potassium Level Greater Than 7 mEq/L)
Electrolyte abnormalities may result in arrhythmias and metabolic acidosis if uncorrected.
Terminally Ill Patients
In the setting of imminent death, nephrostomy placement may not improve quality of life to a significant degree.
Pregnancy
The use of fluoroscopy should be avoided in pregnancy, especially during the first trimester. While percutaneous nephrostomy insertion may be performed solely using ultrasound, limited fluoroscopy may be needed.
Preprocedural Evaluation
Imaging
Preprocedural imaging of any contributory modality should be reviewed to evaluate the location and anatomy of the target kidney, with attention to the expected needle trajectory to avoid transgressing the bowel, pleura, liver, or spleen. Ultrasonography is commonly the first-line imaging for obstructive disease as it can assess renal morphology and degree of hydronephrosis. The proximal ureter and bladder are also often evaluated. CT and MR have the advantage of the ability to evaluate the entire urinary tract and all surrounding abdominal and pelvic structures. The size, number, and configuration of calculi are readily seen on non-contrast CT.
Laboratory Analysis
It is good practice to review all available laboratory studies, not limited to platelets and the patient’s coagulation profile. Abnormal coagulation and electrolyte results should be addressed and corrected, if the clinical scenario permits, prior to any intervention. A preprocedural documentation of the baseline serum creatinine can function as an indicator of improved renal function and postprocedural success.
Technical Aspects of Procedure
Anatomic Considerations/Patient Positioning
Standard positioning on the fluoroscopic table is prone or oblique prone, unless an anterior approach is indicated as often the case in transplant kidney interventions.
Prior to cleaning the skin, the targeted kidney may be imaged with ultrasound, CT, cone beam CT, or fluoroscopy to confirm prior imaging results, planned approach trajectory, and laterality.
Access through a specific calyx is occasionally required for stone extraction.
Antibiotics
The use of prophylactic antibiotics for PCN placement is directed toward preventing urosepsis from intravasation of organisms into the bloodstream during manipulation of the infected urinary system (Table 19.1) [6]. The need for such prevention may be stratified based on the at risk patient. Most authors recommend the use of prophylactic intravenous antibiotics for all obstructed patients; and all agree that high-risk patients with underlying comorbidities or immunosuppressed patients that are at high risk for infectious complications require prophylactic antibiotic coverage.
Table 19.1
Commonly used antibiotics for genitourinary procedures
Antibiotic | Dosage | Indication |
|---|---|---|
Cefazolin IV | 1 g | |
Ceftriaxone IV | 1 g | |
Ampicillin and gentamicin IV | 2 g and 1.5 mg | |
Vancomycin or clindamycin+ aminoglycoside | If penicillinallergic |
Sedation
Adequate sedation is a critical part of safe and effective nephrostomy placement. However, in situations when a patient cannot tolerate sedation and/or general anesthesia, a nephrostomy can be placed using only local anesthesia.
Prior to the procedure, the patient should refrain from oral intake for 8 h. Sedation should be performed in accordance with ASA guidelines, including maintained intravenous access and continuously monitored physiologic measurements throughout the procedure. Procedures in which the patient is in the prone position often facilitate airway management if the patient is breathing spontaneously with a supplemental oxygen mask or nasal cannula. Suction and supplemental oxygen must be immediately available and administered as needed. Securing the airway in the prone position with an endotracheal tube (ETT) is recommended for prolonged procedures, in patients with cardiopulmonary instability or when aspiration and loss of airway are likely [7].
According to ACR-SIR guidelines, intravenous sedative/analgesic drugs are given in incremental doses that are titrated to the desired endpoints of sedation and analgesia [8].
Access Approach
Methods
The practitioner performing the procedure should be aware that inspiratory effort is reduced with sedation; thus the kidney may be oriented cephalad compared to preprocedural imaging or access planning.
With the patient in the prone position, the laterality is confirmed and the overlying skin is prepared and draped. The skin and subcutaneous tissues overlying the intended entry site are infiltrated with local anesthetic and a small dermatotomy may be made with a scalpel. Needle access for PCN is usually obtained with sonographic guidance, fluoroscopic puncture, or varying levels of combining both methods. An 18 gauge trocar needle allows for less deflection and often has an echogenic diamond-shaped tip. The larger caliber 18 gauge needle has the ability to accept a 0.035 in. guidewire, thus minimizing the overall number of exchanges. If a 20–22 gauge trocar needle is used to access the renal pelvis, then a 0.018 in. wire and coaxial introducer catheter must be utilized prior to introducing a 0.035 in. stiff guidewire. The floppy tip of the wire reduces access trauma. Once there is enough purchase into the renal pelvis, the coaxial introducer is advanced over the guidewire and into position using the radio-opaque band to identify tip location of the sheath. Removal of the introducer and stiffening cannula permits advancement of a .038 in. diameter guidewire through the lumen of the sheath.
Usually, access to the collecting system is through a posterolateral approach with the needle directed toward a mid-to-lower pole calyx. Dryer [4] et al. describe the two tactile “pops” appreciated as the needle first traverses the renal capsule, followed by successful entry into the collecting system. Despite the access method, calyx entrance is confirmed by the chosen guidance modality and by aspirating urine. If the fluid is cloudy or there is a high clinical suspicion for infection, opacification of the collecting system may be avoided or performed with gentle caution.
Ultrasound
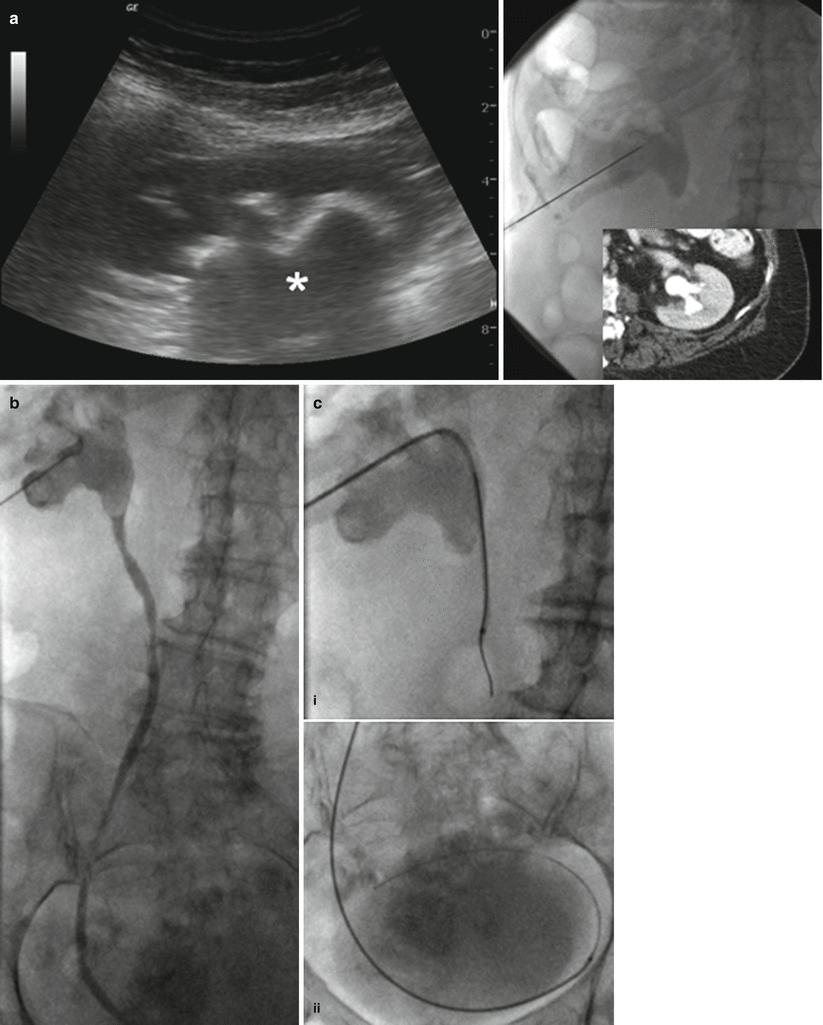
Under real-time sonographic guidance, an 18–22 gauge access needle is advanced directly into the target calyx using a “single-stick” technique. One should attempt to puncture a mid or lower pole calyx at an angle of 20–25° to the coronal plane (Fig. 19.2) [9].
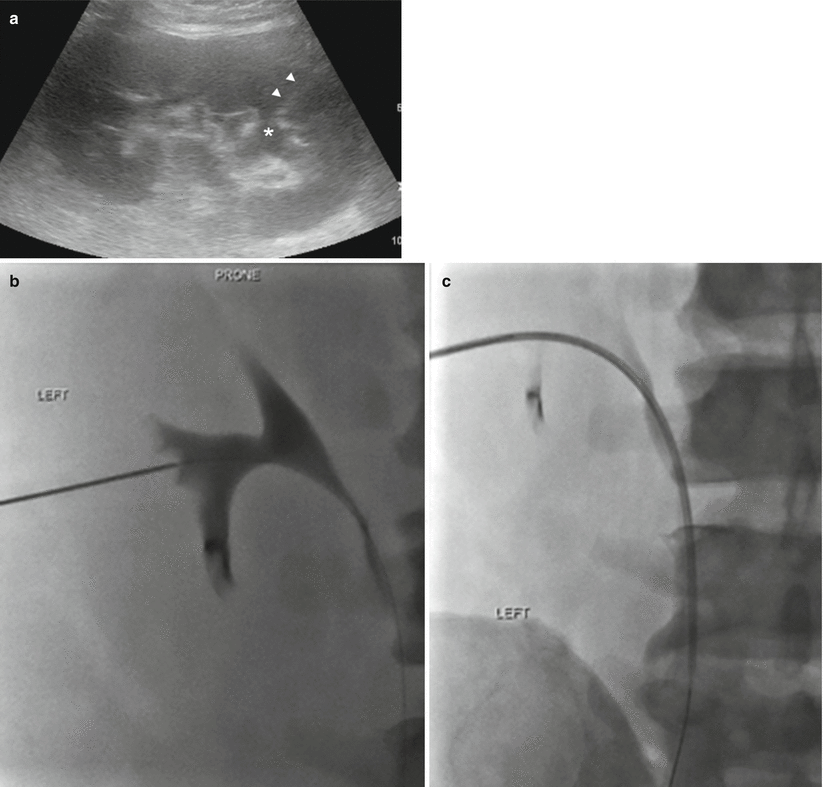
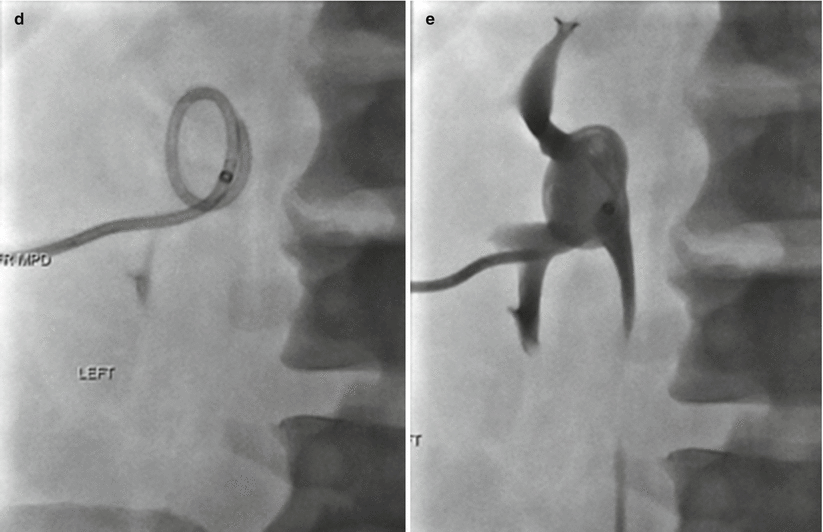


Fig. 19.2
Ultrasound-guided needle access for percutaneous nephrostomy. (a) Gray-scale ultrasound image of the left kidney. Lower pole calyx is denoted with an asterisk. Needle track is identified by the arrowheads with the intended target being a lower pole calyx. (b) Fluoroscopic images demonstrate contrast opacification of the renal collecting system. The operator proceeds with needle exchange over 0.018 in. wire for coaxial introducer sheath with contrast. (c) Subsequently, the operator passes a 0.035 in. guidewire through the coaxial introducer sheath and the lower pole calyx and down to the proximal ureter. (d) The introducer catheter is exchanged over 0.035 in. stiff wire for 8.5 French nephrostomy. Percutaneous nephrostomy drain is passed beyond the inner metal stiffener and into the ureter. The metal stiffener is stationed at the beginning of the curve of the wire and subsequently removed to allow for formation of pigtail end. The pigtail is positioned within the renal pelvis, and the pigtail loop is locked. (e) Injection of contrast confirms adequate placement of the pigtail catheter in a decompressed system, without filling defect
Access is confirmed via sonography, the tactile “pop,” and aspiration of urine. Contrast material may be injected under fluoroscopy for evaluation of calyceal location, morphology, and degree of hydronephrosis. Next, the access wire is advanced through the needle with an attempt to loop the wire within the renal pelvis which facilitates nephrostomy tube deployment. One must attempt to form the pigtail within the renal pelvis, as attempting to do so within the ureter or in a calyceal infundibulum may be difficult and traumatic.
Fluoroscopy
Fluoroscopy alone may be used to access the renal collecting system. If there is a radio-opaque calculus or indwelling ureteral stent or if the collecting system is opacified secondary to the administration of radio-opaque intravenous contrast or via retrograde injection through an existing ureteral catheter, these structures may be targeted in order to access the collecting system via a single or two-stick process. If no radio-opaque target is present, a 22 gauge needle may be advanced trans-retroperitoneal under fluoroscopy in a “blind” manner, guided by the renal outline or, if the outline cannot be seen, osseous landmarks and preoperative images [10]. Attempt to aspirate urine is performed by the operator as the needle is gently retracted. Upon aspiration of urine, the system may be opacified so the desired renal calyx may be defined and targeted with a second 18–22 gauge access needle.
Combination Method
Sonographic guidance of a 21–22 gauge needle into a renal calyx performed. Once in place, air can be injected to define the posterior calyx or the system can be opacified with contrast, in order to provide a radio-opaque target for larger needle access under fluoroscopy. There is a risk of causing an air embolus, if the needle is not within the collecting system.
CT
Although a less utilized form of guidance, a CT may be necessary in patients with variant renal anatomy, such as horseshoe or pelvic kidneys. Barbaric et al. [11] described a combine CT-fluoroscopy technique performed in a modified room where a C arm is fitted at the end of a CT table, allowing for fluoroscopy with the patient on the CT table. The CT-fluoroscopy technique was performed with the patient in the supine-oblique position, reporting no major complications and a 2 % minor complication rate.
Cone Beam/Fluoroscopic
Cone beam CT, especially with the use of needle guidance software, results in improved anatomic visualization compared to conventional CT and fluoroscopy. Newer hybrid systems can perform high-speed rotational scanning with or without simultaneous contrast injection and complete post processing within seconds. Siegel [12] describes the use of such software, allowing for the postprocessed dataset to be used to guide percutaneous interventions with the aid of interactive needle-path planning and guidance software.
Outcomes
Success rates for PCN vary depending on the clinical scenario. The success rate in dilated systems approach is 99 % at most institutions, with lower rates reported in patients with non-dilated collecting systems, complex stone disease, and staghorn calculi. In complicated cases, reported success rates drop to 85 %.
Complications
The Society of Interventional Radiology stratifies the type of complication by outcome as listed below:
1.
Minor complications:
(a)
No therapy, no consequence
(b)
Nominal therapy, no consequence; includes overnight admission for observation only
2.
Major complications:
(a)
Require therapy, minor hospitalization (<48 h)
(b)
Require major therapy, unplanned increase in level of care, prolonged hospitalization (>48 h)
(c)
Permanent adverse sequelae
(d)
Death
Minor and major complications combined occur in approximately 10 % of patients [13] with sepsis and bleeding as the two major procedure-related complications.
Hemorrhage occurring after PCN is usually self-limited and resolves in a few days. If the urine remains bloody past 72 h with frank hemorrhage, upsizing of the nephrostomy tube may be considered. If bleeding persists and the urine remains grossly bloody after 3–5 days or when there is a drop in hematocrit, patients should undergo angiographic evaluation with embolization of injured vessels [3].
Other sources of a drop in hematocrit after PCN include retroperitoneal hemorrhage and should be considered in the appropriate clinical setting, especially if the urine is clear. Suspected retroperitoneal hemorrhage is best evaluated with CT.
The reported rate of sepsis is 1–3 %, with a higher incidence in patients undergoing PCN for suspected urosepsis or pyonephrosis.
Pleural complications rarely occur (approximately 0.2 % of decompressive nephrostomies) and include pneumothorax, empyema, hydrothorax, and hemothorax.
Nephroureteral Stents/Catheter/Tubes
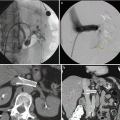
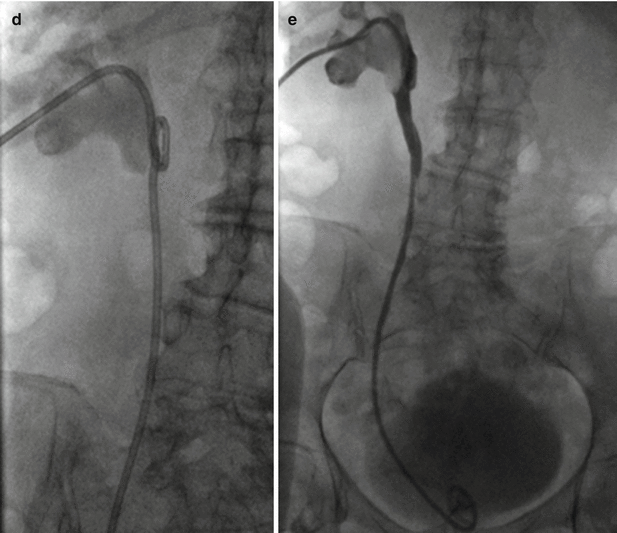
A nephroureteral catheter is a unibody multiple side-hole internal/external drainage catheter. Typically the proximal locking loop, with drainage holes, is positioned in the renal pelvis, and the distal loop, also with drainage holes, is positioned in the bladder. This unibody catheter combines an external drainage tube and ureteral stent confers several advantages. The ability to drain patients both internally and externally provides maximal drainage. Simultaneous internal and external drainage maximally decompresses the patient ensuring that obstruction to urinary flow will be minimized. This may be desirable to facilitate healing of a urinary tract injury or to rapidly normalize renal function. Internal/external access may be desirable to facilitate stone extraction (Fig. 19.3). Finally, internal/external access may improve patient quality of life. In instances where ureteral stents are unable to be exchanged cystoscopically, nephroureteral tubes can remain capped and exchanged at regular intervals. This configuration provides emergency external drainage and facilitates catheter maintenance.
Get Clinical Tree app for offline access


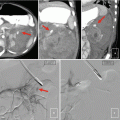
Fig. 19.3
Nephroureteral stent placement. Nephroureteral stent was placed in this patient with a staghorn calculus to provide access for percutaneous and endourologic stone extraction. (a) Gray-scale ultrasound, fluoroscopic, and CT images demonstrate needle puncture and access to kidney with large staghorn calculus (asterisk). (b) Contrast injection confirms access to the collecting system. (c) Catheter and wire are advanced to the proximal ureter (i




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree