With chest pain representing one of the most common presenting symptoms in the emergency department (ED), radiologists are frequently called upon for the noninvasive evaluation of aortic and pulmonary vascular diseases. Adequate assessment relies on the ability to understand the spectrum of the pathologic conditions and the significance of each of these entities.
Thoracic Aorta
Anatomy
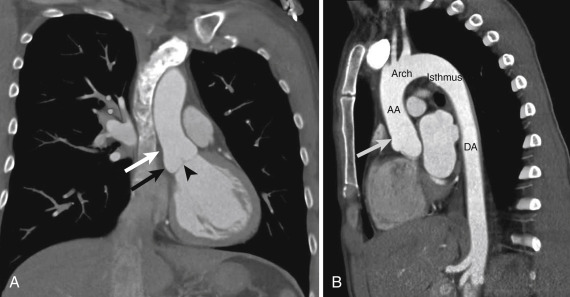
The thoracic aorta begins at the level of the aortic valve and extends to the level of the diaphragmatic hiatus ( Fig. 10-1 ). The aortic root is said to be made of three outpouchings or sinuses of Valsalva and the aortic valve with its annulus. The tubular ascending aorta begins just above the sinuses at a waist known as the sinotubular junction . At the level of the innominate or brachiocephalic artery the ascending artery gives way to the arch. Just distal to the left subclavian artery the descending aorta begins. The most proximal portion of the descending aorta is referred to as the aortic isthmus . A majority of the thoracic aorta is extrapericardial in its location. Only the first 2 to 3 cm of the ascending aorta resides within the pericardial sac.
The aortic wall has three layers. Although not distinguishable by computed tomography (CT) or magnetic resonance imaging (MRI), these layers include the intima, media, and adventitia. The media is the muscular layer in which many of the acute aortic syndromes may propagate. The inner two thirds of the media has concentric fibers that are orthogonal to the outer one third consisting of longitudinal muscle fibers. The net effect is a natural cleavage plane. If blood can access the space within the media, it can easily travel along the course of the media as either a false lumen or intramural hematoma (IMH).
Imaging
Computed tomography has remained the workhorse for aortic imaging in the ED. Its ubiquitous use comes from the proximity of many scanners to the ED, often with 24-hour technologist staffing and high speed of image acquisition. In addition, many of our surgical colleagues are quite comfortable with CT image review. The advent of multidetector computed tomography (MDCT) has only strengthened the role of CT in the evaluation of acute aortic pathologic conditions. With increased detector rows, thinner section images can be obtained at faster speeds. These thinner sections allow for higher quality multiplanar and three-dimensional reconstructions. In the modern era CT has close to 100% accuracy in depicting acute aortic pathologic conditions. Neither echocardiography nor MRI is more accurate.
Our nontraumatic aortic CT protocol begins with a noncontrast acquisition. The noncontrast images are helpful for looking for high attenuation within the wall, indicative of IMH. The IMH should be higher in attenuation than the blood pool, but not as high as calcium. It usually involves only a portion of the wall, not 360 degrees. The images are acquired as 3-mm thick images with a 3-mm reconstruction interval to avoid any gap. The noncontrast images are followed by a CT angiogram using bolus tracking software and a rapid injection rate of 4 to 5 mL/sec. These images are reconstructed as 1- to 2-mm thick images with 1- to 2-mm reconstruction intervals. A noncontrast study should not be used to exclude acute aortic pathologic conditions. If the patient cannot receive intravenous contrast, a noncontrast study can be performed to exclude aortic rupture, but it should be followed by another test, either echocardiography or MRI.
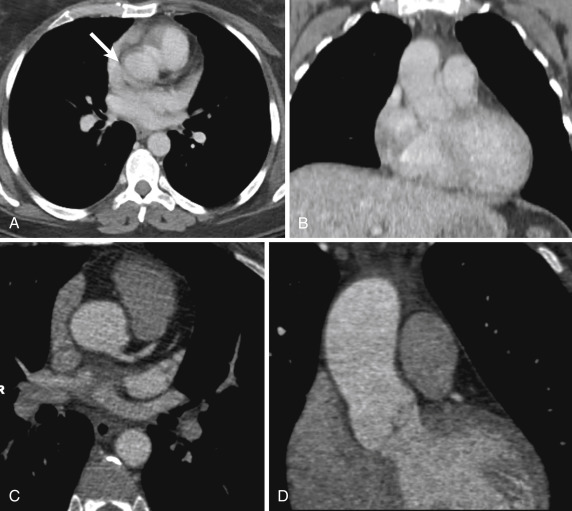
Although MDCT allows for electrocardiogram (ECG) gating, it is not routinely used in our ED for assessment of the aorta. With fewer than 4% of these studies being positive for aortic pathologic conditions, the potential for increased radiation and the added technical prowess of ECG gating have not been justified in our practice. Instead, we have relied on ECG gating as a problem-solving tool once an aortic pathologic condition is encountered. If a question arises as to whether the ascending aorta is truly involved by a process, the patient may be rebolused and rescanned using ECG gating ( Fig. 10-2 ). With the advent of dual-source CT, we have also begun to rely on ultrahigh-pitch imaging (pitch of 3.2) for the evaluation of the ascending aorta. With its lower radiation dose profile, this technique has shown greater promise in evaluating the ascending aorta on a more regular basis.
Oblique sagittal reconstructions are created directly at the scanner for all aortic CTs. The candy cane reconstruction allows for a close approximation of an angiogram view and allows for clear delineation in the z-axis of any aortic process. Volume-rendered and shaded surface images are rarely performed in real time. Although the images can be very artistic, they rarely add to the diagnosis.
Magnetic resonance imaging is increasingly being used as a secondary study when the patient is unable to receive iodinated contrast material or if a question arises from the CT. With technologic improvements and improved MR angiography techniques, MRI offers a nonradiation alternative. Unfortunately, distance from the ED and longer examination times have kept MRI as a second-line test at many institutions. Although gadolinium may allow for images that appear more similar to CT, contrast material is not required for diagnosis of acute aortic conditions. This allows for the potential of noncontrast MRI in the evaluation of the aorta in patients in renal failure. An added benefit of MRI is the use of black-blood sequences and fat-suppressed T2-weighted or short tau inversion recovery (STIR) images. These sequences can be useful for the evaluation of increased signal within the aortic wall, which may be indicative of an IMH and may help clarify a subtle CT finding.
No discussion of aortic imaging in the ED would be complete without discussing conventional radiography and angiography. Conventional radiography is useful when findings are positive because it allows the team to mobilize while support lines are being placed before CT. Radiography findings include widening of the mediastinum, effacement of mediastinal lines, and mass effect on the trachea and the left mainstem bronchus. A majority of the time the radiography signs can be subtle. The most useful finding is change in the mediastinal contour compared to a prior radiograph. A negative radiograph should not be used to exclude acute aortic pathologic conditions.
Angiography was once considered the gold standard for the evaluation of the acute nontraumatic aorta. In the current era, angiography is mainly used in the deployment of endovascular stents.
Mimics and Pitfalls on Computed Tomography
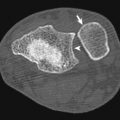
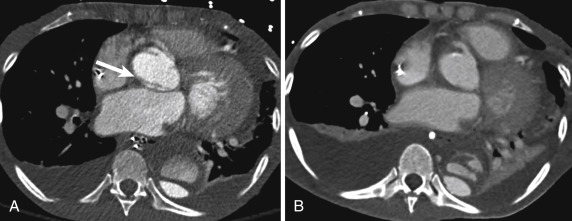
Artifacts and mimics of aortic disease are more commonly encountered than real aortic pathologic conditions. To avoid sending a patient for needless surgery, one must be comfortable with some of the more common mimics of acute disease. The most commonly encountered artifact is pulsation of the aorta at the root ( Fig. 10-3 ). This pulsation artifact can simulate a dissection flap at the root. Despite advances in CT technology, pulsation is still encountered. Clues to pulsation artifact include visualization of a flap in the pulmonary artery at the same level, lack of pericardial effusion or aortic wall thickening, and an ill-defined craniocaudal extension on multiplanar imaging. Although pulsation can usually be excluded, sometimes an echocardiogram or a repeat CT with ECG gating or ultrahigh-pitch imaging is required.
Other artifacts may stem from other diseases, such as a vasculitis or an adjacent neoplasm that may simulate aortic wall thickening or hematoma. The presence of lymphadenopathy can be helpful in suggesting these other entities.
Aortic Aneurysms
An aneurysm can be defined as failure of tapering of the aorta or even dilatation of the distal aorta. In the ascending aorta 4 cm is often used as the minimal size of an ascending aortic aneurysm; in the descending aorta, 3 cm is the minimal size. Measurements must be performed orthogonal to the long axis of the aorta. Occasionally this can be performed on transverse images, but usually these measurements require multiplanar reconstructions. Although patients with known aneurysms are often followed to determine a rate of change (if any) over time, in the ED the radiologist is not often afforded the luxury of a prior study for comparison. It becomes imperative then that certain imaging signs are used to determine when a thoracic aneurysm is unstable.
One of the most important findings is the shape of the aneurysm. Most aneurysms can be described as fusiform (diffuse concentric enlargement) or saccular (eccentric outpouching). Although fusiform aneurysms can become unstable, saccular outpouchings are more often indicative of a pseudoaneurysm that has the potential of being unstable. Most saccular aortic aneurysms are indicative of infection, traumatic injury, or a postoperative complication, such as dehiscence of a suture line. The one major exception is the pseudoaneurysm from a penetrating atherosclerotic ulcer (PAU; see later discussion).
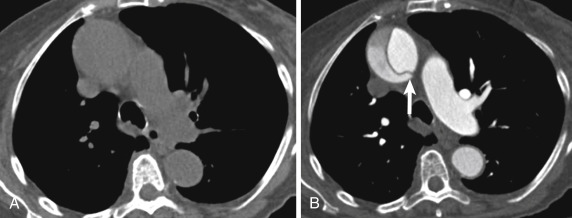
Other imaging findings of an unstable aneurysm include high attenuation within the wall (sometimes referred to as the impending rupture sign ) ( Fig. 10-4 ), irregular shape, calcium that is somewhat tangential to the arc of the aorta, and periaortic stranding. All of these signs require careful attention. Usually more than one of these are present. The impending rupture sign and tangential calcium may be easier to see on noncontrast images.

Rarely, high attenuation indicative of blood may be seen in the mediastinum and in the pleural space (hemothorax). More commonly, mild fat stranding is seen with a low-attenuating pleural effusion. This transudate is felt to represent a sympathetic effusion. In fact, it is very unusual to see an unstable aorta without a left pleural effusion. Care must be taken to not mistake the concomitant left-sided atelectasis, which often briskly enhances, for contrast extravasation. In the past the visualization of blood would result in termination of the scan and immediate contact with the surgeon. In the era of endoluminal therapy the presence of blood in the mediastinum or pleura results in immediate contact with the surgeon, but often the postcontrast images are continued because they are key in planning appropriate therapy. Even more rarely, extravasation of intravenous contrast may be seen. Of all the signs, this one has the gravest prognosis.
Acute Aortic Syndromes
Over the past decade the term acute aortic syndrome has come into use as an aortic equivalent of the acute coronary syndrome as an explanation for an aortic cause of chest pain. Although some authors include unstable aneurysms in this grouping, most reserve the term for the spectrum of aortic medial pathologic conditions, namely, aortic dissection, IMH, and PAU. These entities are rare but can be lethal and require a high index of suspicion. Because CT is critical in the diagnoses of these entities, every radiologist working in the ED must be familiar with their imaging appearances.
Aortic Dissection
Aortic dissection represents the most common of the acute aortic syndromes to present to the ED and the most common to result in premature death. Simply put, an aortic dissection represents a breach of the aortic intima with access of blood to the media. As described earlier, the inner two thirds of the media travel circumferentially within the wall, whereas the outer one third of the media travels longitudinally parallel to the long axis of the aorta. If flowing blood can gain access to the plane between the two orthogonally directed fibers, a false passage can easily be created. Entities that either weaken the medial wall or exaggerate the space between the planes of the media can predispose to dissection as can those conditions that increase aortic luminal pressures. These include conditions associated with cystic medial necrosis (such as Marfan syndrome or bicuspid aortic valve), aortic hypertension, and conditions that transiently elevate aortic pressures, such as pregnancy, cocaine use, and weight lifting. Atherosclerotic disease may increase the risk for dissection by way of a PAU but does not usually directly result in dissection.
Once flowing blood gains access to the media, two lumens are created (a true lumen corresponding to the actual aortic lumen and a false lumen) ( Fig. 10-5 ). The wall that separates the two channels is often referred to as an intimal flap , though technically it is an intimomedial flap (because it contains the intima and the inner two thirds of the media).
Accurate delineation of the true from the false lumen is key on cross-sectional imaging in case surgical treatment is required. In the ascending aorta the true lumen is usually contiguous with the aortic valve orifice and is the smaller of the two lumens. The true lumen tends to be on the left of the false lumen (closer to the pulmonary artery). When a fenestration is present in the ascending aorta, the flaps on both sides of the defect point away from the true lumen, a finding sometimes referred to as the intimomedial rupture sign . In the arch and descending aorta the most reliable way to distinguish the true from the false aortic lumen is the ability to directly trace the suspected true lumen to the intact “normal” lumen of the proximal aorta. Other findings include the size, because the true lumen is usually smaller than the false lumen, and the presence of calcifications, which are usually in the intima surrounding the true lumen. Care must be used in chronic dissections because the false lumen can occasionally calcify. In the descending aorta the true lumen is usually anterior and to the right of the false lumen. Although variable, classically the celiac artery, superior mesenteric artery, and inferior mesenteric artery arise from the true lumen, and the left renal artery tends to arise from the false lumen. As a result, left renal ischemia with resultant left flank pain may be the presenting symptom of an acute aortic dissection. Another helpful finding in the false lumen is the presence of thin strands, representing fibers of the media that remain as blood has dissected in this false space. These are occasionally referred to as aortic cobwebs and are indicative of the false lumen. The angle created by the flap and the outer aortic wall can also be used for separating the true from the false lumen. Generally, the angle is more acute in the false lumen, a finding sometimes known as an aortic beak or beak sign (see Fig. 10-5 ).
A mainstay of aortic dissection treatment is blood pressure control, but clearly some patients require surgical intervention. The location of the dissection flap is the main determinant. Classification of a dissection is usually based on the location of the flap. Two main categorization schemes include the Stanford and DeBakey classifications. At our institution, we use the Stanford classification, in which involvement of the ascending aorta is considered a Stanford type A dissection, requiring surgery. Arch and descending aortic involvement alone are considered Stanford type B dissections and may be amenable to medical therapy alone. Of note, although some authors have previously described the left subclavian artery as the landmark for delineation between Stanford A and Stanford B, increasingly ascending aortic involvement is used. In other words, only when the ascending aorta is involved is the dissection considered type A. In the DeBakey scheme, ascending aortic and descending aortic involvement is considered type I, ascending involvement alone is considered type II, and descending aortic involvement alone is considered type III.
Ascending aortic involvement prompts surgical intervention because of the risk for acute aortic valve regurgitation, coronary artery involvement, or tamponade from hemopericardium. The risk for death from the dissection exceeds the potential risk from surgery (including paralysis). Although most isolated descending aortic dissections are considered medical lesions, occasionally these dissections can become surgical. Potential rupture of the false lumen and end-organ ischemia may prompt either surgical or endoluminal therapy. A concise report therefore must include signs of unstable false lumen. These are exactly the same as those for the unstable aneurysm described earlier. The report should also include information about end-organ ischemia and the nature of the ischemia. If the ischemia is from propagation of the flap into the vessel for that organ, the patient may be stented. If the flap occludes the orifice of the feeding artery without directly involving it, the patient may undergo fenestration at that level.
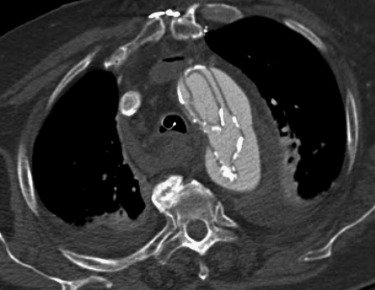
Most aortic dissections will present with a linear filling defect in the aortic lumen that corresponds to the intimomedial flap. Occasionally the aortic dissection can take on an unusual appearance. Rarely the dissection will involve 360 degrees of the aorta so that the false lumen completely surrounds the true lumen. In this instance the intimomedial flap may become free of the remainder of the aorta and form an irregular filling defect. Occasionally this circumferential flap will begin to infold or intussuscept similar to bowel intussusception. This unusual dissection is referred to as an intimointimal intussusception ( Fig. 10-6 ). In another variation a dissection involving the ascending aorta may rupture just above the valve plane into a shared sheath with the pulmonary artery. The blood in the wall of the pulmonary artery will compress the pulmonary artery and may occlude it. Sometimes the blood will dissect around the pulmonary artery to the segmental arterial level. A third unusual manifestation of aortic dissection is a focal dissection at the level of the aortic root near the sinotubular junction. This localized process has been referred to as an incomplete aortic dissection and is almost always associated with cystic medial necrosis and aortic root enlargement. This incomplete dissection can mimic pulsation artifact. As a general guideline, one should have a low threshold for reimaging the aortic root when a linear defect is encountered in the setting of annuloaortic ectasia or in a patient with known Marfan syndrome or bicuspid valve.
Focal dissections of the abdominal aorta are rare in the absence of prior trauma or aortic catheterization. However, imaging of an aortic dissection should include the abdominal aorta and the iliac vessels to the level of the common femoral arteries. Inclusion of the abdomen and pelvis is performed to look for any end-organ ischemia and to delineate the extension of the dissection. Increasingly surgeons are relying on peripheral cannulation for initial bypass in aortic repair. The role of imaging is to help distinguish true from false lumen so that any peripheral cannulation for bypass avoids cannulating the false lumen, which would preclude adequate bypass and could potentially be lethal.
Intramural Hematoma
Intramural hematoma refers to blood within the aortic media that does not have a communication with the aortic lumen. In other words, no fenestration is noted within the intima. The effect on imaging is that the aortic wall will be eccentrically thickened with blood, but the thickening will not enhance with intravenous contrast. The blood will result in high attenuation on CT (usually better seen on noncontrast images) or high signal on black-blood MRI ( Fig. 10-7 ). In an idiopathic IMH the blood is believed to arise from rupture of the vasa vasorum into the media. An IMH may also be secondary to a PAU (see next section).
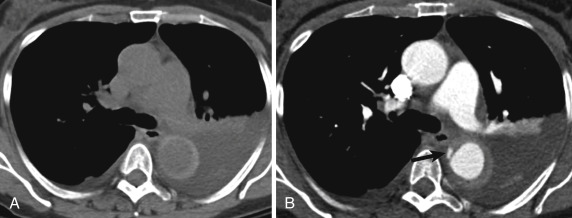
As with dissections, IMH is associated with hypertension and cystic medial necrosis. Patients tend to be older than dissection patients. An IMH may resolve with medical therapy, but it may also progress into a true aortic dissection. If the pressure in the media exceeds luminal pressure, the media may create a fenestration with the true lumen. This process has been sometimes referred to as a reverse dissection , although the imaging will mimic a true aortic dissection that begins on the luminal side. Rarely the IMH will rupture. As with the other entities, signs of impending rupture include irregular shape of the IMH and adjacent fat stranding. As with dissection, an IMH may rupture into the shared sheath with the pulmonary artery. This event can result in a very unusual appearance of the pulmonary arteries and can be quite confusing to the unaware reader.
Because of the potential for progression of an IMH to dissection or rupture, an IMH is often treated similarly to aortic dissection. Many centers will classify an IMH with the Stanford or DeBakey classifications described in the prior section. Intramural hematoma involving the ascending aorta is usually treated surgically, whereas descending aortic IMH is often treated medically. Literature mostly coming from Asia has suggested that in certain patients there may be a role for close observation of a patient with an ascending aorta IMH instead of surgery. These patients may do as well (if not better) than patients undergoing median sternotomy. Other literature has suggested that thickness of the IMH in the ascending aorta or presence of an ulcer may predict which patients are at increased risk for rupture. These features of IMH are currently under investigation. For now the ED radiologist should be aware that most patients with ascending aortic involvement will undergo surgical repair, but that a minority fitting certain criteria may be closely observed instead.
Occasionally a contrast blush may be seen within an IMH (see Fig. 10-7 ). This may arise from the lumen and a tiny ulcerlike projection or from retrograde flow from an intercostal artery. These blushes can lead to confusion in the categorization of which entity of acute aortic syndrome is present. The key is to clearly report whether the ascending aorta is involved and if possible, why the blush is present. Some of the literature has shown that a blush from an ulcerlike projection is at increased risk for progressing to a true aortic dissection. These patients may be more closely followed with subsequent imaging.
Penetrating Atherosclerotic Ulcer
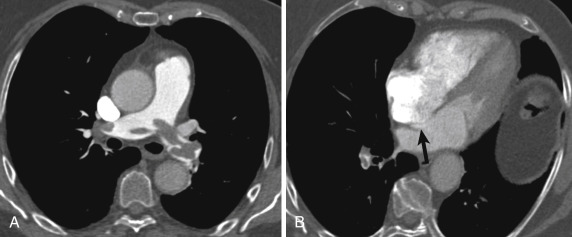
The PAU is the newest entity to have been described, with many of the early papers appearing in the late 1980s. The PAU represents ulceration of an atherosclerotic plaque so that blood from the lumen now has access to the aortic media. The belief is that the macrophages within the plaque weaken the intima and allow for this focal ulceration. The key from an imaging perspective is to make sure that the ulceration truly penetrates through the intima and does not simply represent an ulcerated plaque without an intimal breach. From an imaging perspective, most PAUs will have an eccentric outpouching of the outer wall (saccular aneurysm; Fig. 10-8 ).
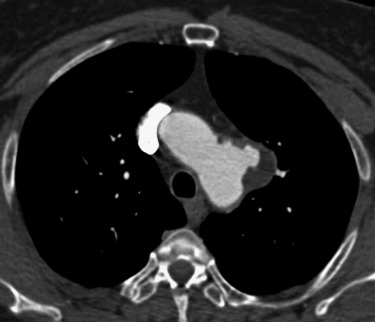
Unlike aortic dissection and IMH, PAUs have a high association with aortic atherosclerosis. Patients with PAUs tend to be older and tend to have a greater atherosclerotic burden. PAUs may spontaneously resolve, but they may also present with an IMH. Because of the intimal breach, flowing blood in a PAU will occasionally create an aortic dissection. Rarely a PAU may go on to rupture. Signs of rupture are similar to those of the unstable aneurysm. The treatment goal in a patient with PAU is to make the patient free of chest pain. If the patient has persistent pain, there is increased risk for IMH, dissection, or rupture. These patients will be closely followed and if possible, may undergo endoluminal treatment. Location of the PAU and the size of the neck will play a role in determining which patients will receive endoluminal repair.
Aortic Thrombus
Occasionally a patient with chest pain will undergo CT and a large filling defect will be seen within the aorta ( Fig. 10-9 ). This acute aortic thrombus is increasingly being recognized as an embolic source. The thrombus is believed to arise from an area of intimal irregularity. Some authors have postulated that a scar from the ductus arteriosus may serve as a nidus for thrombus formation. Others have suggested that differential flow may promote an area of relative stasis along the lesser curve of the aorta at the level of the isthmus.
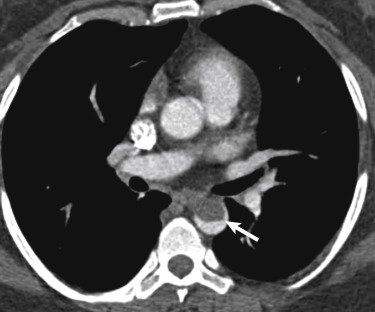
The thrombus itself should not present with chest pain, but emboli from the clot may result in symptoms of abdominal or extremity pain. These filling defects are usually encountered within the aorta at the level of the isthmus and within the descending thoracic aorta. They should not enhance with intravenous contrast, but when they are chronic, areas of recanalization may create pseudoenhancement if they are included in measurements of attenuation on CT or signal on MRI. Radiologists should be aware of the acute aortic thrombus to avoid confusion with an aortic tumor. The latter almost always has soft tissue that extends beyond the aortic wall. Treatment for an acute aortic thrombus begins with anticoagulation but may include a thrombectomy and rarely endoluminal stent placement to trap the thrombus.
Aortic Fistula
Another rare manifestation of aortic disease in the ED is an aortic fistula. Unfortunately, the imaging of an aortic fistula can be quite subtle despite the high lethality of this entity. Clinically the course of a patient with a fistula may be tricky as well. These patients usually report a large episode of hematemesis or rectal bleeding (the herald bleed). Because the fistula is only intermittently patent, the bleeding subsides, so the patient is hemodynamically stable enough to present to the ED and undergo cross-sectional imaging.
Aortic fistulas can be primary, usually from rupture of an aortic aneurysm or in the setting of an infection, such as mediastinitis. When findings of mediastinitis are encountered (such as gas or diffuse mediastinal fat infiltration), careful inspection of the aortic contour is required. Any subtle outpouching of the aorta may be indicative of the development of a fistula ( Fig. 10-10 ). Patients with aortic fistulas from mediastinitis tend to have less of an atherosclerotic burden than those with PAUs. Rarely an aortic aneurysm may rupture and fistulize to an adjacent organ. This phenomenon is more common in the abdomen and has been reported with adjacent bowel and the inferior vena cava.
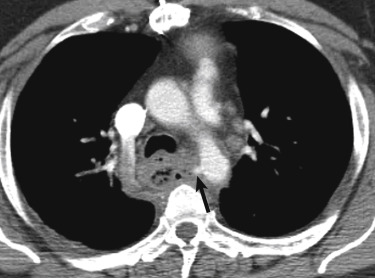
An aortic fistula is more likely to result as a complication from prior surgery. In the thorax the fistula may be to the esophagus or an adjacent bronchus (usually the left main bronchus). Fistulas may follow either open or endoluminal surgery. Clues to the diagnosis rest on seeing gas next to the repair site, irregular contour of the aorta, and clinical history. Sometimes endoscopy is used to confirm a suspected fistula.
Pulmonary Embolism
The pulmonary arteries serve as the main conduit of blood from the right ventricle to the lungs and serve as the main path of oxygenation of the circulatory system. They begin as a main pulmonary artery that bifurcates into a right pulmonary artery, which travels slightly posterior and inferior to the right main bronchus, and a left pulmonary artery, which travels superior to the left main bronchus. The arteries continue to divide into lobar, segmental, subsegmental arteries, eventually reaching a microscopic capillary level. The result is that these smallest of vessels serve as a net to stop any emboli from gaining access to the left heart and the systemic (aortic) circulation. In other words, the pulmonary arteries should be the final recipient of emboli from the venous system to prevent systemic complication. Many types of embolic disease can manifest within the lungs. In thromboembolic disease the thrombi form in peripheral veins from an antecedent condition (e.g., long period of sitting still, hypercoagulability) and then dislodge to the lungs.
When acute, the embolism may result in right heart failure and even death. Usually the acute emboli go unnoticed clinically. In fact up to 3% to 5% of outpatients may have asymptomatic embolic disease. If the emboli result in a cascade of cytokine-mediated scarring in the pulmonary arteries, pulmonary pressures may elevate, and the result is chronic pulmonary emboli (PEs) or chronic thromboembolic pulmonary hypertension. This is occurs in about 1% to 3% of patients with acute embolic disease. Sometimes the embolized thrombus is infected. These septic emboli are unlikely to result in right heart failure (because the burden is small), but they tend to be markers of a bacteremic patient.
Not all emboli arise from embolized thrombi. In trauma, bone marrow may be embolized, as can amniotic fluid in the setting of a difficult delivery. These latter two entities result in a microvascular disease with characteristic CT appearance. Other emboli include tumor and iatrogenic material. As imaging for suspected emboli increases, radiologists must be familiar with the entire spectrum of embolic disease.
Acute Pulmonary Embolism
Acute PE represents the third most common cause of cardiovascular death in the United States, following stroke and myocardial infarction. As with the acute aortic syndromes and the other entities previously described, clinical history is only modestly helpful. Patients with PE range from being asymptomatic to asystolic. The nonspecific clinical presentation has resulted in the proliferation of imaging that any radiologist with even one night in the ED can appreciate.
Computed tomography has become the main imaging technique for acute PE. Since its introduction in the 1990s, CT has risen to become the dominant modality for imaging of acute PE, especially in the ED. The popularity of CT for PE, as with acute aortic syndromes, rests on its availability in most EDs and its rapid turnaround times. In the emergency setting, ventilation-perfusion ( <SPAN role=presentation tabIndex=0 id=MathJax-Element-1-Frame class=MathJax style="POSITION: relative" data-mathml='V˙/Q˙’>V˙/Q˙V˙/Q˙
V ˙ / Q ˙
) scintigraphy has taken a backseat to CT, owing largely to the limited availability of technologists and the longer examination times. Another explanation for the dominance of CT is its ability to diagnose alternative conditions. In many centers only 5% to 10% of CT scans for PE will be positive for PE. Up to one third may reveal another explanation for the patient’s presentation. Often these alternative diagnoses are not possible by <SPAN role=presentation tabIndex=0 id=MathJax-Element-2-Frame class=MathJax style="POSITION: relative" data-mathml='V˙/Q˙’>V˙/Q˙V˙/Q˙
V ˙ / Q ˙
scan.
Computed tomography findings of PE rely mostly on direct visualization of the PE ( Fig. 10-11 ). These include a central filling defect that usually expands the vessel. Contrast may be seen between the embolism and the wall. When the vessel with embolism is cut in cross-section, it is referred to as the rim sign , and when the vessel is in plane, the surrounding contrast is sometimes called a tram-track sign . Key in diagnosing an acute PE is to remember that every bronchus should have a corresponding artery. This point is key in preventing confusion of a vein for an artery and key in detecting an occluded artery. Another potential pitfall in the diagnosis of an acute PE is the confusion of a mucoid impacted bronchus for PE. Again, if the radiologist remembers that a bronchus should accompany an artery, he or she will realize that the enhancing structure is the artery and the apparent adjoining filling defect is actually in the bronchus.