18 On the Horizon: Going Beyond Conventional MR Contrast Agents
18.1 Why We Need More Than What Is Available
In patients with brain tumors, contrast-enhanced magnetic resonance imaging (MRI) with gadolinium (Gd)-based agents is essential for characterization of disease extent, planning surgical resection, delineating appropriate radiosurgical target volumes, and following patients for disease recurrence. 1 , 2 Effective management of patients with brain tumors depends on accurate detection and delineation of lesions. Enhancement of intra-axial brain tumors using Gd chelates is based primarily on disruption of the blood–brain barrier and/or abnormal vascularity, which allows the agent to accumulate within the lesion. 3 Contrast enhancement thus improves the demarcation of pathological tissue from normal brain.
Several factors affect the degree of signal enhancement of a lesion from Gd chelates, including magnetic resonance (MR) field strength, sequence parameters, and individual properties of the contrast agent itself, as well as the dose and concentration of the agent. The ideal MR contrast agent would perfectly delineate the margins of the tumor, thus showing its size and extent and clarifying its relationships with other structures; moreover, it would enable tumor enhancement to be distinguished from treatment-related enhancement. The ideal MR contrast agent would also make it possible to obtain reliable information on tumor grade based on assessment of the microvasculature with functional techniques. However, the intrinsic infiltrative nature of some brain tumors, particularly glioblastomas, makes it a real challenge to reliably delineate their margins and extent. 4
Chelate-bound Gd ions (Gd3+) were first used for MRI purposes in the early 1980s, and many Gd-chelate contrast agents for MR brain imaging are now commercially available. 5 , 6 Contrast agents are used in approximately one third of all MRI scans of the central nervous system. 7 Gd-based agents can be classified based on molarity and protein-binding capacity. The most widely used contrast agents are gadopentetate dimeglumine (Gd-DTPA), gadoteridol (Gd-HP-DO3A), gadoterate (Gd-DOTA), gadodiamide (Gd-DTPA-BMA), gadoversetamide (Gd-DTPA-BMEA), and gadobenate dimeglumine (Gd-BOPTA). These are water soluble, extracellular, non-tissue-specific compounds that do not bind to proteins (except Gd-BOPTA, which has transient weak protein-binding properties). 5 These extravascular contrast agents are all supplied at a standardized concentration of 0.5 mmol/mL. Although their molecular structures differ, these small-molecular-weight compounds have similar pharmacokinetic profiles and physicochemical properties that result in their quick diffusion from the blood into the extracellular space for both normal and pathological tissues and transient accumulation in areas with an abnormal blood–brain barrier, leading to faster T1 relaxation times and demonstrable contrast enhancement. 8
Extravascular contrast agents have similar T1 relaxivities (4.3–5 L/mmol/s) and are routinely given at a standard dose of 0.1 mmol/kg body weight. Numerous studies have shown that the enhancement characteristics of these non-protein-binding contrast agents are virtually indistinguishable at this dose. 9 , 10 , 11 , 12 , 13 , 14 , 15 , 16 , 17 Another extravascular contrast agent, gadobutrol (Gd-BT-DO3A-butriol), is a double-concentrated, nonionic macrocyclic Gd chelate with potential advantages for clinical imaging. 18 , 19 The low osmolality and viscosity of gadobutrol make it possible to double the concentration of Gd chelate. 20 Recent studies have shown that the higher relaxivity of gadobutrol in comparison with other non-protein-binding Gd-based agents can improve enhancement in different ways, increasing in vivo T1 shortening per injected volume at the standard dose. 21 , 22 , 23 , 24
On the other hand, the protein-binding properties of a Gd-based contrast agent can also affect brain tumor contrast enhancement substantially. As the protein-bound fraction of contrast agent increases, the time that the contrast material remains inside blood vessels also increases. This increased intravascular availability improves tumor enhancement. Previous studies have demonstrated that even the weak protein-binding properties of gadobenate dimeglumine provide better tumor enhancement, resulting in superior lesion depiction and delineation, compared to non-protein-binding agents such as gadopentetate dimeglumine or gadoterate dimeglumine. 8 , 9 , 10 , 11 , 12
To overcome the limitations of extravascular contrast agents, including rapid clearance from the blood pool and consequent suboptimal reduction of T1 relaxation times in blood, new, intravascular contrast agents have been designed for MRI (Table 18.1). 25 Examples include Gd-based molecules with significantly stronger protein binding and various macromolecular contrast agents (MMCAs), including superparamagnetic iron oxide nanoparticles. Experimental studies and early clinical trials have shown the benefits of intravascular contrast agents, including increased sensitivity and diagnostic accuracy.
Conventional MR contrast agents | Newer MR contrast agents | |
Examples |
|
|
Paramagnetic agent | Gd | Gd, irona |
Molecular weight (kDa) | < 1 | > 10–30 |
Distribution | Interstitial, extravascular | Intravascular, intracellularb |
Plasma half-life (hours) | 1.5 | > 14 |
Excretion | Renal | Renal/MPSc |
Mechanism of enhancement | Equal distribution throughout extracellular space with abnormal blood–brain barrier; nonbinding to serum proteinsd; untargeted agents | Intravascular and extravascular enhancement; capable of binding to serum proteins; targeted agents |
Dosage (mmol/kg body weight) | 0.1–0.3 | 0.03e |
T1 relaxativity (R1; mmol/s) | < 5 | > 19 |
MR perfusion imaging | First-pass imaging | First-pass imaging, steady-state imaging |
Advantages | Determination of disease extent; planning surgical resection; delineation of radiosurgical volumes | Greater tumor enhancement; persistent high intravascular enhancement; better delineation of microvessels; long window scan imaging; lower dosage; convection-enhanced delivery of drugs using liposomes |
Drawbacks | Vascular half-life relatively short; enhancement only a few minutes after administration | Most remain in preclinical stage |
aIron oxide nanoparticles and some liposomes. bUltrasmall superparamagnetic iron oxide (USPIO) particles. cIron oxide nanoparticles accumulate into the mononuclear phagocytic system (MPS). dTransient weak protein-binding properties for gadobenate dimeglumine. eGadofosveset trisodium. | ||
Gadofosveset trisodium (MS-325, Ablavar, Lantheus Medical Imaging, North Bilerica, MA), an intravascular contrast agent, forms reversible noncovalent bonds with albumin. It has relatively low relaxivity in its unbound state, but unbound gadofosveset accounts for only about 15% of the compound in human blood; the remaining 85% is bound to albumin (Fig. 18.1). 26 This slows the tumbling rate of the complex, resulting in a four- to fivefold increase in blood relaxivity at 1.5 T in comparison with extracellular Gd-based agents. 27 In addition, like MMCA, gadofosveset has a long terminal half-life and remains confined to the vascular pool. Furthermore, there is a high concentration of albumin in the interstitial space of gliomas, and it is reasonable to expect that gadofosveset will bind to this albumin in addition to the albumin in blood, leading to greater lesion enhancement. 28 Recently, improved enhancement with gadofosveset trisodium in comparison with extravascular agents has been demonstrated in a variety of human brain tumors. 29 , 30

Detailed characterization of the tumor vasculature provides a better understanding of the complex mechanisms associated with tumor growth and is especially important in monitoring therapy. In this direction, experimental and some early clinical studies have shown the potential of MMCAs for characterizing tumor angiogenesis by dynamic contrast-enhanced MRI based on the ability to assess microvessel permeability and fractional plasma volume. Although most MMCAs are still under development, these contrast media will potentially enable qualitative and quantitative, physiologically based imaging evaluation of tumor microvessels and the response to angiogenesis inhibitors. Although many are not yet approved for clinical use, they do show promise for brain tumor imaging applications.
This chapter reviews several groups of the newest MR contrast agents for brain tumor imaging. We discuss the advantages and limitations of each group, using examples from our experience and from the literature, with the understanding that newer contrast agents are needed to overcome the current limitations and improve our diagnostic capabilities.
18.2 Blood Pool Agents
Gd-based agents can be classified based on molarity and protein-binding capacity. The typical extravascular contrast agents have similar pharmacokinetic and physicochemical profiles that result in their transient accumulation in areas with an abnormal blood–brain barrier. Extravascular contrast agents have similar T1 relaxivities (4.3–5 L/mmol/s) and are given at a standard dose of 0.1 mmol/kg of body weight. They exhibit rapid clearance from the blood pool, which results in unsatisfactory reduction of T1 relaxation times in blood. To overcome this problem, new protein-binding Gd compounds have been designed for use as intravascular blood pool agents.
Although not currently approved for brain tumor imaging in the United States, gadofosveset is the first blood pool agent approved for clinical use with MR angiography in the European Union, Canada, and the United States. Gadofosveset binds strongly but reversibly to albumin in plasma, leading to higher relaxivity (4 or 5 times higher) and better vascular retention, resulting in longer intravascular half-life (~16 h vs. ~90 min). The diagnostic accuracy of gadofosveset-enhanced MRI for evaluating vascular stenoses and aneurysms of abdominal vessels in patients with aortoiliac occlusive disease is similar to that of conventional angiography. 31 A recent report showed that gadofosveset MR angiography is also useful in the follow-up of intracranial aneurysms. 32
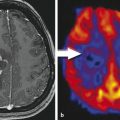
The usefulness of gadofosveset trisodium in imaging glioblastomas has recently been evaluated. Puig et al 30 demonstrated advantages for gadofosveset over gadobutrol, an extravascular contrast agent, for imaging histologically proven glioblastoma in a clinical setting. Interestingly, in the subjective analysis, readers rated images obtained 6 hours after gadofosveset administration more useful for diagnosis, and this impression was confirmed by the increases in quantitative enhancement parameters (Fig. 18.2). The greater enhancement seen 6 hours after gadofosveset injection may be attributable to greater accumulation in the interstitial fluid of tumors due to the so-called trapping effect of contrast molecules: after unbound gadofosveset molecules pass through the impaired blood–brain barrier, they bind with albumin within the tumor, resulting in strongly reduced backflow rates and increased relaxivity. These findings are in line with previous preclinical studies performed using rat brain glioma models, which concluded that the pharmacodynamic effect of protein binding increases the relaxivity of the contrast agent, thereby increasing the MR signal. Adzamli et al 33 found two- to threefold stronger enhancement with albumin-binding agents (including gadofosveset) compared to the nonbinding extravascular contrast agent gadoversetamide, on a per mole basis. Furthermore, Wintersperger et al 34 demonstrated that the protein-binding capacity of experimental gadolinium chelates (bound fraction of ~50–90%) increased contrast-to-noise ratio and contrast enhancement in rat brain glioma in comparison with the non-protein-binding agent gadopentetate dimeglumine and the weak protein-binding agent gadobenate dimeglumine. This is important because gadobenate dimeglumine has been extensively evaluated in brain tumors, and significantly greater lesion-to-brain contrast has been demonstrated in multiple clinical trials and in experimental models in comparison with other extravascular agents. 7 , 35 , 36 , 37

The more pronounced and prolonged enhancement achieved with gadofosveset trisodium that results in improved diagnostic end points can be attributed to the markedly greater T1 relaxivity of this agent at 1.5 T (19 ± 1 L/mmol/s compared with 4.7 ± 0.2 L/mmol/s for gadobutrol). 18 , 33 The relaxivity of gadofosveset is dramatically increased when the complex is bound to protein because transient interactions of the contrast-effective moiety of gadofosveset with albumin decrease the tumbling rate of the molecule (called the receptor-induced magnetization enhancement effect) and increase its persistence in blood. 27 Extravascular Gd-based agents have a blood half-life of 90 minutes; they diffuse rapidly into the tumor, so enhancement peaks within the first few minutes after they are administered. 5 By contrast, the intravascular contrast agent gadofosveset has a blood half-life of approximately 16 hours, and peak enhancement is not achieved until several hours after administration. 26 Immediately after injection, the unbound fraction of gadofosveset molecules (about 15%) passes through the blood–brain barrier and also causes the glomerular filtration rate to increase. However, after a few circulation cycles, the remaining molecules of gadofosveset are homogeneously distributed throughout the vascular blood in the albumin-bound state (about 85%) (see Fig. 18.1). 38
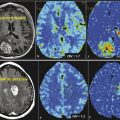
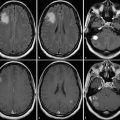
Serum proteins like albumin can extravasate from blood into tumors as a consequence of the breakdown of the blood–brain barrier (through openings in the endothelial junction or by pinocytosis) and increased vascular permeability. 28 , 39 Previous studies have shown that the bound fraction of gadofosveset has significantly stronger relaxation effects than the unbound fraction as a result of the larger size and resultant longer rotational correlation time of the molecule. 27 , 33 , 38 , 40 Six hours after gadofosveset administration has been proposed as the optimal time for scanning to evaluate glioblastoma in humans (Fig. 18.3, Fig. 18.4, Fig. 18.5, and Fig. 18.6).




A contrast agent for MRI of brain tumors should provide high T1 contrast. Contrast enhancement of brain tumors is based on complex pathophysiological changes in the tumor and the adjacent brain tissue, primarily due to disruption of the blood–brain barrier and increased capillary permeability. 3 Two types of brain tumor enhancement are important. Extravascular enhancement occurs when the contrast agent permeates through the vessel wall and accumulates in the perivascular interstitial fluid. Intravascular enhancement occurs when blood flow or volume increases due to the tumor’s increased or pathological vascularity; contrast material in blood results in enhancement proportional to increases in blood flow or volume, reflecting the vascular component or angiogenesis of the tumor. The increase in tissue relaxivities brought about by gadofosveset’s binding to intravascular and extravasated serum albumin, together with its longer half-life, results in greater enhancement in tissues with increased microvascularization or extravasation, making brain tumors more conspicuous on T1-weighted images and enabling better delineation of their extension (see Fig. 18.4, Fig. 18.5, and Fig. 18.6). Gliomas usually extend beyond the areas demarcated on T2-weighted or conventional Gd-based contrast-enhanced T1-weighted MRI, so improved delineation of the borders and extension of gliomas can be useful for planning surgical resection and/or radiation therapy and might contribute to a more accurate understanding of brain tumors. 41 , 42
One aspect of tumor physiology that may represent tumor aggressiveness is vascular leakage. Vascular leakage reflects the spread of increased vascularity, the rupture of the blood–brain barrier, or both; it arises from both focal areas with severe vascular abnormalities and regions with mild vascular abnormalities in which leakage often extends into the unenhanced abnormal region (peritumoral edema). 43 Peritumoral edema, which is unenhanced on T1-weighted images after the administration of classical contrast agents, has been described as an area of infiltration surrounding the tumor in which tumor cells coexist with tumor-secreted vascular permeability factors. 44 A high degree of vascular permeability indicates an abnormality of the blood–brain barrier and presumably reflects a larger size pore or disruption of the endothelial junctions. 45 Thus the magnitude of the opening in the blood–brain barrier seems to be a surrogate marker for tumor aggressiveness. Recently, Cao et al 43 , 46 determined the volume of vascular leakage from the transfer constant of the extravascular contrast agent Gd-DTPA from plasma to tissue in dynamic contrast-enhanced imaging; they found that vascular leakage volume predicted survival better than the fluid-attenuated inversion recovery (FLAIR) and post-Gd T1 tumor volumes, suggesting that vascular leakage could be a surrogate marker for the aggressiveness of high-grade gliomas. In Puig et al’s 30 study, enhancement had dispersed into the area of peritumoral edema 24 hours after gadofosveset trisodium injection in slightly more than half of the glioblastomas.
This peritumoral enhancement could be explained by the presence of albumin in the peritumoral edema, which is supported by the finding of albumin concentrations as high as 19.2 mg/mL in peritumoral edema in cat brains after xenotransplantation of cell clones from primary rat tumors. 47 Recently, Pronin et al 48 described the dispersion of Gd-DTPA enhancement into the area of peritumoral edema in the time interval between 5 minutes and 6 hours after injection in an 11-patient cohort study, with the highest average increase in volume of contrast observed at 6 hours. On the other hand, an increase in the volume of enhancement over time has also been demonstrated in animal models of glioblastomas, although this increase might not depend on the infiltrative and edematous patterns or might not be closely related with peritumoral edema. 49 Given that the extent of vascular leakage estimated by the transfer constant of Gd-DTPA might help predict tumor aggressiveness and survival in patients with high-grade glioma, 43 further research should address whether dispersion of gadofosveset over time from the initial site of enhancement to the periphery on T1-weighted imaging is useful for predicting survival.
Moreover, in the overwhelming majority of tumors with a central necrotic and/or cystic cavity, enhancement was observed in the cavity 24 hours after gadofosveset administration. 30 This finding, which has also been reported after gadopentetate dimeglumine and gadobenate dimeglumine administration in experimental brain tumors, 7 , 50 reflects the delayed wash-in and delayed washout of the contrast agent from the poorly vascularized necrotic area. 51 , 52 The leakage of intracellular proteins into the extracellular space after lysis of necrotic cells could also contribute to these phenomena.
Another advantage associated with the high relaxivity of gadofosveset is the possibility of reducing the dose of Gd (by a factor of 4) in terms of number of Gd ions administered, compared to conventional Gd chelates. This is important because whether nephrogenic systemic fibrosis develops depends not only on the stability of the Gd-based agents applied but also on the cumulative Gd dose administered. 53 Although gadobutrol is considered a relatively safe agent because its macrocyclic structures favor higher kinetic stability than linear chelates, 22 , 54 , 55 gadofosveset at a dose of 0.03 mmol/kg body weight yielded better enhancement than the gadobutrol at a dose of 0.1 mmol/kg body weight; thus gadofosveset represents a lower risk of nephrogenic systemic fibrosis without compromising image quality.
The experience to date suggests that the use of gadofosveset trisodium might have important clinical implications, particularly in intraoperative imaging for the real-time assessment of tumor resection. Generally, the first step in the multimodal treatment of glioma patients is surgery aiming to resect as much of the tumor as possible without inducing disabling neurologic deficits. Gross total resection in which no contrast-enhancing tumor remains after resection is associated with extended overall survival and progression-free survival in low-grade and high-grade gliomas (> 7 month increase in median survival), although no class 1 supporting evidence is available. 56 , 57 , 58 , 59 , 60 The accurate intraoperative assessment of tumor margins can be difficult for neurosurgeons; moreover, the functional relevance of brain tissue precludes widening resections to include a safety margin.
To optimize the extent of resection, intraoperative imaging methods, including computed tomography (CT), ultrasonography, 5-aminolevulinic acid, and MRI have been established in operating rooms, enabling surgeons to better see the extent of lesions and serving as immediate resection control. Of these intraoperative imaging methods, high-field intraoperative MRI scanners provide the highest resolution for detecting contrast-enhancing tissue tumor remnants and are considered essential in the surgical management of glioblastomas. 58 Intraoperative MRI could influence the course of surgery by enabling additional resection in 10 to 70% of cases, and it could increase the number of gross total resections to nearly 100%. 58 , 60 The greater enhancement provided by gadofosveset, at least in the first 6 hours after a single dose, might enable greater volumes of tumor to be resected, thus increasing the percentage of patients in whom the maximum amount of tumor can be safely resected. However, the dispersion of gadofosveset at 24 hours follow-up is also a potential source of errors: a long delay in scanning after contrast injection might lead to overestimation of the volume of enhancing tumor and misinterpretation of the extent of the zone of enhancement, resulting in the resection of initially nonenhancing white matter.
Blood pool contrast agents such as gadofosveset are likely to play a central role in the evaluation of tumor angiogenesis. Angiogenesis is vital to tumor growth. When a tumor exceeds a diameter of 1 to 2 mm, diffusion from the surrounding vasculature alone is no longer sufficient to provide nutrients to the outer cells of the tumor; the tumor induces the growth of new vessels to supply nutrients and possibly to remove biological end products secreted by rapidly dividing cancer cells. Blood pool contrast agents result in both intravascular enhancement, related to blood flow, and interstitial enhancement, related to vessel permeability. The persistent strong intravascular enhancement of gadofosveset allows for the acquisition of high-resolution images in the steady state, enabling better delineation of vessel pathology, abnormal vascular beds, and increased vascularity associated with fast-growing tumors (Fig. 18.7). There is an urgent clinical need for accurate, noninvasive imaging biomarkers to quantify tumor angiogenesis and to monitor the response to treatment. Furthermore, if blood pool contrast agents are validated as surrogate markers for treatment outcome, they will be very useful in the development of new tumor angiogenesis inhibitors. Thus the ability of MR techniques using blood pool contrast agents to reveal changes in tumor microvascularity is likely to accelerate the development, testing, and monitoring of new antiangiogenic drugs.
Overall, the future of blood pool contrast imaging is promising. However, although it seems reasonable to expect that the greater diagnostic information available on gadofosveset-enhanced images would help improve the efficacy of therapeutic procedures, particularly with regard to better definition of the target volumes at radiosurgery, a more comprehensive understanding of the precise extent to which greater diagnostic information contributes to improved patient management would be required to prove this point conclusively. Future studies are necessary to fully explore the true clinical potential of gadofosveset as well as its advantages over the currently established Gd-based agents for contrast-enhanced MRI in the diagnosis and follow-up of brain tumors.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree