34 Crystal-induced Osteoarthropathies and Related Diseases
The term crystal-induced osteoarthropathy describes a heterogeneous group of joint diseases that are caused by disturbances in biochemical processes. Intraarticular precipitation and deposition of defined metabolic products sometimes induced characteristic osteoarthropathies. Crystal-induced osteoarthropathies include hyperuricemia (gouty arthritis), chondrocalcinosis (calcium pyrophosphate dihydrate [CPPD] crystal deposition disease), calcium hydroxyapatite crystal deposition disease, hemochromatosis, Wilson disease, alkaptonuria, and oxalosis. Typical radiographic findings characterize these disease entities.
Gouty Arthritis/Hyperuricemia
Pathoanatomy and Clinical Symptoms
Gouty arthritis is caused by elevated concentrations of uric acid in the extracellular space. The measurable correlate is uric acid in serum (normal value up to 6.5 mg/dl). By far the most common cause of primary gout is a combination of a genetically determined (so far not fully elucidated) disorder in renal excretion of uric acid (hereditary gout) with increased alimentary purine intake. The enzymatic defect (in phosphoribosyltransferase) responsible for the Lesch-Nyhan syndrome is a rare cause of increased synthesis of uric acid. Secondary gouty arthritis can be caused by a number of diseases that are not directly associated with the purinemetabolism but result in an increased accumulation of uric acid (e.g., myeloproliferative syndromes, leukemias) or to a decrease in renal excretion of uric acid (e.g., chronic kidney diseases). Gouty arthritis most commonly affects males in their fifties, with a predilection for the joints of the feet. Women are affected later and have milder disease and polyarticular manifestations.
When the solute capacity of uric acid is exceeded, sodium urate crystals precipitate in the kidneys and especially in the bradytrophic tissue of the joints. Gout of the joints has the following clinical symptoms:
Gouty arthritis most commonly presents as acute monarthritis (arthritis urica) in the following order of incidence: metatarsophalangeal joint of the big toe (podagra), metacarpophalangeal joint of the thumb (chiragra), the rest of the joints of the fingers and hand, and, less commonly, the large limb and iliosacral joints. During an acute attack of gouty arthritis, the urate crystals cause an extreme inflammatory reaction with redness, swelling, hyperthermia, and painful restriction of movement. Polarizing light-microscopic evidence of sodium [natrium]-monohydrate crystals in the synovial fluid constitutes proof of gout, but is usually unnecessary because of the unambiguous clinical presentation.
After a complaint-free interval, chronic recurrent attacks of gouty arthritis can set in.
Chronic gouty arthritis is characterized by the development of tophi in bones, bradytrophic tissues, and joints. Aside from polyarticular destructive osteoarthropathies, chronic deposition of urate crystals results in tophi in the subcutis, tendon sheaths, and bursae, and occasionally in Heberden nodules. In contrast to the remission state of acute gouty arthritis, chronic pain appears only in the stage of advanced joint destruction.
A special form, juvenile gouty arthritis (usually the Lesch-Nyhan syndrome), can have an aggressive course.
Diagnostic Imaging
Radiography
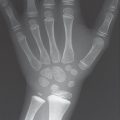
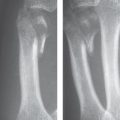
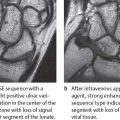
Radiographic examination is unremarkable in the first attack. In recurrent attacks, reversible periarticular osteopenia can appear. Typical articular lesions that can be seen in radiographs appear only in chronic cases lasting 5–10 years (Figs. 34.1a, 34.2a , 34.3 ), especially :
When calcinoses have developed in the presence of urate deposits (the urate crystals themselves are less radiodense)
When the initially slow destruction of the articular cartilage has become extensive.
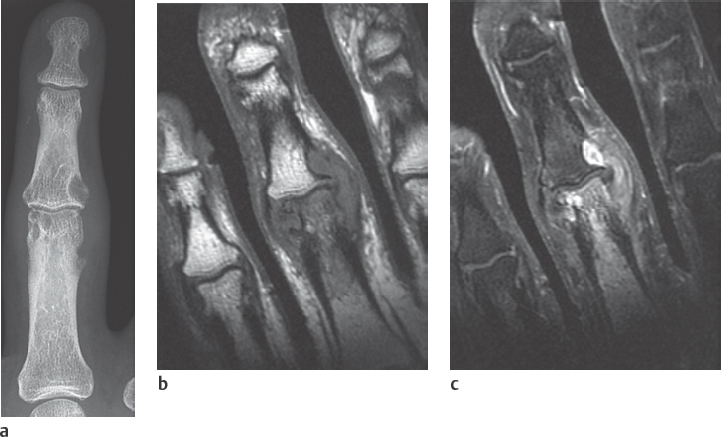
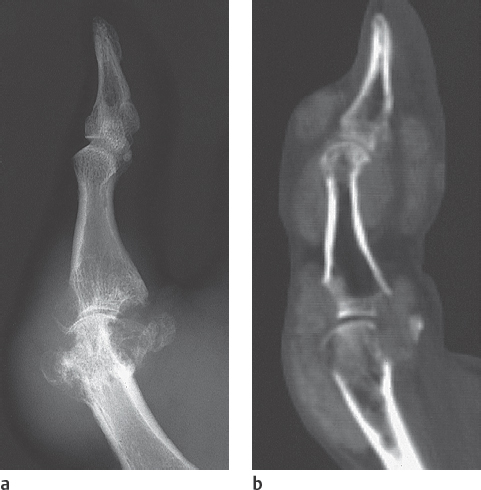
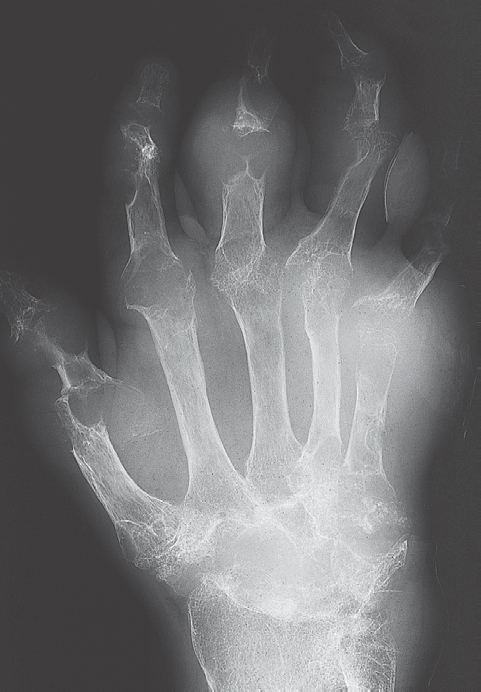
Table 34.1 summarizes the radiographic signs of gout.
Pharmacological treatment can make the soft-tissue calcifications disappear entirely, whereas erosive joint lesions and deformities remain without any reparativeprocesses.
Ultrasonography
Gout tophi are characteristically hyperechoic in high-resolution ultrasonography. Their hyperechoic appearance confirms the presence of gout, since lesions of nearly all other inflammatory entities are hypoechoic.
Computed Tomography
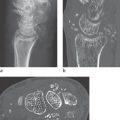
In CT, urate tophi have densities of about 170 HU (Fig. 34.2b). Although differentiation from calcified tophi and loose calcifications of other origin based on density can be problematic, gout can almost always be reliably differentiated from noncalcified soft-tissue masses, like xanthomas, in CT. Marginal erosions are characteristic.
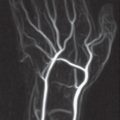
The anatomic neighbourhood of urate deposits in the anatomically complex area of the carpus can be visualized better in CT than in projection radiography.
Magnetic Resonance Imaging
Inflammation of soft tissues, synovium, articular cartilage, and bones found in gouty arthritis can be detected in MR imaging earlier than in radiography (Fig. 34.1b, c). The inflamed areas have inhomogeneous, partially decreased signal intensity in T2-weighted sequences due to inclusions of tophi and possible hemorrhaging. In T1-weighted sequences, the tophi have an intermediate signal intensity. After intravenous administration of gadolinium, there is a moderate contrast enhancement in marginal erosions due to synovitis and less enhancement within the tophi.
Differential Diagnosis
During the initial manifestation, tenosynovitis and acute calcium deposition with hydroxyapatite crystals must also be considered.
The characteristic radiographic signs alone can exclude septicemic bacterial arthritis, rheumatoid arthritis, CPPD deposition disease, and diabetic osteoarthropathy during disease progression.
Therapeutic Options
Nonsteroidal anti-inflammatory drugs (NSAIDs) are the therapy of first choice for both acute gout attacks and chronic hyperuricemia. Acetylsalicylic acid is an exception because it decreases uric acid excretion. Glucocorticoids can be administered directly into the affected joint or systemically in the acute stage. Colchicine should only be prescribed when NSAIDs and corticoids are contraindicated because of its numerous side-effects. Chronic hyperuricemia is treated with diet and uricostatics or uricosurics.
Calcium Pyrophosphate Dihydrate (CPPD) Deposition Disease (Pyrophosphate Arthropathy)
Pathoanatomy and Clinical Symptoms
In this disease, calcium-pyrophosphate dihydrate (CPPD) crystals are deposited on the surfaces of the articular cartilage and the fibrous cartilage, and in the capsuloligamentary system. The two forms listed in Table 34.2 are differentiated pathoanatomically according to the type of crystal deposition.
Asymptomatic calcium pyrophosphate dihydrate deposits are often observed among the elderly, when the condition is termed chondrocalcinosis. In a high percentage of persons over 80 years of age, chondrocalcinosis is an incidental finding in the articular cartilage and causes no complaints. The most common sites of manifestation are the triangular fibrocartilage complex of the wrist, the menisci in the knees, the symphysis, and the articular cartilages. The familial occurrence of the disease indicates a genetic disposition. The manifestation generally begins before the age of 60 years in these cases.
If calcium pyrophosphate crystals are found, however, in the synovial fluid, CPPD deposition disease can become symptomatic.
calcium deposits in periarticular soft tissues cause monarthritic or polyarthritic complaints (pseudogout). During the pseudogout attack the joint is swollen, reddened, hyperthermic, and painful on pressure.
Deposits of CPPD in the joints can cause chronic destruction. Such cases are referred to as CPPD arthropathy.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree