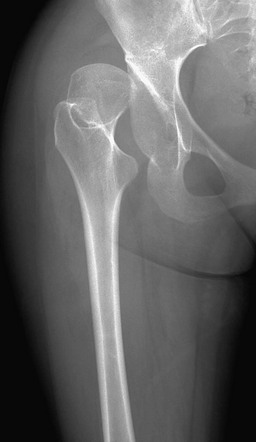
Chapter 134 Malalignment of bony components of the extremities may be congenital or acquired. A defect that results in malalignment may be primary to bone or secondary to soft tissue or neuromuscular disorders. Malalignment is often seen with congenital deformities characterized by embryologic failure of development, discussed in Chapter 128. One of the most common alignment disorders is developmental dysplasia of the hip, discussed in Chapter 133. Etiologies, Pathophysiology, and Clinical Presentation: Erb palsy is caused by birth trauma to the C5 and C6 nerve roots. It is associated with prolonged, difficult delivery (dystocia) and with larger infants. Infants present soon after birth with decreased motion of the involved extremity. Children with a persistent defect will hold their arms in internal rotation with pronation of the forearm and flexion of the wrist. The brachial plexus injury is the primary insult leading to secondary glenohumeral dysplasia. Imaging: At birth, radiographs serve to exclude fractures of the clavicle and humerus. Ultrasonography and magnetic resonance imaging (MRI) have been used to directly evaluate the brachial plexus;1,2 however, the primary goal of advanced imaging is to evaluate orthopedic anatomy to determine function and orthopedic treatment since primary repair of the brachial plexus injury is difficult. Progressive deformity with secondary glenohumeral dysplasia manifests as a small, flattened humeral head, which may sublux, usually in the posterior direction relative to a small, shallow, abnormally retroverted glenoid (Fig. 134-1).3 In normal shoulders, the glenoid is mildly retroverted by approximately 5 degrees posteriorly relative to a line perpendicular to the axis of the scapular body.4 With Erb palsy, glenoid retroversion averages 25 degrees. The scapula is hypoplastic and elevated; the acromion is tapered and inferiorly directed, as is the coracoid; the clavicle is shortened. Figure 134-1 Erb palsy in a 9-year-old girl. Many of these findings are seen on radiography; however, computed tomography (CT) or MRI can be used to quantify the degree of glenoid retroversion, deformity of the glenoid and humeral head, glenoid–humeral head congruence, and relative muscle volume and quality of the affected shoulder.5–9 The glenoid normally has a concave shape. With glenohumeral dysplasia, the glenoid becomes progressively flat, convex, or biconvex with a pseudoarticulation with the humeral head. MRI is preferred in young children (<5 years old) (e-Fig. 134-2) and CT in older children. To determine glenoid version, the articular cartilage should be used when MRI is performed; the glenoid cortical bone should be used when CT is performed. In infants, ultrasonography may be used to assess instability of the glenohumeral joint.10–12 e-Figure 134-2 Erb’s palsy in a 2-year-old girl. Treatment: Microsurgery techniques may be attempted to address the underlying traumatic neural injury.13 Controversy exists as to their role and proper timing. The main focus of treatment is on the reconstructive surgical techniques of the shoulder, elbow and forearm, and hand and wrist, aimed at preserving joint integrity and maximizing function.13,14 Therefore, imaging optimization should be tailored for orthopedic anatomy rather than the brachial plexus. Etiologies, Pathophysiology, and Clinical Presentation: In Madelung deformity, the radius is short and its distal articular surface tilted toward to ulna.15 In most cases, the cause of Madelung deformity is unknown. Madelung deformity occurs more often in girls.16 Patients may have pain; however, treatment is more often sought because of deformity or limited range of motion. With an underlying syndrome, the deformity is more likely to be bilateral. Madelung deformity is occasionally seen with Turner syndrome and is a characteristic in dyschondrosteosis (Léri-Weill syndrome) (Fig. 134-3).15 Among these cases, 10% to 15% are familial. A Madelung-like deformity may also be seen in patients with hereditary osteochondromatosis or enchondromatosis, also suggesting a defect in normal distal radial maturation. Madelung deformity may occur as a complication of infection or trauma that results in medial and volar radial physeal growth disturbance.17 Figure 134-3 Madelung deformity in a 10-year-old girl who presented with right wrist pain. Imaging: The distal articular surface of the radius is tilted in an ulnar and volar direction.18,19 The radius is short and bowed dorsally and laterally (“bayonet deformity”). Secondary distortion of the carpus is observed with an abnormally narrow carpal angle and proximal lunate migration.18,19 The distal ulna is subluxed. The distal radial growth plate may prematurely fuse along its ulnar aspect. CT or MRI may be used to assess the extent of distal radial physeal fusion.20 Etiologies, Pathophysiology, and Clinical Presentation: Trauma, infection, inflammatory disease (juvenile idiopathic arthritis) or other causes of premature fusion may lead to shortening of the ulna relative to the radius (negative ulnar variance) or shortening of the radius relative to the ulna (positive ulnar variance) (Fig. 134-4).21 Most cases are idiopathic. Processes associated with hyperemia, for example, fracture healing, infection and vascular lesions, may lead to over growth of one of the bones. Imaging: At skeletal maturity, the distal radial and ulnar articular surfaces are nearly at the same level, and the radial styloid projects 9 to 12 mm distal to the ulnar articular surface.21,22 With negative ulnar variance, the ulna ends more proximally, and with positive ulnar variance, the ulna ends more distally (e-Fig. 134-5).21,23 Ulnar variance may be exaggerated with forearm pronation and decreased with forearm supination. e-Figure 134-5 Method of measuring ulnar variance. Treatment: Osteotomy with ulnar or radial shortening may be indicated to reduce symptoms and avoid sequelae. Negative ulnar variance is associated with the development of avascular necrosis of the lunate (Kienböck disease).24 Positive ulnar variance is associated with ulnolunate impaction syndrome (Fig. 134-6) and triangular fibrocartilage complex degeneration. Etiologies, Pathophysiology, and Clinical Presentation: The normal neck-shaft angle of the proximal femur is approximately 150 degrees at birth and decreases to 120 to 130 degrees in adulthood.25 External or internal rotation of the hip or femoral anteversion may affect measurement.26 Functional coxa vara occurs with disorders that result in femoral neck shortening, as in trauma, infection, or epiphyseal osteonecrosis.27 True coxa vara occurs as a congenital anomaly that is caused by bone softening (e.g., rickets, osteogenesis imperfecta, fibrous dysplasia) or to abnormal growth (e.g., spondyloepiphyseal dysplasia congenita, spondyloepimetaphyseal dysplasia, cleidocranial dysplasia).28,29 Children with developmental coxa vara present with a limp (unilateral deformity) or a waddling gate (bilateral deformities). Coxa vara may occur as the result of abnormal growth at the proximal femoral physis that results in abnormal angulation of the physis. Congenital coxa vara occurs with a congenital short femur (i.e., proximal focal femoral deficiency) and does not spontaneously resolve.28 With infantile or developmental coxa vara, the hip is normal at birth, and deformity is noted when the child begins to walk.28,30 Infantile coxa vara may be self-limited. Acquired coxa vara is caused by another process such as trauma.28 Imaging: In coxa vara, the femoral neck-shaft angle is decreased from normal. A measurement below 110 degrees is considered coxa vara.31 Fragmentation and sclerosis may be seen at the medial margin of the proximal femoral metaphysis (Fig. 134-7).30 The Hilgenreiner epiphyseal angle is the angle between the Hilgenreiner line and a line drawn through the physis (e-Fig. 134-8).28,31 If it is less than 45 degrees, progression is unlikely. If over 60 degrees, progression is likely. If 45 to 60 degrees, prognosis is less predictable. Figure 134-7 Bilateral congenital coxa vara in a 4-year-old boy. e-Figure 134-8 Hilgenreiner epiphyseal angle. Treatment: Surgical management may be warranted for progressive disease, especially if asymmetric or associated with pain, leg length discrepancy, or both.28 Valgus osteotomy is performed, and physeal fixation or tendon transfers may also be performed to deter progression and improve mechanical function. Etiologies, Pathophysiology, and Clinical Presentation: With coxa valga, the neck-shaft angle of the proximal femur is increased.32 Coxa valga is most often seen in patients who are nonambulatory and nonerect, such as those with cerebral palsy and other neuromuscular disorders (Fig. 134-9).26,33 Imaging: The femoral neck-shaft angle is measured on radiographs. External rotation may mimic coxa valga and can be differentiated through the positioning of the greater trochanter.26 With external rotation, the greater trochanter projects through the femur, whereas with true coxa valga, it is located laterally. Increased femoral anteversion may cause the femoral neck-shaft angle to be overestimated.32 Acetabular dysplasia and femoral subluxation are frequently concomitant findings.32 Etiologies, Pathophysiology, and Clinical Presentation: Increased femoral anteversion may hinder proper localization of the femoral head relative to the acetabulum. Increased femoral anteversion is seen in hip deformity caused by developmental dysplasia of the hip, Legg-Calvé-Perthes disease, and cerebral palsy.32 With increased anteversion, in-toeing of feet is noted. Femoral version is the angulation of the femoral neck in the transverse plane measured relative to the femoral condyles distally (Fig. 134-10). If the femoral neck is anteriorly angulated with respect to the femoral condyles, the femur is anteverted. If the femoral neck is posterior with respect to the femoral condyles, the femur is retroverted. Normal femoral anteversion is 35 to 50 degrees at birth, decreasing steadily to 10 to 15 degrees in adulthood (Fig. 134-11).27 Figure 134-10 Schematic diagram of the right femur as viewed from below. Figure 134-11 Normal femoral anteversion. Imaging: On CT, femoral anteversion is the angle between the axis of the femoral neck and the transcondylar axis at the distal femur (Fig. 134-12).34–36 Limited low-dose axial images are usually performed; however, axial oblique or three-dimensional images may improve measurement.37,38 Figure 134-12 Computed tomography method for assessing femoral anteversion in a 17-year-old girl with a history of right femoral fracture. Etiologies, Pathophysiology, and Clinical Presentation: Tibial torsion is the degree of rotation of the distal end of the tibia relative to the upper end of the tibia. Newborns have internal tibial torsion relative to older children and adults. Lack of normal progression results in in-toeing. Internal tibial torsion is the most common cause of in-toeing in the preschool age group. Imaging: Assessment of tibial torsion is often performed with assessment of femoral version. Limited axial low-dose CT images are obtained through the proximal and distal tibias.41 Tibial torsion is best measured as the angle between the posterior epiphyseal cortical margin of the tibia proximally and a bimalleolar line distally. Normal values are 5 degrees of external rotation in a newborn, which progresses to 15 to 20 degrees of external rotation in an adult.40,42 Etiologies, Pathophysiology, and Clinical Presentation: Bowleg deformity manifests as separation of the knees, with the legs in anatomic position. Pathologic causes include rickets, osteogenesis imperfecta, neurofibromatosis, skeletal dysplasias (i.e., campomelic dysplasia, achondroplasia), focal fibrocartilaginous dysplasia, congenital bowing, Blount disease, and, occasionally, growth plate trauma.43–46 Recently, greater prevalence of bowleg and tibia vara has been noted in some adolescent athletes, most notably soccer players.47–49 Repetitive stress on the proximal tibial physis may play a role in this.48 Most lateral bowing in otherwise normal infants and children younger than 2 years of age is normal and developmental (“physiologic”) and resolves without treatment.44,50–52 Similar to Blount disease, exaggerated physiologic bowing is seen in early walkers, African Americans, and heavier children.44 Imaging: Ozonoff described the following findings as characteristic of physiologic lower extremity bowing: (1) The tibia is abducted relative to the femur, and both bones are intrinsically bowed laterally; relative tibial torsion produces external rotation of the upper tibia relative to the distal tibia; (2) margins of the distal femoral and proximal tibial metaphyses are mildly accentuated with small beaks; (3) medial cortices of the tibia and femur are thickened; (4) distal femoral and proximal tibial epiphyses are not well ossified medially and are wedge shaped; (5) the distal tibial growth plate may be tilted lateral.53 Radiographically, the femur and the tibia are also mildly bowed anteriorly, with beaking occurring posteriorly (Fig. 134-13). Physiologic bowing is usually more marked in the tibias. Occasionally, lateral bowing may almost exclusively be seen in the distal femur (e-Fig. 134-14). The varus deformity is common in normal infants and converts to valgus between 18 and 36 months of age. Degree of valgus reduces spontaneously by 6 to 7 years of age to a mild degree that remains throughout life. Approximate normal angles are 17 degrees varus in a newborn, 9 degrees varus at 1 year, 2 degrees valgus at 2 years, 11 degrees valgus at 3 years, and 5 to 6 degrees valgus at 13 years (e-Fig. 134-15).52,54,55 Figure 134-13 Bilateral idiopathic bowed legs in a 22-month-old boy. e-Figure 134-14 Femoral bowed legs (femora vara): Endogenous stress trauma at the knees without weightbearing. e-Figure 134-15 Development of the tibiofemoral angle during growth. Radiographs should be taken with the patient bearing weight as soon as he or she is able to stand (Fig. 134-16). The radiographs may suggest an underlying disorder such as rickets or a dysplasia. Figure 134-16 Bowed legs of a 12-month-old girl who started to walk at 8 months. Treatment: Persistent varus with delayed conversion to valgus may indicate a higher likelihood of Blount disease (tibia vara). In the second year, it may be difficult to distinguish normal physiologic bowing from Blount disease.56 Exaggerated varus during the second year is likely developmental or physiologic and does not require treatment.50,51,54 Any varus at the knee after 2 years of age should raise concern. Such patients must be monitored to exclude progression to Blount disease.55,57 Realignment surgery is rarely needed in isolated bowleg without underlying dysplasia, metabolic bone disease, or Blount disease. Etiologies, Pathophysiology, and Clinical Presentation: Blount disease (tibia vara; osteochondrosis deformans tibiae) is a progressive deformity affecting the proximal tibia (Fig. 134-17).58–60 It is theorized that stress on the posteromedial proximal tibial physis causes growth suppression. Figure 134-17 Progressive changes in Blount tibia vara.
Alignment Disorders
Overview
Upper Extremities
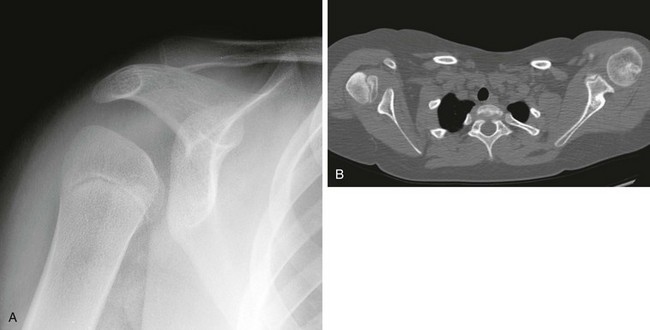
A, On radiography, the humeral head appears small and abnormally contoured, the glenoid margins are indistinct, and the acromion is curved. B, Axial computed tomography shows the right glenoid to be small and retroverted. The right humeral head is small and squared in contour but appears normally located relative to the glenoid. Note the asymmetric muscle volume of the right shoulder girdle compared with the left shoulder girdle.
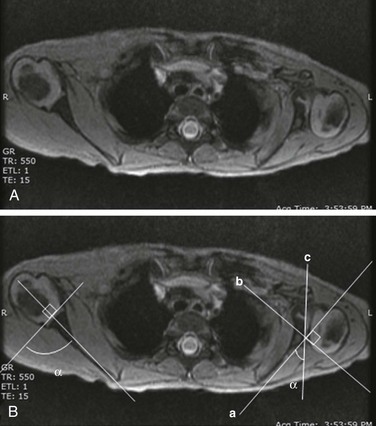
A, Gradient recalled echo axial sequence demonstrates severe retroversion and dysplastic convex left glenoid and posteriorly subluxed humeral head as well as global left shoulder girdle muscle atrophy. B, Same sequence with glenoid version measurements. Version is calculated by first (a) a line drawn along scapula, (b) a perpendicular line drawn with respect to line a, and (c) a line drawn along articular surface of glenoid. The glenoid angle is determined between line a and c. If the glenoid is directed posteriorly with respect to scapula, the glenoid is in retroversion. If the glenoid is directed anteriorly, the glenoid is in anteversion.
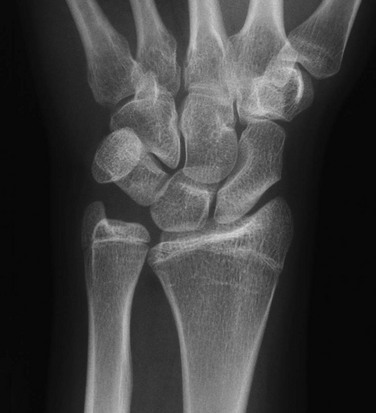
Madelung Deformity
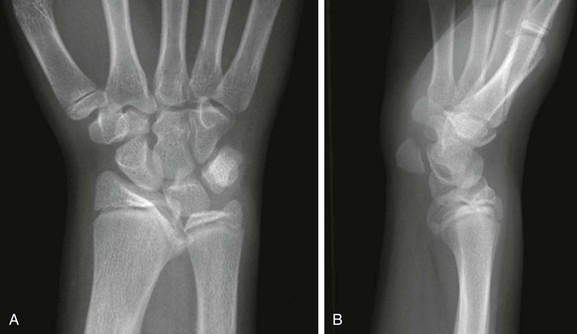
A, Posteroanterior radiograph shows medial tilt of the distal radial articular surface and physis with thinning of the medial aspect of the distal radial epiphysis. B, Lateral radiograph shows anterior tilt of the distal radial articular surface with mild anterior displacement of the carpus.
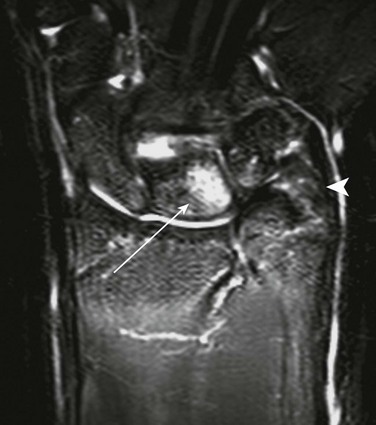
Ulnar Variance
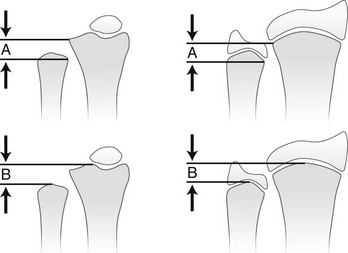
A, Distance from the most proximal point of the ulnar metaphysis to the most proximal point of the radial metaphyses. B, Distance from the most distal point of the ulnar metaphysis to the most distal point of the radial metaphysis. (Modified from Hafner F, Poznanski AK, Donavan JM. Ulnar variance in children—standard measurements for evaluation of ulnar shortening in juvenile rheumatoid arthritis, hereditary multiple exostosis and other bone and joint disorders of childhood. Skeletal Radiol. 1989;18:513-516.)
Lower Extremities
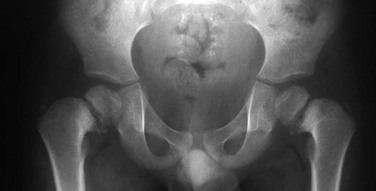
Proximal femoral growth plates are tilted, medial side down. Slight fragmentation is noted at the medial aspect of each proximal femoral physis.
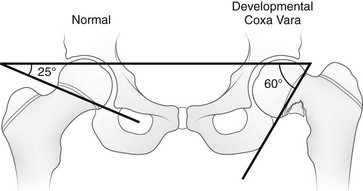
This angle is created by a single line through the triradiate cartilage and its intersection with another line through the physis. The normal angle is about 25 degrees. (From Beals RK. Coxa vara in childhood: evaluation and management. J Am Acad Orthop Surg. 1998;6:93-99.)
Coxa Valga
Femoral Anteversion
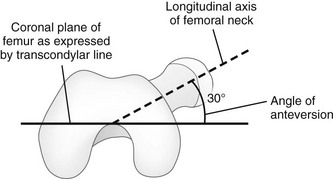
The femoral neck is angulated anterior (anteversion) relative to the transcondylar axis of the distal femur. (Modified from Greenspan A. Orthopedic imaging: a practical approach. 4th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2004.)
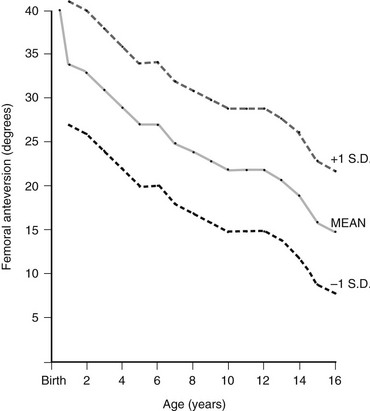
The mean value is an average of those reported by Shands and Steele (1958), Crane (1959), and Fabry and associates (1973). Standard deviations are only an approximation and are based on the range of those determined by Fabry and associates. (From Ozonoff MB: Pediatric orthopedic radiology. 2nd ed. Philadelphia: WB Saunders; 1992.)
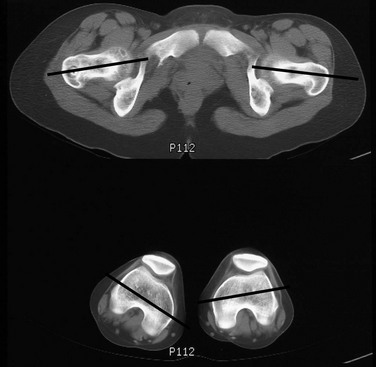
Lines denote the axes of the femoral necks and the transcondylar axes of the distal femora. An angle of 49 degrees of anteversion was measured on the right and an angle of 14 degrees was measured on the left.
Leg
Bowleg (Genu Varum)
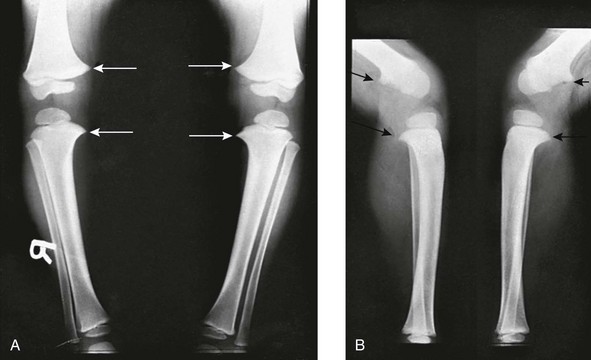
A, Frontal projection. Arrows point to the medial beaking of the femoral and tibial metaphyses at the knees. The increased stress of weightbearing has thickened the medial and dorsal cortical walls of the tibias inward. The medial femoral epiphyseal ossification centers are too small, which are under greater stress of weightbearing when the legs are bowed. B, Lateral projection. Arrows point to dorsal beaking of the femoral and tibial metaphyses. After correction of bowed legs, these “stress” phenomena disappear after several months.
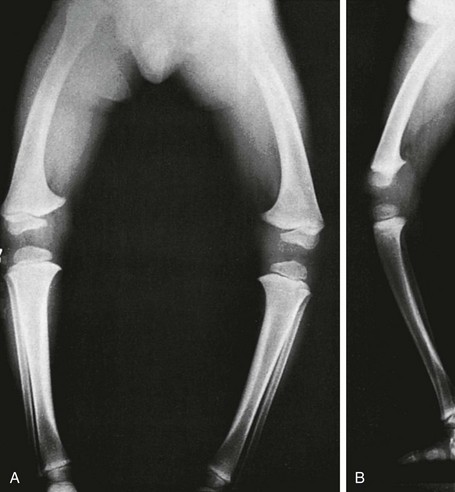
Anteroposterior projection shows marked bowlegs, in which the tibias are straight and both femora are bowed abruptly lateral near their distal ends (femora vara). Femoral epiphyseal ossification centers are small and flattened on their medial halves.
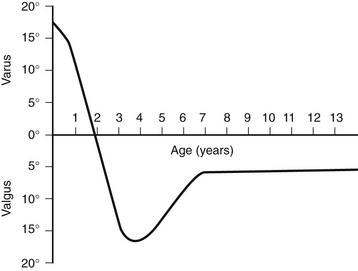
This graph shows the normal physiologic progression of bowlegs to knock-knees and then to normal during the early years of growth. (From Bruce RW Jr. Torsional and angular deformities. Pediatr Clin North Am. 1996;43:867-881.)
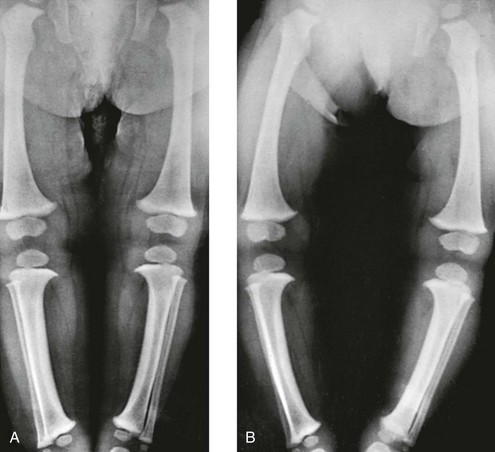
A, In recumbent position, the tibias and femora are bowed, but the legs are not bowed because the knees and ankles are in apposition. B, In erect position, during weightbearing and with ankles in apposition, the legs are bowed, with a gap of 12 cm at the knees. The real clinical deformity of bowed legs is shown most accurately when the patient is erect and is bearing weight.
Blount Disease (Tibia Vara)
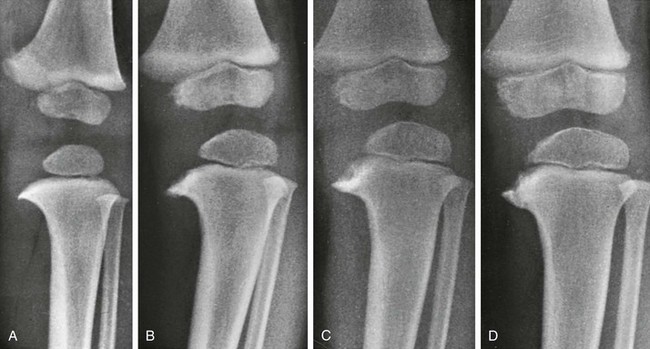
A, At 17 months, the medial segment of the tibial metaphysis is widened and sharpened into a short beak or spur that is bent slightly caudad. B, At 26 months, the spur is longer, sharper, and more bent; a radiolucent strip on its upper edge represents noncalcified cartilage. C, At 32 months, the amount of cartilage is increased and the spur thickened. D, At 38 months, the beak of the spur is displaced caudad, possibly owing to trauma; the medial edge of the ossification center is flattened, and the femur has shifted mediad in relation to the tibia.![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree