div epub:type=”chapter” role=”doc-chapter”>
Atlas and Anatomy of PET/MRI
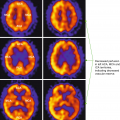
Hybrid positron emission tomography/magnetic resonance image (PET/MRI) has undergone rapid evolution during the last years, moving from a predominantly research field to clinical practice. With the advances in faster silicon photomultiplier detectors, MRI-based attenuation correction, and image reconstruction, significant improvements in equipment and image quality have been achieved. Currently, there are fully integrated PET/MRI systems that allow simultaneous and more rapid acquisition, improving not only the technical quality but also the experience for patients who need a low radiation dose [1–3]. With this technology comes the possibility of performing multiparametric MRI studies, where detailed anatomical evaluation and functional evaluation are possible, not only considering the qualitative and quantitative data of PET but also integrating multiple parameters such as perfusion (contrast-enhanced sequences), cellularity (diffusion-weighted sequence), metabolites (spectroscopic analysis), and texture analysis. Additionally, recent developments are very promising in giving the possibility of incorporating advanced data and biomarkers to integrate with bioinformatics and allow a better understanding of the disease, as well as an efficient evaluation, prediction of response to treatment, and follow-up [4–7].
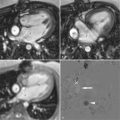
With the growing availability of PET/MRI, its main and differential applications have also been clarified. Nonspecific 18F-fludeoxyglucose (FDG) PET/MRI continues to be the most widely used, and thus new radiotracers are expanding the field to be explored. Among the most frequent applications of 18F-FDG PET/MRI, where its superiority over PET/CT has been demonstrated, are the evaluation of head and neck, colorectal, gynecological, bone and soft tissue tumors, as well as the evaluation and characterization of primary or secondary liver lesions [8–12]. It has also shown good results in non-tumor pathology such as epilepsy, inflammatory bowel disease, and cardiac sarcoidosis [4, 13].
The creation of new radiotracers that can be imaged both with PET/MRI and PET/CT, depending on the case and availability, has allowed great advances in the evaluation of other oncological and non-oncological pathologies. In the case of neuroendocrine tumors and prostate cancer, targeting somatostatin receptors with 68Ga-DOTATOC, targeting PSMA with 68Ga-PSMA-11 among others available tracers, and the inclusion of 177Lu agents have revolutionized the diagnosis and treatment of these pathologies respectively [14, 15]. In the field of neuroimaging, the wide availability of radiotracers has made it possible to improve the evaluation of multiple targets different from glucose metabolism (FDG), such as DNA synthesis (18F-fluorothymidine), protein synthesis (11C-methionine, 18F-fluoroethyl-L-tyrosine [FET], 18F-fluoro-L-3,4-dihydroxyphenylalanine [DOPA]), and hypoxia (18F-fluoromisonidazole) [16, 17]. In the field of degenerative diseases, where much remains to be explored and research is very promising, examples of emerging invaluable applications are amyloid PET and Tau PET for Alzheimer’s disease, as well as 18FP-CIT PET for Parkinson’s disease [18, 19].
In this chapter, we present multiple demonstrative examples of the different uses of PET/MR, with the most relevant anatomical references for each case.
1 Head and Neck
1.1 Case 1

1. Left superior frontal gyrus
2. Left precentral gyrus
3. Left postcentral gyrus
4. Peritumoral edema, right parietal lobe
5. Primary central nervous system lymphoma involving right parietal white matter
6. Primary central nervous system lymphoma involving corpus callosum
1.2 Case 2

1. Metabolically active lymphoma adjacent to the posterior horn of the left lateral ventricle
2. Normal FDG uptake in gray matter at the frontal cortex
3. Normal FDG uptake in the white matter at the frontal lobe
4. Anterior horns of the lateral ventricles
5. Posterior horns of the lateral ventricles
6. Septum pellucidum
7. Anterior cerebral arteries
8. Falx cerebri, frontal region
9. Perilesional edema
10. Choroid plexus at right lateral ventricle
11. Skull, left parietal area
12. Left temporal muscle
13. Scalp, left parietal area
14. Superior sagittal sinus
1.3 Case 3

1. Right eye retinoblastoma with minimal diffuse FDG uptake
2. Right medial rectus muscles
3. Right temporal lobe
4. Cerebellar vermis
5. Left optic nerve
6. Left lateral rectus muscle
7. Pons
8. Ethmoid air cells
9. Left eye, anterior chamber
10. Left eye, vitreous chamber
11. Left temporal arachnoid cyst
12. Pituitary gland
13. Left temporal bone
14. Basilar artery
15. Fourth ventricle
1.4 Case 4

1. Metabolically active tumor in the upper olfactory recess
2. Thalamus
3. Pons
4. Genu of corpus callosum
5. Lateral ventricle anterior horn
6. Splenium of corpus callosum
7. Pineal gland
8. Straight sinus
9. Cerebellum
10. Nuchal ligament
11. Spinal cord
12. Sphenoidal sinus with secretion due to obstruction
13. Left optic nerve
14. Basilar artery
15. Cerebellar vermis
16. Right temporal lobe
17. Left eye, vitreous chamber
18. Left ethmoid air cells
19. Optic chiasm
20. Fourth ventricle
1.5 Case 5

1. Metabolically active bilateral orbital lymphoma infiltration
2. Right anterior ethmoid air cells
3. Right posterior ethmoid air cells
4. Sella turcica (pituitary gland)
5. Right temporal lobe
6. Right superior eyelid with lymphoma infiltration
7. Frontal sinuses
8. Right temporal muscle
9. Pons
10. Right eye lens
11. Right eye vitreous chamber
12. Right optic nerve
13. Left medial rectus muscle
14. Left lateral rectus muscle
15. Left internal carotid artery
16. Basilar artery
17. Fourth ventricle
18. Midbrain, red nucleus
19. Aqueduct of Sylvius
20. Crista galli
21. Left olfactory cortex
22. Left Sylvian fissure
23. Anterior cerebral arteries
24. Midbrain, sustancia nigra
1.6 Case 6

1. Nasal septum
2. Right maxillary sinus
3. Right masseter muscle
4. Right temporalis muscle
5. Right lateral pterygoid muscle
6. Right medial pterygoid muscle
7. Right mandibular ramus
8. Right external auditory canal
9. Right internal carotid artery
10. Right mastoid air cells
11. Metabolically active tumor at the right fossa of Rosenmüller

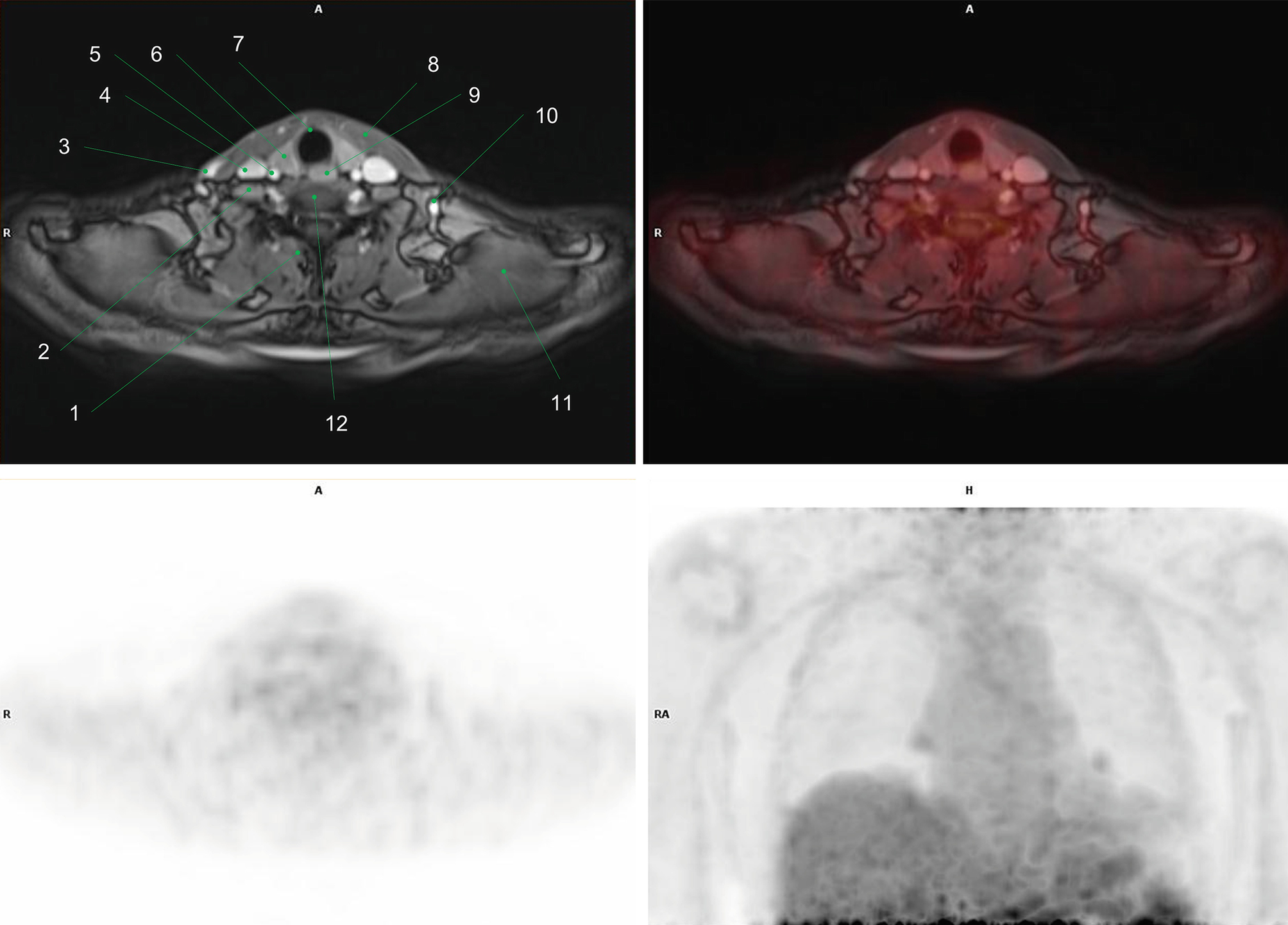
1. Tongue
2. Right molar teeth with artifact due to dental implant
3. Right masseter muscle
4. Right mandibular ramus
5. Right medial pterygoid muscle
6. Right palatine tonsil
7. Right external maxillary vein
8. Right parotid gland
9. Metabolically active LN metastasis, right neck level II
10. Oropharynx
11. Spinal cord
1.7 Case 7

1. Metabolically active tumor in the right sublingual space
2. Metastatic LN, right neck level II
3. Genioglossus muscle
4. Epiglottis
5. Larynx
6. Cervical vertebral body
7. Right sternocleidomastoid muscle
8. Posterior cervical muscles (inner to outer): semispinalis, splenius cervicis, and splenius capitis
9. Left submandibular gland
10. Right common carotid artery
11. Left sublingual space
12. Mandible, body
1.8 Case 8

1. Metabolically active epiglottic tumor
2. Intrinsic tongue muscles (genioglossus)
3. Left submandibular gland
4. Spinal cord
5. Pituitary gland and stalk
6. Sphenoid sinus
7. Nasopharynx
8. Uvula
9. Oropharynx
10. Larynx
1.9 Case 9

1. Metabolically active right parotid tumor, involving both superficial and deep lobes
2. Right masticator space
3. Spinal cord
4. Left parapharyngeal space
5. Left carotid space
6. Left paraspinal space
7. Left buccal space
8. Oropharynx
9. Longus capitis muscles
1.10 Case 10

1. Metabolically active hypopharyngeal tumor
2. Hypermetabolic metastatic neck lymph nodes: left, level III
3. Hypermetabolic metastatic neck lymph node: left, level II
4. Hypermetabolic metastatic neck lymph nodes: left, level IV
5. Right carotid artery
6. Retropharyngeal space
7. Right vertebral artery
8. Epiglottis
9. Left submandibular gland
10. Left carotid artery
11. Left jugular vein
12. Trachea
13. Left thyroid lobe
14. Left common carotid artery
15. Left second rib
16. Vocal cords
1.11 Case 11

1. Diffusely increased activity along the spinal cord
2. Focal increased activity at the vocal cords, physiologic
3. Sphenoid sinus
4. Clivus
5. Nasopharynx
6. Uvula
7. C2, odontoid process
8. Nuchal ligament
9. Oral cavity
10. Oropharynx
11. Left parapharyngeal space
12. Left parotid gland
13. Left vertebral foramen in C1
14. Brainstem
15. C6–C7 Intervertebral disc
16. Trachea
17. Cerebellum
18. Cisterna magna
19. Ill-defined high signal intensity lesions (T2WI)
20. Right vertebral artery
21. Hard palate
22. Longus capitis muscles
23. Left vertebral artery
24. Cerebrospinal fluid
1.12 Case 12

1. Metabolically active recurred tumor in the surgical flap
2. Cerebellum
3. Lateral ventricles
4. Thalami
5. C2, odontoid process
6. Nuchal ligament
7. Medulla
8. Epiglottis
9. Tongue
10. Nasopharynx
11. Left occipital condyle
12. Left cervical paraspinal muscles (multifidus, longissimus capitis, splenius capitis)
13. Right maxillary sinus
14. Right masseter muscle
15. Right mandibular ramus
16. Left nostril
17. Left pterygoid muscles
18. Left cerebellar hemisphere
19. Nasal septum
20. Prevertebral muscles (longus capitis and rectus capitis muscles)
21. Proximal vertebral arteries
22. Surgical graft
23. Left mandibular condyle
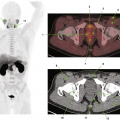
2 Chest
2.1 Case 1

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree