Chapter 5 Assessing Response to Therapy
Introduction
Cancer continues to be a major health problem as one in four deaths in the United States is due to cancer. However, we continue to see incremental improvements over time with the relative 5-year survival rate for cancer in the United States at 68%, up from 50% in the mid 1970s. Cancer death rates fell 21.0% among men and 12.3% among women during the 1991 to 2006 period in the United States. The American Cancer Society estimates that the cancer incidence decreased 1.3%/yr among men from 2000 to 2006 and 0.5%/yr from 1998 to 2006 among women. This decline is attributed mainly to falling smoking rates, improved cancer treatments, and earlier detection of cancer.1
For both the individual patient and clinical trial patients, close communication between the interpreting doctor and the referring physician is necessary for deciding the most appropriate imaging technique to use and when to perform a follow-up study to assess response. Appropriate care in planning the imaging component of clinical trials is essential, which may include proper imaging techniques, analysis, reporting, image transfer, and designing forms that may need to be filled out for these studies. Ideally, these imaging modalities and measurements are identical in both individual and trial patients, which may make clinical imaging research easier to perform or even to incorporate an individual patient into a clinical trial. Such planning will avoid added costs of repeat imaging or the need to go back and reanalyze images. Many of these imaging strategies could be made easier by accreditation of the imaging facility by the ACR (American College of Radiology) because it will ensure that the imaging equipment and qualifications of staff and physicians are registered, which then makes it easier to participate in clinical imaging trials such as ACRIN (American College of Radiology Imaging Network). Ensuring the high quality of imaging benefits primarily our patients but also allows easy participation in clinical research, which is the foundation of continuing improvement in our various specialties.
General Imaging Strategies
Key Points
• Multidisciplinary approach to cancer care will require multimodality, subspecialty imaging.
• Novel therapies will require improved imaging indices to assess extent of disease and response.
• Multimodality imaging expertise and rapid communication between physicians and reporting are a must.
• Integration of imaging into clinical trial planning and accreditation is encouraged.
Why do We Need to Monitor Tumor Response?
History of an Evolving Imaging-based Response Assessment
An early study to assess response was done by Moertel and Hanley,2 in which 16 experienced oncologists were asked to measure 12 simulated tumors, placed underneath foam, using their clinical methods, which entailed physical examination with a ruler or caliper. Although seemingly crude, this was an appropriate simulation of the clinical setting in which a physician will palpate a tumor and then estimate its size before and after administering the treatment. This paper suggested that a 50% reduction in the perpendicular diameters of the tumors done at approximately 2 months is an acceptable objective response rate. This 50% reduction in bidimensional measurement of a single lesion was adopted in the World Health Organization (WHO) guidelines in 1979. Miller3 and coworkers recommended that a partial response be identified if there is a 50% reduction in the bidimensional measure of tumor area or, if multiple tumors are present, the sum of the product of the diameters. This study also described unidimensional measurements for “measurable” disease, bone metastases, and criteria for “nonmeasurable” disease. Tumor volume estimates were based on conventional radiography techniques by measuring the two longest perpendicular diameters and their product. Although widely used, obvious shortcomings of the WHO guidelines were the clinical foundation of the criteria without accounting for the improvements in imaging to determine tumor volumes. Tumors are rarely round or symmetrical, thus making these measurements difficult to implement, particularly by using a ruler or calipers. The lack of distinction between a complete versus a partial response in 50% to 90% decrease in tumor volume was an obvious flaw.
The European Organization of Research and Treatment of Cancer (EORTC) and the National Cancer Institute (NCI) of the United States and Canada set up a study group (RECIST)4 to standardize assessment criteria in cancer treatment trials. The objective was to simplify and standardize the methods to assess tumor response by more precisely defining tumor targets with proposed guidelines on imaging methods. Revisions on complete response, partial response, stable disease, and progressive disease were done. Unidimensional measurements were established for lesions of 2 cm or larger for CT, MRI, plain film, and physical examination and 1 cm or larger for spiral CT scan. The sum of the unidimensional tumor measurements was used for evaluation of response, which may decrease the sources of error.
RECIST 1.1: The Current Standard
The major changes of RECIST 1.1 include the number of lesions required to assess tumor burden for response determination has been reduced to a maximum of five total (and two per organ, maximum). Assessments of pathologic lymph nodes with a short axis of 15 mm are considered measurable and assessable as target lesions. The short-axis measurement should be included in the sum of lesions in calculation of tumor response. Nodes that shrink to less than 10 mm on the short axis are considered normal. Confirmation of response is required for trials with response primary endpoint but is no longer required in randomized studies because the control arm serves as appropriate means of interpretation of data. Disease progression is clarified in several aspects: in addition to the previous definition of progression in target disease of 20% increase in sum, a 5-mm absolute increase is now required as well to guard against overcalling progressive disease when the total sum is very small. Furthermore, guidance is offered on what constitutes “unequivocal progression” of nonmeasurable/nontarget disease, a source of confusion in the original RECIST guidelines. Finally, a section on detection of new lesions, including the interpretation of 2-[18F] fluoro-2-deoxy-D-glucose (FDG)–PET scan assessment, is included. Finally, the revised RECIST 1.1 includes a new imaging appendix with updated recommendations on the optimal anatomical assessment of lesions.5
Although these anatomic criteria have been evolving to affect better response criteria, the RECIST criteria and now, quite likely, the RECIST 1.1 criteria are or will be used in virtually every clinical trial of new solid tumor therapeutics, because response is essentially always measured. Regulatory agencies have accepted RECIST as the standard in response assessment for clinical trials in most countries. Familiarity with the implications of trials in which response is measured using the WHO, RECIST, and RECIST 1.1 criteria is essential because they are not identical and do not produce identical results. Table 5-1 presents RECIST 1.1 overall response criteria for both measurable and nonmeasurable lesions.
Table 5-1 RECIST 1.1 Target Lesions Response Criteria
| Objective response | RECIST 1.1 target lesions* change in sum of LDs, maximum of two per organ up to five total. |
| Complete response | |
| Partial response | Decrease in target LD sum ≥ 30%, confirmed at 4 wk. |
| Progressive disease | |
| Stable disease | Does not meet other criteria. |
CT, computed tomography; FDG, fluoro-2-deoxy-D-glucose; LD, lesion diameter; RECIST, Response Evaluation Criteria in Solid Tumors.
* Measurable lesion, unidimensional (LD only: size with conventional techniques ≥ 20 mm, with spiral CT ≥ 10 mm; nodes: target short axis ± 15 mm, nontarget 10- to 15-mm nodes, normal < 10 mm). Nonmeasurable: all other lesions, including small lesions; evaluable is not recommended.
From Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228-247.
Highlighting Sources of Errors in Response Evaluation
Possible Problems Related to Image Acquisition
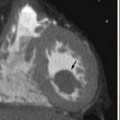
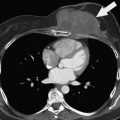
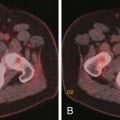
Multiple imaging modalities are utilized and are continuously updated over time, presenting differences in equipment capabilities from within a manufacturer and across competing imaging equipment makers. CT has undergone significant evolution in a short period of time and it is the most commonly used modality for therapeutic clinical trials. Image acquisition protocols vary as to body region or organ being evaluated. The proper phase of contrast has to be consistently used, as is proper windowing and ensuring that the entire tumor or organ is imaged (Figure 5-1
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree