Chapter 11 Pancreatic Ductal Adenocarcinoma
Epidemiology and Risk Factors
Worldwide, approximately 230,000 people are afflicted yearly with pancreatic cancer. It is the fourth leading cause of cancer-related deaths in the United States, as reported by the American Cancer Society, for 2009. It is estimated that 33,000 to 34,000 patients will die of pancreatic cancer in the United States, representing approximately 6% of total U.S. cancer deaths.1
Pancreatic cancer is typically a cancer of the elderly. Only 13% of cases occur in patients younger than 55 years, whereas 69% of cases occur in those older than 65.2 There is a slight predilection for men over women in most countries.3 It has been speculated that, although there may be a hormonal element accounting for this, it is more likely secondary to the higher rate of smoking for males.4
Tobacco smoke exposure plays a significant part in the development of pancreatic adenocarcinoma in as many as 20% to 30% of pancreatic cancers.5 A study of 194 patients showed that the risk of pancreatic cancer doubled in those with a history of smoking.6 In contrast, although excessive alcohol consumption is a common risk factor for chronic pancreatitis, it has not been shown to result in an increased risk of pancreatic cancer.4
The relationship of diabetes to pancreatic cancer is complex. Pancreatic cancer itself can induce diabetes secondary to its destructive effects on the pancreatic parenchyma. Nevertheless, a meta-analysis of 20 studies showed a doubling of the pooled relative risk of pancreatic cancer in those patients with diabetes for 5 years.7 The findings suggest that mechanisms for this increased risk included insulin resistance, elevated insulin levels, and decreased glucose tolerance. This may also explain the finding that moderate physical activity, such as walking as little as 1.5 hours every week, was associated with reducing the risk of pancreatic cancer by half.8
Findings have been mixed regarding the impact of diet on the incidence of pancreatic cancer.9,10 Whereas some studies suggest excessive intake of saturated fat may have a detrimental effect,9 others have failed to show such a pattern.10
The activation of oncogenes and the inactivation of tumor suppressor genes play a significant role in the manifestation of pancreatic cancer.11 Mutations such as DPC4, p53, and p16 are each present in over half of all cases. Overall, it has been estimated that between 5% and 20% of pancreatic cancers are hereditary, depending on the population that is being studied.2,12 The degree of risk increases greatly with the number of affected family members.13
A variety of syndromes are also associated with an increased risk of pancreatic cancer. Individuals who are carriers of the germline BRCA2 mutation have up to a 10-fold greater risk of developing pancreatic cancer over the general population. Other syndromes, and their associated genetic alteration, include hereditary pancreatitis (PRSSI), hereditary nonpolyposis colorectal cancer—the Lynch II variant (hMSH2, hMLH1), familial atypical multiple mole melanoma (FAMMM) syndrome (p16), Peutz-Jeghers syndrome (STK11/LKB1), and ataxia telangiectasia (ATM).2
Anatomy and Pathology
The pancreas, measuring between 12 and 15 cm in length, is located deep within the retroperitoneum and lacks a capsule, which allows surrounding fat to extend into clefts between glandular components.14
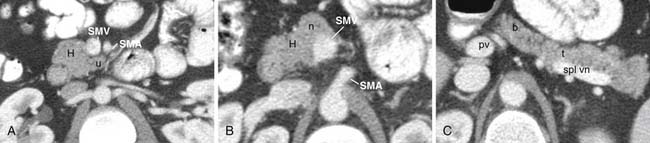
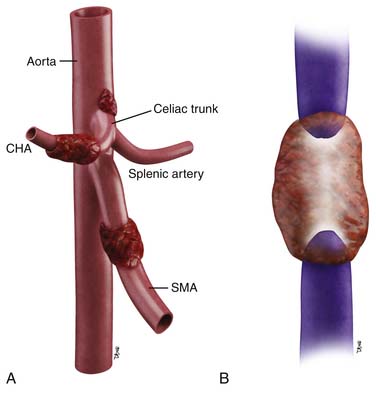
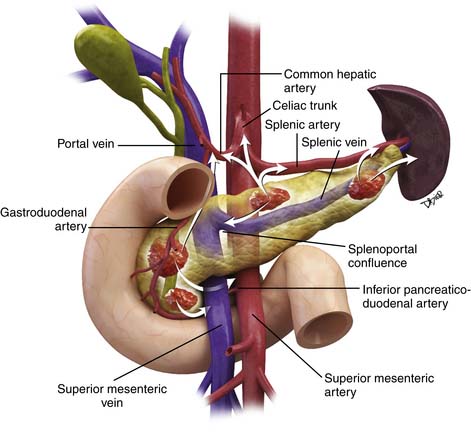
The pancreas can be divided into five components: the head, uncinate process, neck, body, and tail (Figure 11-1). It is bounded on the right by the descending duodenum, inferiorly by the horizontal duodenum, and anteriorly by the stomach. The pancreatic head is defined as that portion to the right of the left border of the superior mesenteric vein (SMV). Extending to the left of the pancreatic head is a narrow projection called the uncinate process posterior to the SMV. In contrast, the neck is a narrowed segment that joins the pancreatic head to the body. The body is defined as that portion located between the left border of the SMV and the left border of the aorta. The tail extends from the left border of the body laterally into the region of the splenic hilum.14 The pancreatic head is bounded on the right by the descending portion of the duodenum and inferiorly by the horizontal portion of the duodenum. The pancreatic body is located posterior to the stomach. Vascular structures also provide important boundaries of the pancreas that can be utilized to localize disease either inside or outside of the pancreas. The gastroduodenal artery, originating from the common hepatic artery (CHA), forms the right lateral and anterior boundary of the pancreatic head. The inferior pancreaticoduodenal artery courses along the posterior border of the pancreatic head and the SMV, as described previously, is an important landmark.
The arterial supply of the pancreas is as follows: the gastroduodenal artery from above and the inferior pancreaticoduodenal artery from below supply the anterior and posterior pancreaticoduodenal arcades to supply the pancreatic head and the uncinate process. Branches of the dorsal pancreatic artery, which typically arises from the celiac trunk, also provide supply to the pancreatic head and anastomose with the pancreaticoduodenal arcade and small vascular branches from the gastroduodenal artery and CHA. The pancreatic body is supplied by branches of the dorsal pancreatic artery and the great pancreatic artery, which in turn, arises from the splenic artery. Multiple splenic artery branches provide supply to the pancreatic tail.15 Knowledge of the vascular supply is important because it also provides a pathway for spread of tumor.
The venous drainage of the pancreas is via branches that drain back to the SMV and splenic veins, which in turn, fuse to form the portal vein (PV) at the splenoportal confluence posterior to the pancreatic neck.16 The splenic vein runs along the posterior pancreatic body, which is the etiology for frequent occlusion of the structure secondary to pancreatic cancer or pancreatitis.
Both sympathetic and parasympathetic innervations of the pancreas are present. The sympathetic supply controls pancreatic blood flow, and the parasympathetic innervations, originating from the posterior vagus nerve and the celiac plexus, promotes pancreatic secretions.17 Both the parasympathetic and the sympathetic tracts contain nerve fibers that transmit pain. These nerve fibers provide a means for tumor migration; their involvement is also a source of debilitating pain.
Histologically, the pancreas is composed of both exocrine and endocrine cell types. The exocrine component consists of acinar and ductal cells responsible for a variety of digestive enzymes and bicarbonate that are ultimately emptied, at the ampulla of Vater, into the duodenum to facilitate digestion.17 The endocrine cellular types include alpha cells (that produce glucagon), beta cells (which produce insulin), delta cells (which produce somatostatin), and PP cells (that produce pancreatic polypeptides).17
Ductal adenocarcinoma is the most common solid malignant neoplasm of the pancreas. This tumor type is an invasive epithelial form that, at least focally, shows sites of ductal or glandular differentiation.18 Typically, it induces an intense desmoplastic reaction made up of a variety of cellular types including myofibroblasts, inflammatory cells, such as lymphocytes and plasma cells, in addition to dense collagen.19 The tumor cells in the well-differentiated type form well-defined glands with mild pleomorphism.18 Moderately differentiated forms exhibit poorly defined glands, whereas poorly differentiated forms do not show well-defined glands. The poorly defined forms grow as individual cells or in sheets and with extensive nuclear pleomorphism.20 Growth tends to be haphazard and most forms show vascular invasion and lymphatic as well as perineural invasion.20
On gross examination, these tumors form firm, but poorly defined, masses that are white to yellow in color. They are typically of variable size, and vary from poorly seen small forms to those that are large, with central regions that can be necrotic, show cystic change, or manifest mucinous features.20
Clinical Presentation
Because the pancreas is located deep within the retroperitoneum, clinical symptoms typically do not manifest until tumor has involved local vessels, caused perineural infiltration, or in pancreatic head tumors, biliary obstruction. Therefore, patients usually present with jaundice, weight loss, and/or abdominal pain.21 In addition, symptomatology will depend on a tumor’s location within the pancreas and extent of metastatic disease. Tumors of the pancreatic head, neck, and sometimes, the body often obstruct the common bile duct, causing jaundice. Tumors in the pancreatic tail often cause left-sided pain, whereas those in the pancreatic body often manifest with midepigastric pain.22 Other symptoms include fatigue, new-onset diabetes, and steatorrhea related to pancreatic insufficiency.17,22 There should be a high suspicion for pancreatic cancer when patients without discernible risk factors present with pancreatitis.23
Unfortunately, symptoms are typically nonspecific, resulting in a median time of 6 months between onset of symptoms and presentation.17 Uncommon symptoms include nausea and vomiting secondary to gastric outlet obstruction and increasing abdominal distention secondary to accumulation of ascites.2 Unexplained progressive weight loss is a common symptom and can be related to jaundice, nausea, or catabolic syndrome and is noted in patients with resectable as well as advanced pancreas cancers. Patients with pancreatic cancer are prone to hypercoagulable states including deep venous thrombosis, pulmonary embolism and less often aseptic/marantic endocarditis—sometimes these are part of the initial presentation.
Physical signs may be very limited. Cachexia with temporal wasting is noted in patients with advanced disease and significant weight loss. Only a third will have a palpable gallbladder (Courvoisier’s sign), and an even smaller percentage will have other palpable masses or ascites.24 Virchow’s (left supraclavicular) or Sister Mary Joseph (periumbilical) adenopathy may be present.2
Biliary obstruction typically causes elevated serum bilirubin levels and transaminitis. Liver function tests can be significantly abnormal with extensive liver metastases as well. Serum amylase may be normal, but when greater than 300, advanced disease is typically present. Often, serum glucose levels are mildly to moderately elevated.2,25
A variety of tumor markers have been considered for screening for pancreatic cancer such as carcinoembryonic antigen (CEA), CA50, and cell adhesion molecule 17-1 (CAM 17-1), but the most useful so far has been CA19-9.2,26 Unfortunately, CA19-9 is not specific for pancreatic cancer but does serve as a prognostic indicator in some patients. It is elevated in a variety of gastrointestinal tumors including stomach, colon, and biliary tree and is often elevated in the setting of biliary obstruction alone.2 In addition the 10% of patients who are Lewis antigen a and b negative have undetectable or markedly suppressed levels of CA19-9.27
Staging Classification
Accurate staging is vital to stratify patients correctly to the most appropriate care, given the severe prognosis of this cancer. It is important to balance the need to give potentially curative surgery to the small percentage of patients who may benefit, at the cost of remaining months of fair to good quality of life with postoperative morbidity in those patients who undergo surgery without potential benefit because of locally advanced or metastatic disease. A review of cases from the United States from 1996 to 2002 shows that the vast majority had metastatic or regionally advanced disease with only a small fraction being resectable.28 When patients presenting with all stages are pooled together, it is found that 5-year survival overall is only 5%.29 Importantly, surgery provides no survival benefit if microscopic or gross positive margins are present.30,31
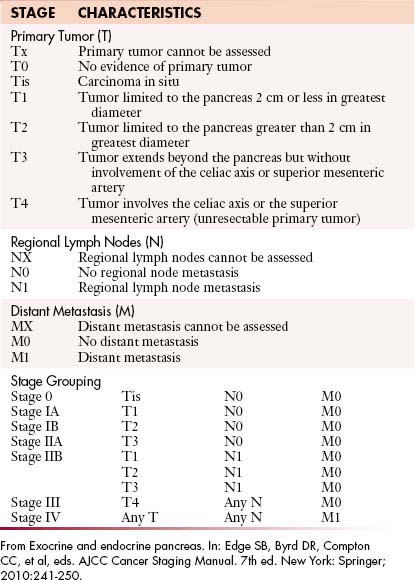
The staging system most extensively utilized is that from the American Joint Committee on Cancer and the International Union Against Cancer (Table 11-1). This system evaluates the primary tumor (T), the presence or absence of nodal disease (N), and the presence or absence of metastatic disease (M), to come up with TNM grades that are, in turn, utilized to determine staging.
Also important is pathologic staging that, by definition, is limited to patients who have undergone surgery. This assessment alone provides truly accurate information regarding nodal disease. Full assessment includes examination of the surgical specimen with no less than 10 lymph nodes resected from the region including those along such major vasculature structures as the CHA and celiac trunk as well as the regions near the distal stomach/pylorus and near the splenic hilum.26
Primary Tumor (T)
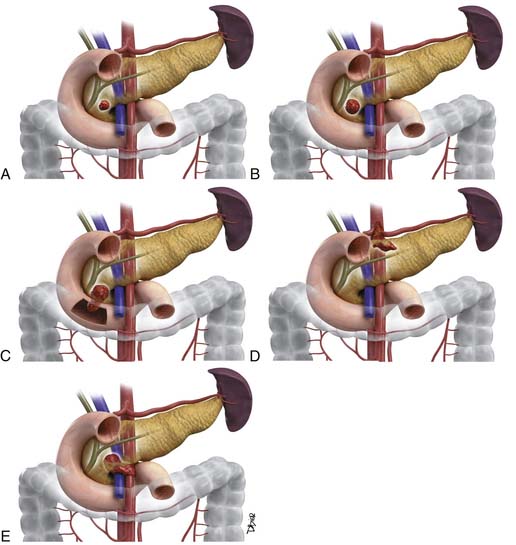
As shown in Table 11-1 and Figure 11-2, T staging is based on tumor size, whether it is limited to the pancreas, and whether tumor involves major local arterial structures (the SMA and celiac artery) (Figure 11-3). Tumors that are 2 cm or less in size but confined to the pancreas are identified as T1, but called T2 if more than 2 cm in diameter (while still confined to the pancreas). Once there is extension beyond the confines of the pancreas, tumor is graded at T3. When that extension is such that it involves the celiac axis or the SMA, it is identified as T4, which is unresectable disease in essentially all medical institutions. T3 disease is considered to be in the realm of resectable disease, but this will vary with the institution. It is notable that originally venous involvement was considered to be unresectable disease; surgical advances such as the venous bypass graft have allowed for en bloc resection.32 Nevertheless, such surgical techniques are not practiced at all institutions. Therefore, it is imperative to understand the local criteria for accepting patients for potential surgical resection. For example, some institutions may identify venous occlusion as an absolute contraindication. Others, such as our institution, have developed a criterion of “borderline” resectable pancreatic disease. These are patients in whom the primary tumor meets certain criteria, and in our case, in the setting of preoperative therapy, are considered to be possibly resectable after a prolonged course of preoperative therapy.
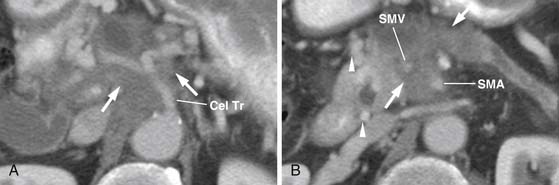
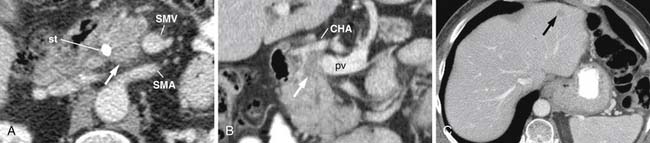
The M. D. Anderson criteria for borderline resectable disease are based largely on the appearance of the tumor on cross-sectional imaging, particularly computed tomography (CT; Figure 11-4). Borderline resectable disease includes abutment of up to 180 degrees of the SMA, short segment encasement/abutment of the CHA (but > 1 cm uninvolved CHA at the level of the origin from the CHA), or venous involvement up to short segment occlusion with suitable conditions above and below this region to allow for placement of a venous bypass graft (Figures 11-5 and 11-6).31 As can be seen from the prior description, the borderline resectable classification supplements or extends the T portion of TNM staging.
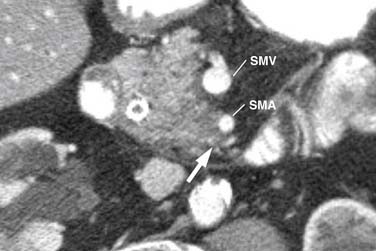
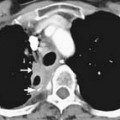
Figure 11-5 Axial contrast-enhanced CT scan shows tumor (arrow) abutting over slightly less than 180 degrees of the SMA.
Nodal Disease (N)
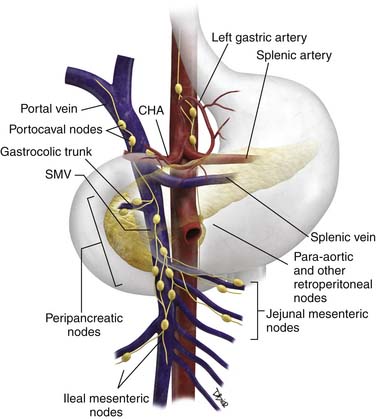
The N descriptor in TNM staging refers to regional nodal disease (Figure 11-7). This criterion is very difficult to assess preoperatively and is described further under “Imaging.” It commonly indicates pathologic staging and, therefore, requires an adequate lymphadenectomy; typically, the histologic evaluation should include no less than 10 regional nodes located in the celiac, CHA, gastric (pyloric), and splenic regions. Recent large studies that performed extended lymphadenectomy including such stations as celiac, greater and lesser omental, portal, SMA, and periaortic nodes have shown no improvement in survival.33,34
Nodal involvement and the more recently described variable of lymph node ratio (number of positive nodes divided by total number examined) have both been described as prognostic factors after resection of pancreatic cancer.35,36 A recent study at our institution of 326 patients showed median survival with nodal status of N0 was 31.9 months and 21.6 months in patients with N1 disease.37
Patterns of Tumor Spread
The most important factor in the spread of a patient’s pancreatic tumor is the location of the tumor within the pancreas (Figure 11-8). This is because of the scaffolding of vessels and neural structures that provide a pathway for tumor to spread outward from its site of origin.38,39
Tumors located within the anterior portion of the pancreatic head extend along the local vasculature, the anterior pancreaticoduodenal arcades superiorly, until reaching their closest supplying vessel, the gastroduodenal artery. Tumor then tends to extend further superiorly along the gastroduodenal artery to its origin from the proper hepatic artery. For this reason, close attention should be paid to this region on cross-sectional imaging for tumors originating in the anterior pancreatic head.
Tumors that are instead located in the much more medially and posteriorly located uncinate process commonly extend along the inferior pancreaticoduodenal arcade to the inferior pancreaticoduodenal artery. Tumor then uses this vessel to infiltrate toward the SMA from which this vessel originates, via a common trunk with the first jejunal artery. Uncinate tumors, therefore, typically involve the SMA before involvement of the celiac trunk or its tributaries. Tumor can also spread via the first jejunal artery into the jejunal mesentery. The SMV is frequently involved and is at risk for occlusion; there also is risk for involvement of the first jejunal venous branch and the ileal branch of the SMV. Tumor can also extend into the ileocolic vasculature.
Typically, the pattern of nodal involvement is that of adjacent regional nodes (see Figure 11-7). Pancreatic head and body tumors are of concern for left gastric, common hepatic, and portacaval nodes, and subsequently, celiac axis nodes. Pancreatic head and uncinate lesions are also of concern for peripancreatic nodes and jejunal mesenteric nodes. Pancreatic body tumors are of concern for left gastric distribution nodes in addition to common hepatic nodes. In contrast, pancreatic tail lesions may be associated with peripancreatic/splenic nodes, although with medial extension, as with body, neck, and head tumors, spread may occur to celiac trunk nodes. Inferior pancreatic head tumors and uncinate tumors can also spread to nodes along the proximal ileocolic vessels where they originate from the ileal vein. With sufficiently advanced disease, tumor can extend to retroperitoneal nodes, and in our anecdotal experience, nodes can occasionally be identified in the periesophageal region.
Metastatic disease typically goes initially to the liver and/or peritoneum. Lesions may initially be very small, requiring careful evaluation by the reading radiologist and close comparison with any available prior studies. Pulmonary metastases typically occur later and, in our experience, can have a variety of forms including small nodules, irregular lesions, small cavitary lesions, and infrequently as lymphangitic spread. Osseous lesions are less frequent, as low as 2.2% in one recent study,40 and in our experience are typically later in appearance than other forms of metastatic disease.