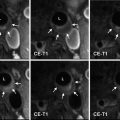
Fig. 1
Axial CT (a) showing acute subarachnoid hemorrhage caused by rupture of a blister aneurysm. The aneurysm was not diagnosed on preliminary CTA, but subsequent DSA (b and c) showed a typical “thorn-shaped” dilation of the internal carotid artery on the anterior-superior surface between the posterior communicating and anterior choroidal artery origins (arrows)
Sequential angiograms often show the aneurysm to enlarge within days of the presenting subarachnoid hemorrhage, which helps to distinguish them from very small non-blister aneurysms [6, 9] (see Fig. 2). This transformation into a larger saccular shape on serial DSA is usually interpreted to indicate dissection or mural hemorrhage as the initiating event for aneurysms at sites other than the internal carotid artery. It creates a potential difficulty in classification for future authors, since blister aneurysms are increasingly managed by endovascular treatment and confirmation of the diagnosis by surgical inspection is not available (see Fig. 3).
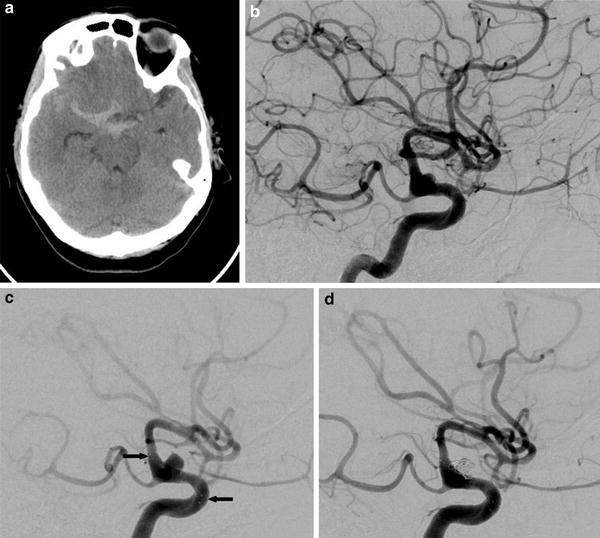
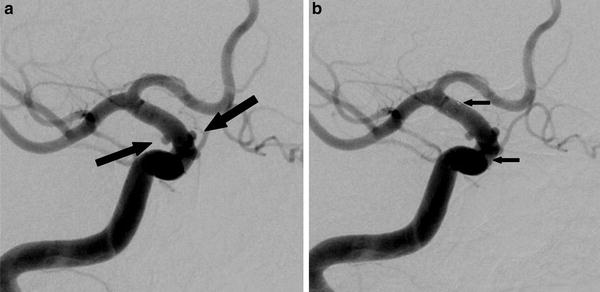

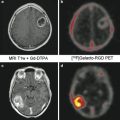
Fig. 2
Axial CT (a) and lateral DSA (b) at presentation 5 days after spontaneous subarachnoid hemorrhage caused by a blister aneurysm of the right internal carotid artery. The aneurysm was treated with a combination of an endovascular stent and endosaccular coils after the patient suffered repeat subarachnoid hemorrhage 12 days later. Control angiograms after deployment of the stent (c) and after insertion of coils (d) show how the aneurysm enlarged in the interval between angiograms and developed a more saccular shape. Small markers can be seen at the two ends of the stent (arrows)

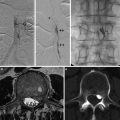
Fig. 3
Unruptured tandem small aneurysms (large arrows) of the right internal carotid artery. DSA performed before (a) and after (b) placing a stent to cover the necks of the aneurysms, which arise from the superior and lateral surface of the artery. Their location is typical of blister aneurysms, but inspection at surgery was not available to confirm the diagnosis. The small arrows show the markers at the ends of the stent (Courtesy of Dr. M. Cellerini)
For the time being, it can be argued that the blister aneurysm is a precursor of a saccular aneurysm, which spontaneous subarachnoid hemorrhage brings to medical attention and after rupture is, for a short period of time, in an unstable state. Whether such aneurysms left untreated inevitably evolve to become saccular aneurysms is unknown, but it has been reported that spontaneous complete regression can occur [14]. In the future, higher resolution imaging than available on current magnetic resonance imagers may be able to better detect mural hemorrhage and provide noninvasive in vivo confirmation of the postulated etiology and diagnosis at other arterial locations.
Management
Blister aneurysms are notoriously difficult to treat regardless of the approach employed, and interventions are associated with overall rates of mortality and morbidity that are significantly higher than those for typical ruptured intracranial aneurysms [9, 15, 16]. The thin, fragile wall of this type of aneurysm, in combination with their small sizes and broad necks, are some of the features that render them particularly prone to intraprocedural rupture when treated using either surgical or endovascular techniques [12, 17]. Blister aneurysms are thus considered particularly dangerous lesions and carry an overall re-rupture risk that is greater than that of most saccular aneurysms [12, 17]. Another feature of these lesions that adds to the high mortality and morbidity rates reported after spontaneous subarachnoid hemorrhage is a frequently observed rapid regrowth of the aneurysmal sac within weeks of apparently initial successful treatment [15]. Blister aneurysms consequently may require multiple treatments, all carrying similar high risks, which significantly increases the total risk of iatrogenic morbidity [15]. Adding to the total risk is the possibility that these small lesions are frequently not diagnosed on primary CT angiography or initial catheter angiograms and are only evident on repeat angiograms after growth of the aneurysmal sac. This delay prolongs the period that patients are at risk of repeat bleeding and additional morbidity if rebleeding occurs [15].
The best treatment of blister aneurysms has, since their first recognition, been controversial, and various strategies have over time been advocated. Most recently, this has centered on the relative strength and weaknesses of open microsurgery compared to endovascular treatments. Parent artery sacrifice (i.e., a deconstructive technique) was for a long time considered the best primary treatment for these lesions and is still by many used as a rescue therapy if other techniques fail. Advances in both microsurgical and endovascular techniques, however, have allowed an increasing proportion of blister aneurysms to be treated preserving the parent artery (i.e., reconstructive techniques) or preserving the blood flow carried through the parent artery to its distal territory through various bypass procedures. These techniques generally appear to offer better anatomical results and patient outcomes than parent artery sacrifice [13, 18].
Other measures in addition to direct interventions to occlude the aneurysm are important adjuvant treatments, which should be considered in order to prevent aneurysm re-rupture. These include tight control of arterial blood pressure, careful drainage of cerebrospinal fluid, and the use of antifibrinolytic drugs such as aminocaproic acid [18].
Most treatment techniques proposed and described in the literature have been directed toward the classical blister aneurysms arising from the internal carotid artery. Similar lesions can, however, as already mentioned sometimes be identified on angiography at other locations. The typical “thorn-shaped” arterial wall dilation (Fig. 1) has for example been demonstrated in aneurysms of the basilar artery, and because of this appearance, these aneurysms have also been termed blister aneurysms [19]. However, since endovascular therapists do not have the possibility of direct visual inspection, it can only be assumed that aneurysms with these appearances at other locations have the same characteristics as those of the ICA.
Surgical Treatment
The thin, frail walls of blister aneurysms easily rupture during surgical dissection or during attempted aneurysm repair [9, 12, 13, 20]. Application of clips directly onto the aneurysm is treacherous, and gentle dissection needs to be performed with careful planning of aneurysm clip placement. Ideally, the clip should be applied completely parallel to the parent vessel, which may be facilitated by clipping under systemic hypotension [9, 12]. Parallel clip placement ensures an even distribution of traction from the aneurysm clip on the vessel wall, minimizing distortion of the wall and allowing a more secure and stable hold of the clip on the wall surrounding the thin-walled and fragile sac of the blister aneurysm [4, 7, 10]. Despite surgical manipulation being performed with the utmost care, avulsion and rupture of the aneurysm wall is not an uncommon occurrence during attempted direct clipping of these lesions [9, 13]. Intraoperative rupture may create a situation where sacrifice of the parent vessel (ICA) to control bleeding is the only possible way to regain control. However, parent artery sacrifice has been shown by Meling et al. to be associated with poor outcomes if performed in SAH patients within 48 h of their bleeding even if collaterals appeared to be adequate on preoperative angiography [13]. These authors suggested that this was due to vasospasm secondary to subarachnoid hemorrhage reducing the available blood flow support to the distal vascular territory via collateral arteries and that parent artery occlusion in the acute phase after hemorrhage should be avoided if at all possible [13].
Due to the high risk of intraoperative rupture of blister aneurysms during attempted clipping, alternative methods for surgical repair have been proposed. Several authors have advocated treatment with various wrapping or combined clipping and wrapping techniques [7, 21]. The vessel-encircling clip graft, or Sundt clip graft, was introduced over 40 years ago, and recently, Park and Meyer described the application of Sundt clip grafts as a rescue technique when vessel dehiscence occurred perioperatively. They achieved good outcomes in 76 % of patients, most of whom were treated for blister aneurysms [22]. Sundt clip grafts have also been successfully applied as a salvage operation after failed endoluminal stenting [23], and their application remains an important tool in the arsenal of surgical treatment techniques for selected patients. However, the encircling clip grafts have important limitations when it comes to the anatomy of the vessel segment around which they are to be applied. The clip grafts cannot be applied to blister aneurysms arising close to branch arteries without occluding them and thereby causing ischemic infarctions and potentially permanent neurological deficits.
Although generally less effective than clipping in preventing re-rupture of saccular aneurysms, different wrapping techniques have been employed successfully by several authors to treat unclippable blister aneurysms [24–26]. Various materials have been used for wrapping including muscle [27], muslin gauze followed by a layer of temporal fibrous fascia [28], and cotton [26]. Blister aneurysms have also been coated with various plastic adhesives [29] including formulated cyanoacrylates, such as Biobond or Aron Alpha [30, 31], and methyl methacrylate [29]. Wrapping using muscle and fascia seems to be of limited value when it comes to preventing re-rupture, most likely because of the gradual necrosis and disappearance of the tissue over time [27, 31, 32]. Wrapping with cotton or gauze, however, either alone or combined with coating the wrapped aneurysm with plastic adhesives, offers better protection over time [32]. A potential disadvantage of using plastic adhesives relates to their potential to cause chronic inflammatory changes with necrosis and fibrosis of the media layer of the vessel wall. Used alone, cotton and muslin gauze cause fibrosis in the adventitia but no changes in the arterial wall media [33]. This selective reaction of the outer layer of the vessel wall is thought to better reinforce the aneurysm sac [27]. Several authors therefore consider cotton used without additional adhesive coating the best material for wrapping ruptured aneurysms [21, 26, 33].
In one study, re-rupture rates for aneurysms wrapped between 1965 and 1975 using muslin gauze with or without additional muscle or adhesives were found to be 8.6 % for early re-rupture, i.e., within the first 6 months, and 1.5 % per annum for late re-ruptures up to 10 years postoperatively [24]. This does not compare favorably to an annual re-rupture risk of around 0.3–0.4 % per year for aneurysms that were clipped in the same period [24]. However, in the cooperative study performed prior to the widespread availability of CT scanning, Nishioka reported a confirmed fatal rebleeding rate of 25 % in untreated patients with all types of intracranial aneurysms [34], and it is thus clear that wrapping of aneurysms offers a significant degree of protection compared to the re-rupture risk of untreated aneurysms. The usefulness of this technique has also been confirmed in the era of microsurgical neurosurgery, and one more recent study reported no rebleeding during a follow-up interval of 5 years for patients with solitary aneurysms wrapped using muslin gauze [25].
A further consideration in treatment planning is the possible late development of chronic granulation tissue after wrapping. Granulomas or so-called gauzomas have been found to sometimes form around wrapped aneurysms [26]. These may cause cranial neuropathies, which is the most frequent complication of aneurysm wrapping apart from re-rupture [26]. To minimize the risk of this happening, the wrapping material should therefore never be placed in direct contact with cranial nerves [21].
Wrapping of blister aneurysms is now usually performed in combination with some form of clip placement. Aneurysmal clips can be placed across the lesion after it has been wrapped with gauze or cotton (e.g., Surgicel) in a technique called “wrap-holding clipping.” Here the wrapping material both reinforces the arterial wall and prevents slippage and displacement of the clip which may cause aneurysm re-rupture. The wrapping material can also be used to encase the arterial segment containing the aneurysm, and an aneurysm clip can then be placed over the wrapping material to hold it in place without incorporating the aneurysm or vessel wall in the clip. This technique is known as “wrap clipping” [35]. Both techniques have been reported to achieve better results for difficult blister aneurysms than clipping or wrapping alone [9]. Wrapping materials commonly in use are cotton (e.g., Bemsheet or Surgicel) [35], rayon [21], Gore-Tex [18, 26], or Dacron [36]. The latter two fabrics are mostly used for wrap clipping.
Delayed aneurysm growth and branch occlusions have been reported after wrap clipping, and frequent follow-up imaging is therefore recommended in particular for patients treated with this technique [35]. Another way to combine wrapping and clipping techniques is to put the wrapping material around a primary clipped lesion. Similar to wrap-holding clipping, this is also performed in order to prevent slippage of the clip, to strengthen the arterial wall, and to provoke formation of scar tissue over time [18]. This technique has been used as a salvage operation together with arterial suturing or tying the blades of a clip to the carotid trunk with silk sutures following perioperative rupture of blister aneurysms during primary clip placement [20].
Over recent years, it has become increasingly clear that blister aneurysms of the supraclinoid internal carotid artery may involve a large portion of the vessel circumference. Often nearly half (i.e., 180°) of the vessel’s circumference is directly involved in the aneurysm with its characteristic diseased thin and fragile vessel wall. This leaves a very limited amount of normal vessel wall onto which it is possible to place aneurysm clips and can make clip reconstruction, with preservation of a vessel diameter sufficient to sustain adequate perfusion in the territory supplied by the artery, impossible [12]. Extracranial to intracranial (EC-IC) bypass followed by parent artery occlusion or trapping has therefore been increasingly advocated by several authors as a means of treating blister aneurysms [13, 18, 37, 38]. Various bypass techniques are used depending on the magnitude of the blood flow the bypass needs to sustain and the inherent availability of collateral blood flow. A low-flow bypass from the superficial temporal artery (STA) to the middle cerebral artery (MCA), a so-called STA-MCA bypass, can only provide limited supportive blood flow. A high-flow bypass between the external carotid artery (ECA) and the MCA with an interposed arterial or venous graft (e.g., radial artery (RA) or saphenous vein (SV)) can provide much more substantial blood flow. When treating aneurysms of the supraclinoid internal carotid artery (ICA), a high-flow bypass such as the ECA-RA-MCA bypass is usually required [37, 38].
In one small study, 4 out of 5 patients with ruptured internal carotid artery blister aneurysms had good outcomes after treatment with ECA-RA-MCA bypass surgery and parent artery occlusion [38]. A temporary low-flow STA-MCA bypass was used during the procedure to provide adequate perfusion to the MCA territory while the permanent anastomosis between the ICA and MCA using a RA graft was established (so-called double-insurance bypass) [38]. This is a complex procedure, but the authors’ results were in line with the results from a large systematic review of different aneurysms in the anterior circulation treated with EC-IC bypass surgery, in which good outcomes (mRS 0–1) were reported in 81 % of the patients [39]. The complication rate with this kind of bypass surgery, however, especially in the setting of acute subarachnoid hemorrhage, is significant, and 25 % of patients in the same review had greater neurological deficits after surgery than before, mostly due to thromboembolic events [39]. Because of this high complication rate, Spetzler and colleagues have recently stated that they consider the EC-IC high-flow bypass and parent artery occlusion as the last option for treating blister aneurysms to be used only after failure of other techniques, i.e., primary clipping or wrap clipping [18]. They point out that although good results are obtainable using bypass surgery, the complication rates of this kind of surgery immediately after subarachnoid hemorrhage remain high in clinical practice [18].
Endovascular Treatment
The standard endovascular treatment for ruptured intracranial aneurysm is performed by packing the aneurysm sac with platinum coils (i.e., endosaccular coiling). This approach, however, like standard microsurgical clipping is not optimal for treating blister aneurysms. Attempted endosaccular coiling of these lesions has at best yielded mixed results and is often not possible because of their broad-based shapes and small sizes [13, 17]. Both these features make it difficult for coils to be retained within the aneurysm sac, and treatment of small-sized aneurysms is generally associated with higher rates of periprocedural rupture. Thus endosaccular coil embolization carries a high risk of coil herniation into the parent artery as well as intraoperative aneurysm rupture [16]. The wide dome and inherent recurrence tendency of blister aneurysms also prevent balloon remodeling from achieving more permanent aneurysm occlusion [17, 40]. Consequently, most blister aneurysms are unsuitable for endosaccular coiling, and parent artery occlusion has historically been considered the more practical and safer endovascular treatment option [17].
However, for the reasons already mentioned, outcomes after parent artery sacrifice are often poor in the acute period after subarachnoid hemorrhage unless supported by bypass surgery [13, 15], even if the native collateral blood flow capacity appeared sufficient when assessed before treatment [13]. Parent artery occlusion also carries the inherent risk of occluding important side branch arteries of the internal carotid artery [15], and therefore in many centers microsurgical repair remained the primary treatment approach for blister aneurysms until self-expanding endovascular stents for intracranial use became available on the commercial market.
Combining endosaccular coil embolization with stenting has several potential advantages. The most important in the context of blister aneurysms is that stents deployed in the parent artery act to retain coils in the aneurysm sac. Coil and stent treatments are performed either by first placing the stent and then introducing coils through the struts of the stent or by catheterizing the aneurysm sac and then placing the stent over the catheter before introducing endosaccular coils into the aneurysm (so-called “jailing” technique) (see Fig. 2). Stent-assisted coiling has allowed a significantly larger proportion of blister aneurysms to be treated successfully with an endovascular approach, and good clinical outcomes have been reported in 62–89 % of patients [16, 40–42]. A majority of treated patients, however, still had to have more than one procedure, either for placement of a second stent within the first or placement of additional coils after aneurysm regrowth becomes evident on follow-up imaging [40, 41]. Early postoperative or perioperative hemorrhage, most often fatal, has also been reported to occur in 13–28 % of blister aneurysm patients treated using stent and coil techniques [16, 41, 42]. The results of initial attempts to utilize stents to augment endovascular treatment by endosaccular coiling were mixed.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree