Infratentorial tumors
Astrocytoma
Medulloblastoma
Ependymoma
Brainstem glioma
Supratentorial tumors
Intraparenchymal
Astrocytoma
Ependymoma
Ganglioglioma/ganglioglioma
Desmoplastic neuroepithelial tumor (DIG)
Primitive neuroectodermal tumor (PNET)
Rhabdoid tumors
Atypical teratoid/rhabdoid tumors
Sellar/suprasellar
Craniopharyngioma
Astrocytoma
Rathke cleft cyst
Germ cell tumors
Extra-axial
Choroids plexus papilloma/carcinoma
Epidermoid/dermoid
Arachnoid cyst
15.2 Neuroimaging of Brain Tumor
Neuroimaging plays a pivotal role as noninvasive tool for the evaluation of pediatric brain tumors, from diagnosis to patient follow-up, allowing the planning of individualized therapy and patient management. In recent years, attention has focused on imaging methods that examine specific tissue properties relevant to the tumor biology, with the aim to elucidate unresolved clinical questions when using conventional structural imaging.
Clinicians expectations from neuroimaging concern the following critical points:
Diagnosis and accurate localization of brain tumor and staging
Definition of tumor borders, extent, and volume
Grading
Guide biopsy and treatment planning
Evaluation of residual tumor after therapy; therapy monitoring
Differentiation between tumor/radiation necrosis and tumor/inflammation
Identification of recurrent tumor
Conventional imaging techniques routinely employed are magnetic resonance (MR) and computed tomography (CT), which reveal morphological tumor features (size and localization) and identify important secondary phenomena such as mass effect, edema, hemorrhage, necrosis, and signs of increased intracranial pressure [1]. MR imaging is the standard neuroimaging method for brain tumor evaluation. Contrast-enhancing components on T1-weighted sequences reflect blood-brain barrier disruption and identify malignant tumor region. Clinical routine MRI characterizes brain tumors primarily by their appearance on T1-weighted images before and after contrast enhancement and on T2-weighted images. Signal abnormalities in T2-weighted/fluid-attenuated inversion recovery (FLAIR) images suggest nonenhancing tumor, perifocal edema, and treatment-related changes such as gliosis or necrosis [2]. However conventional imaging techniques, which demonstrate important structural information with a high sensitivity, have limited specificity, especially after previously applied radiation and/or chemotherapy. Furthermore they fail to provide physiologic and functional information that is crucial for tumor grading, predicting clinical outcome and response to therapy. Otherwise functional and molecular imaging techniques may investigate on tumor cellularity and tissue ultrastructure and tumor metabolism and vascularity, providing noninvasive assessment of tumor biology thus improving the care planning process. These diagnostic tools include magnetic resonance spectroscopy and positron emission tomography (PET) performed with several radiopharmaceuticals. Challenges remain in determining the optimum manner for incorporating these techniques into routine clinical practice, as part of a multimodal protocol.
15.3 Advanced MRI Techniques
Diffusion-weighted magnetic resonance imaging (DW-MRI) is an essential part of MR imaging where T1- and T2-weighted images alone do not provide enough diagnostic information, examining tissue ultrastructure through measurement of the diffusion properties of water [3]. High intensity on DWI indicates restriction of the ability of water protons to diffuse extracellularly. In most tumors there is no restricted diffusion, resulting in a normal, low signal on DWI. However, the peritumoral edema, which exhibits similar diffusion properties as tumor tissue, often precludes precise delineation of the tumor based on diffusion information alone. Diffusion tensor imaging (DTI) is an advanced MR technique that describes the movement of water molecules, useful to study microstructural differences among different tumor types and grades. Indeed tumor type, the degree of invasiveness, and growth rate can affect the diffusion properties [4]. Perfusion MR imaging can play an important role in determining the malignancy grade of brain tumor. The perfusion shows a better correlation with the grade of malignancy than the amount of contrast enhancement [4]. MR spectroscopy (MRS) provides a measure of biochemical changes due to the presence and concentration of various metabolites in tumor that exhibits markedly different spectra from normal brain tissue [5, 6]. It has been shown that brain tumors have decreased N-acetyl aspartate (NAA) signals (neuronal/oligodendrocytic marker) and often also have increased levels of choline (Cho) (increased membrane turnover, cellularity), leading to increased Cho/NAA ratios. The decrease in NAA is interpreted as the loss, dysfunction, or displacement of normal neuronal tissue. The “Cho” signal reflects the increased membrane turnover, correlating with the cellular density and the degree of tumor infiltration into brain. The use of MRS to map Cho levels has therefore been suggested as a method for defining tumor boundaries in treatment planning. Other metabolic changes are elevated signals in the lactate and lipid region of the spectrum and also sometime increased levels of myoinositol (mI), taurine (Tau), glutamine plus glutamate (Glx), myoinositol plus glycine (mI 1 gly), and alanine (Ala) in short echo time (TE) spectra. Taurine has been established as an important biomarker in distinguishing medulloblastomas (the most common posterior fossa neoplasm in children) from other common pediatric brain tumors, such as cerebellar astrocytomas. MRS may provide additional metabolic indices beyond anatomic information for tumor characterization, especially when the differential diagnosis by neuroimaging is difficult, such as in differentiating tumor from infection or radiation necrosis. While the utility of MRS in diagnosis and evaluation of treatment response of brain tumors have been widely documented, MRS has not been widely accepted as a routine clinical tool.
15.4 Nuclear Medicine Imaging
The role of nuclear medicine imaging is increasingly implemented in neuro-oncology, especially if we consider the application in this field of molecular imaging with PET. In fact it may provide important metabolic information of the tumor, promote the development and application of individual treatment, and facilitate prognostic assessment before and after therapy. Different molecular processes have been proposed to be useful for brain tumor evaluation, like glucose metabolism, cell proliferation, membrane biosynthesis, hypoxia, and neo-angiogenesis. The majority of PET radiopharmaceuticals for the evaluation of brain tumor however deepen into increased intratumoral cell proliferation since the evaluation of glucose metabolism in these diseases has important limitations. The single emission tomography (SPET) with gamma emitters plays a marginal and historical role in this setting. In 1990s studies of thallium-201 chloride (201Tl) SPECT in children with recurrent brain tumors had revealed that thallium uptake is increased in pathological areas, but not in radionecrosis with a sensitivity and specificity in tumor detection of 76.9 and 93.3 %, respectively [7]. Tl-avid tumors comprised several histologic types, including astrocytomas/gliomas as well as medulloblastoma and ependymoma. Technetium-99 m hexamethyl-propylene-amine-oxime (99mTc-HMPAO), a perfusion radiotracer, did not prove significant results, due to the extreme variability of HMPAO distribution at the site of tissue abnormality.
Finally, 99mTc-sestamibi scans have been showed to be useful in differentiating neoplastic brain tissue from normal parenchyma. Compared to 201Tl, it offers a better definition of tumor margins and a higher contrast between tumor and background with lower radiation exposure [8]. However, it shows an intense physiological uptake in the choroid plexus of the ventricles, making the detection of lesions in the paraventricular spaces difficult. A relationship between sestamibi uptake and tumor grade was determined in astrocytoma. Brain SPECT with 99mTc tetrofosmin showed comparable results to 99mTc-sestamibi scans [9].
15.5 18F-FDG PET
The glucose analog 18F-FDG is the most frequently used radiopharmaceutical for PET in oncological applications to evaluate the increased metabolic rate of glucose in neoplastic cells. However, it also represents a common pathway of neurochemical activity in the brain providing approximately 95 % of the energy source and being connected to neuronal activity. Consequently the physiological brain uptake is very high in different brain areas, such as the cerebral cortex, the basal ganglia, and the thalamus, which significantly limits the sensitivity for detection as well as the specificity for delineation of adjacent neoplastic tissue. Furthermore not all brain tumors are characterized by increase glucose metabolism, and the metabolic status may vary significantly depending on tumor histopathological grade, proliferative activity, and heterogeneous tissue component (necrosis, hemorrhagic/cystic areas). Spence and colleagues [10] showed that 18F-FDG imaging 3–8 h after injection can improve the distinction between tumor and normal gray matter. 18F-FDG uptake in low-grade tumors is usually similar to that in normal white matter, and uptake in high-grade tumors can be less than or similar to that in normal gray matter, thus decreasing the sensitivity of lesion detection [11–13]. The clinical role of 18FDG PET in pediatric brain tumor is less firmly established compared to its value in adult brain tumors. However, several studies indicate that FDG uptake plays an important prognostic role reflecting malignancy grade and predicting survival. For example, the demonstration of high uptake of FDG in a previously known low-grade tumor suggests the diagnosis of anaplastic transformation. FDG PET may assist stereotaxic biopsy improving the diagnostic yield of the procedure by detecting metabolically active areas of tumor and identify the margins for tumor resection. PET has been shown to be useful for the evaluation of children with brainstem gliomas. Hypermetabolic tumors are more likely to reflect a glioblastoma compared to low or absent 18F-FDG uptake in anaplastic astrocytomas or low-grade astrocytomas. Furthermore tumors with high FDG uptake are associated with a shorter survival time than tumors with absent or moderate FDG uptake. The optimal cutoff levels for distinguishing low- from high-grade gliomas have been reported as 0.6 for the tumor/cortex ratio and 1.5 for the tumor/white matter ratio with sensitivity and specificity of 94 % and 77 %, respectively [14]. 18F-FDG PET may be useful in the differential diagnosis between radiation necrosis and persistent/recurrent tumor. The sensitivity ranges from 75 to 86 % and specificity from 40 to 94 % [15, 16]. Given these results, in up to one-third of the patients, inappropriate treatment would be carried out. Co-registration with MRI may make FDG PET a more sensitive and useful test in distinguishing recurrence from radionecrosis. Despite its recognized limitations in brain tumor imaging, this imaging modality remains the most commonly used tracer nowadays (Figs. 15.1, 15.2, and 15.3).
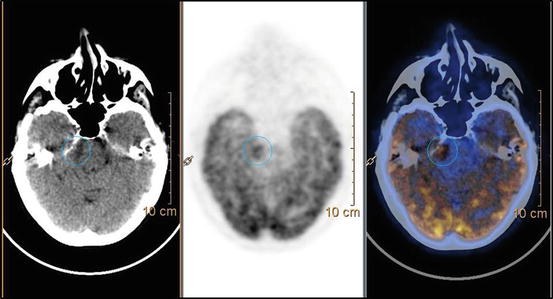
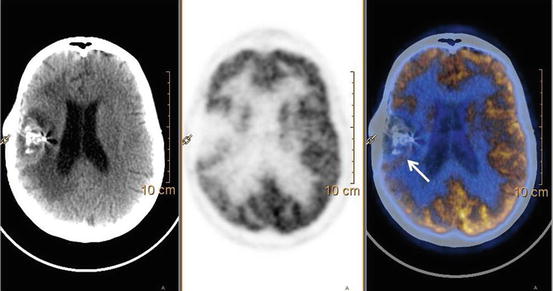
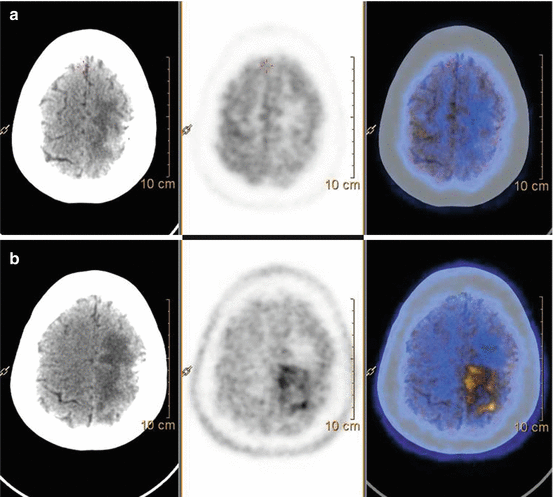

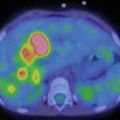
Fig. 15.1
18F-FDG PET-CT in patient with right Gasserian ganglion tumor previously treated. PET finding shows hypermetabolic uptake (blue circle) between the brain stem and the right sphenoid region, consistent with recurrence disease

Fig. 15.2
18F-FDG PET-CT performed in patient with anaplastic astrocytoma of the right temporal region surgically treated shows a faint FDG uptake in the posterior margin of the surgical cavity (white arrow) consistent with recurrent/persistent disease

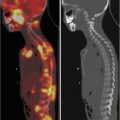
Fig. 15.3
Follow-up imaging in patient with previously left frontal glioblastoma. 18F-FDG PET-CT (panel a) is negative for recurrent disease. 11C-MET PET-CT (panel b) shows abnormal uptake in the left frontoparietal region suspicious for relapse
15.6 Non-FDG PET-CT
Non-FDG brain tumor PET radiopharmaceutical has been developed during the past decades in order to improve the diagnostic sensitivity, specificity, and accuracy of molecular imaging of brain tumors. Appropriate non-FDG tumor radiotracers primarily concentrated on the increased proliferative activity of tumors cells. The upregulation of protein synthesis explored by radiolabeled amino acids is an important molecular target system for the evaluation of tumor growth as well as the DNA synthesis explored by radiolabeled nucleotides and their precursors or the increased of membrane synthesis. Of the radiotracers used for the aforementioned target systems, in brain tumor imaging, the use of 11C-L-methionine (MET) has been reported most frequently, followed by 18F-fluoroethyl-l-tyrosine (FET), 11C-choline, 18F-fluorothymidine (FLT), 11C-acetate, and 18F-fluoro-DOPA.
15.6.1 Amino Acid Transport and Protein Synthesis
Radiolabeled amino acids were introduced in 1982 as suitable PET tracers in brain tumors [17]. The use of amino acids is based on an increased amino acid utilization in cancer cells, which is known to play a pivotal role in cell proliferation, as well as in extracellular matrix synthesis. For this purpose, a variety of radiolabeled amino acids, like 11C-MET and aromatic amino acid analogs, like 18F-fluorotyrosine (18F-TYR), 18F-FET, 18F-fluoromethyltyrosine (18F-FMT), and 18F-DOPA, have been studied. Active amino acid transport is upregulated in the cell membranes of tumor cells, regardless of the phase of the cell cycle, thus not representing a limiting factor for successful PET imaging. High-contrast images can be acquired due to high amino acid uptake in tumors and low uptake in normal brain tissue compared to FDG. Possible problem may be caused by unspecific increase of amino acid uptake due to damage of the blood-brain barrier [18]. Methionine uptake correlates not only with cell proliferation but also with microvessel density, suggesting the possibility of mapping tumor neovascularization. The accumulation of MET in macrophage is irrelevant overcoming the limitation of FDG due to uptake in inflammatory process. The uptake of 11C-MET gradually increased with age until 20 years especially in the frontal lobes and cerebellum, possibly reflecting high activity of the neutral amino acid transporter and brain protein synthesis correlated with brain metabolism. The highest uptake is found in the cerebellum and occipital cortex, lower in the white matter [19]. The clinical application of 11C-MET has been proved both for tumor diagnosis (tumor detection and characterization, extent for treatment planning, biopsy guidance) and for treatment evaluation (therapy monitoring, detection of recurrent/persistent disease). The specificity of MET for brain tumor evaluation is generally high (85–100 %), and tumor/background radio superior to 1.5 is considered indicative of tumor. The use of this radiopharmaceutical is particularly interesting in suspicious low-grade glioma that cannot easily identify with FDG PET. The MET uptake may be overlying among grades I and II glioma and grades III and IV; however, it may provide a prognostic index in patients with low-grade glioma [20]. Furthermore, 11C-MET is the radiopharmaceutical of choice for PET-guided biopsy. The distribution of MET in brain lesion reflects the histological heterogeneity of the tumor, indicating more accurately the areas to be biopsied [21]. It has been shown that MET-guided biopsy reduces the number of attempts to obtained diagnostic samples, providing a more sensitive mark compared to FDG. Another crucial point in the management of brain tumor is the distinction between recurrence and post-therapy changes. The MET PET has been shown to be superior to FDG (sensitivity 100 %, specificity 72 %, PPV 81 %, NPV 100 %) in differentiating viable tumor from radiation injury [22–24]. Infrequently a mild MET uptake may be observed due to the proliferation of glial cell that may be present after radiation therapy. To increase the diagnostic performance, it has been suggest that tumor-background ratio superior to 1.5–2 should be considered indicative of tumor presence. 11C-methionine is probably the tracer of choice at this time, in terms of its sensitivity, specificity, and accuracy, but its major disadvantage is the short half-life of 11C (20 min) which implies on-site cyclotron. 18F-amino acid has been shown comparable results, overcoming the problem of radiopharmaceutical production [25]. 18F-fluoro-ethyl-tyrosine (18F-FET) is an artificial amino acid taken up into upregulated tumoral cells but not incorporated into proteins (contrary to natural amino acids such as 11C-methionine). 18F-FET PET has excellent performance for the diagnosis of brain tumor and glioma in the initial evaluation of newly diagnosed brain lesions. Initial comparison studies demonstrated that 18F-FET uptake ratios correlated with 11C-methionine uptake but with a lesser uptake by inflammatory cells, allowing a better discrimination between infectious and tumoral lesions or between tumor recurrence and radionecrosis [26]. Among other applications, 18F-FET PET has shown value in the diagnosis of brain tumor recurrence after initial surgery or radiotherapy and for directing biopsy or radiosurgery. Furthermore early changes in 18F-FET uptake have been suggested as a surrogate to predict tumor response to treatment and prognosis. 18F-DOPA is also an amino acid analog which is taken up in normal brain by the neutral amino acid transporter. The highest radiopharmaceutical uptake in tumor and cerebellum generally occurred between 10 and 30 min after injection, while the uptake in the striatum reaches the peak until 50 min after injection. 18F-FDOPA has a sensitivity of 96 % and a specificity of 43 %, with an overall accuracy of 83 % for the detection of primary brain tumors [27]. Compared to FDG, 18F-DOPA has shown a better accuracy with a higher sensitivity and a similar specificity, allowing an excellent visualization of both high-grade and low-grade tumors. The contrast between tumor and normal tissue is higher than that with 18F-FDG because of the low 18F-DOPA uptake in normal brain tissue. Tripathi and colleagues [28] in a series of 15 patients demonstrated that 18F-DOPA is superior both to FLT and FDG for detection of primary and recurrent low-grade gliomas. 18F-DOPA should demonstrate good feasibility in detecting local recurrence and residual tumor not clearly seen on MR alone; however, the evaluation of this aspect is still limited in literature.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree