Chapter 25 Cervical Cancer
Introduction
Cervical cancer is the third most common gynecologic malignancy in the United States. In 2011, 12,710 new cases and 4290 deaths1 are expected in the United States. However, in the underdeveloped world where screening remains underutilized, cervical cancer is the second most common cancer in women, with 275,000 deaths worldwide in 2002.2
In the past 50 years, there has been a steep decline in mortality from cervical cancer that can be attributed to the use of the Papanicolaou (Pap) smear, one of the most effective and widely used screening tests. However, exfoliative cytology obtained with the Pap smear permits early detection of only early squamous cell carcinoma; this has led to a relative increase in the incidence of cervical adenocarcinoma.3
Epidemiology and Risk Factors
There are numerous predisposing factors in the development of cervical cancer; however, early epidemiologic studies implicated an infectious agent as the most important factor. In 1983, this was identified as human papillomavirus (HPV). Although HPV infection is widespread, the majority of infections are cleared by cell-mediated immunity within 2 years and less than 10% of individuals will develop persistent infection. It is this persistent infection with HPV that plays a central role in the development of cervical cancer and can be identified in almost all cases. There are numerous HPV genotypes, of which HPV-16 and -18 have been determined as the most potent carcinogens. HPV-16 alone accounts for almost 60% of cervical cancers.3 In addition to its causative role in the development of squamous cell carcinoma of the cervix, HPV has also been implicated in the development of cervical adenocarcinoma and neuroendocrine carcinoma.
1. Multiple sexual partners and high-risk sexual partners.
2. History of sexually transmitted diseases (e.g., chlamydia or herpes simplex).
3. Prolonged use of oral contraceptives.
6. Young age at first intercourse.
Anatomy and Pathology
Cervix
The uterus has three distinct anatomic parts: the corpus or body, the lower uterine segment, and the cervix. The cervix is divided into suprvaginal and vaginal portions. The vaginal portion, or portio vaginalis, is covered by stratified nonkeratinized squamous epithelium, which also lines the vagina. This meets the columnar epithelium of the endocervix at the external os. This is the squamocolumnar junction, or transformation zone, described previously. In this area, a gradual transformation of columnar to squamous epithelium proceeds through life, leading to a gradual migration of this zone from the level of the external os in young women to a position within the endocervical canal in older women. This has consequences on the location of squamous tumors, which may present as endocervical masses in older women.
Pelvic Anatomy
A basic overview of pelvic anatomy is important for staging cervical cancer because an understanding of pelvic ligaments, vessels, peritoneal reflections, and pelvic lymph node stations is vital in evaluating cross-sectional computed tomography (CT) and MRI.4
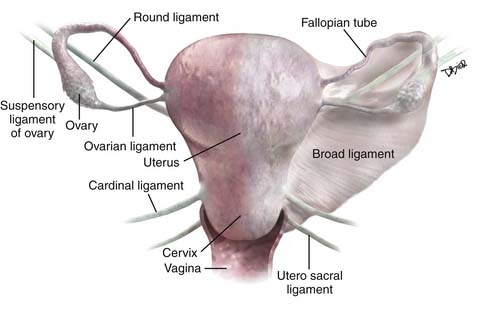
The important ligaments from the imaging perspective that are found in relation to the uterus, cervix, and ovaries include the broad ligament, round ligaments, uterosacral ligaments, cardinal ligaments, and suspensory ligament of the ovary (Figure 25-1).
The broad ligament is a peritoneal reflection that extends from the uterus to the pelvic sidewall. It contains the fallopian tubes along the upper margin; the cardinal ligament runs in the base of the broad ligament. It contains fat, connective tissue, the uterine and ovarian vessels, lymphatics, and the ovarian and round ligament. It is difficult to see except in the presence of ascites. The round ligament runs from the anterior wall of the uterus through the inguinal canal to the labia and is easy to see on CT. The cardinal ligament is an important structure that runs from the cervix and upper vagina to the obturator internus muscle. The uterine arteries run along the superior aspect of this ligament and help define this structure. As these arteries course from their origin from the internal iliac arteries to the edge of the uterus, they arch over the ureters creating the “arc sign,” which can frequently be seen on CT. The uterosacral ligament arises from the cervix and upper vagina to arc on either side of the rectum to the S2-S3 segments of the sacrum. This structure is thickened after radiation therapy. The suspensory ligament of the ovary carries the gonadal vessels to the ovary and is defined by the course of these vessels on cross-sectional imaging.
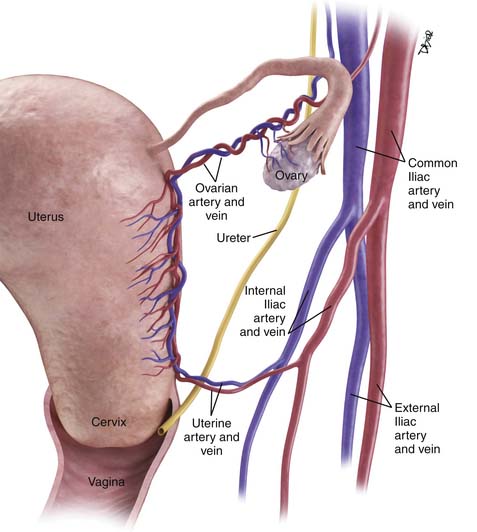
The main vascular supply to the uterus is the uterine arteries that arise from the internal iliac vessels, run along the superior aspect of the cardinal ligament, and then ascend on either side of the uterus to trifurcate and supply the fallopian tubes, fundus of the uterus, and ovaries (Figure 25-2). The ovarian arteries arise from the aorta below the renal arteries; the vaginal arteries arise from the internal iliac arteries.
Pelvic Nodal Anatomy
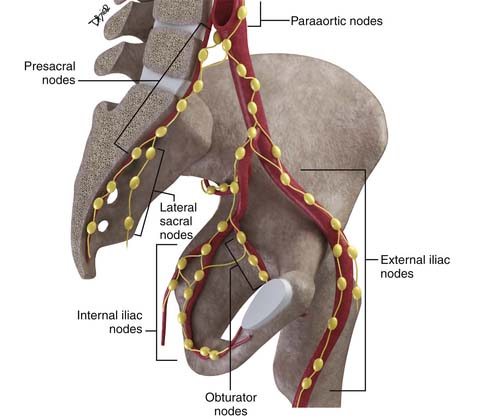
Disease spread to pelvic nodes is the most common pathway of tumor dissemination from the cervix. An understanding of the pathways of spread is essential for image analysis because these are the nodes that should be most closely scrutinized when evaluating cross-sectional imaging studies (Figure 25-3).
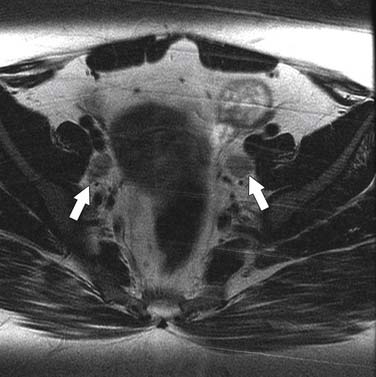
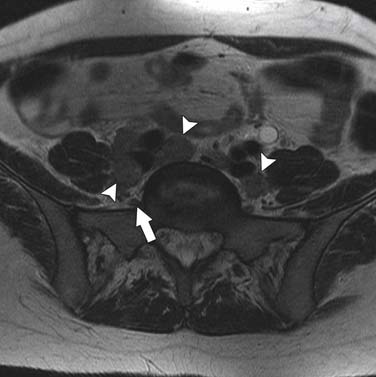
The nodal group most commonly first involved by tumor spread from pelvic tumors is the perivisceral nodes, which in the instance of cervical cancer are the parametrial nodes. This is followed by spread to pelvic sidewall nodes. Lymphatic spread from cervical tumors can spread to the pelvic nodes by three routes: (1) the lateral pathway of spread toward external iliac nodes, (2) the hypogastric route toward nodes along the internal iliac vessels, and (3) the posterior route, where lymphatics course along the uterosacral ligament to nodes along lateral sacral vessels and nodes anterior to the sacral promontory.5 The nodes along the external iliac vessels are subclassified into middle, medial, and lateral groups. The lateral chain nodes are located, as the name implies, lateral to the vessels; the middle chain nodes are between the external iliac artery and vein; and the medial chain nodes are located posterior and medial to the artery and vein. The nodes medial to the external iliac arteries are the group most commonly involved by metastatic spread from cervical cancer. These nodes are located in close proximity to nodes along the obturator vessels and are frequently grouped together with obturator nodes, although there is some controversy in this regard (Figure 25-4). All the pelvic nodal chains drain to the common iliac nodes. The common iliac nodes are also classified similar to external iliac nodes into middle, lateral, and medial subgroups. The middle subgroup is located posterior to the common iliac vein in the lumbosacral fossa, which is bordered posteriorly by the sacral vertebral body.6 This node is in close proximity to the L5 nerve root and can impinge on this root when enlarged, causing back pain (Figure 25-5). Spread from common iliac nodes is most commonly to the para-aortic nodes.
Key Points Anatomy, pathology, and epidemiology
• Persistent infection with HPV-16 and -18 is the most important risk factor in the development of cervical cancer.
• Cervical cancer has a long premalignant phase and, therefore, is accessible to early intervention and screening.
• Squamous carcinoma accounts for 85% of cervical tumors and arises from the zone located at the level of the external os and may present as exophytic or infiltrative masses.
• The uterine arteries and ureters as they course on the surface of the cardinal ligament define the level of the supravaginal cervix and site of parametrial invasion.
Patterns of Tumor Spread
As with all tumors, it is important to define regional and nonregional nodal groups; involvement of the latter upstages the tumor to stage IV because these nodes are viewed as M1 nodes. In the case of cervical cancer, parametrial, internal iliac, obturator, external iliac, common iliac, presacral, and lateral sacral are viewed as regional nodes, whereas inguinal, para-aortic, mediastinal, and supraclavicular are viewed as nonregional nodes, and consequently, qualify as metastatic disease.6
Key Points Patterns of tumor spread
• The most common pathways of tumor spread are direct invasion through the cervical stroma into the parametrium and adjacent pelvic structures and lymphatic spread.
• Nodal spread in cervical cancer occurs in a stepwise progressive manner from the parametrial to external, presacral, or internal iliac nodes followed by common iliac and para-aortic nodes.
• Para-aortic and inguinal adenopathy constitute nonregional nodes and, consequently, metastatic disease.
Staging
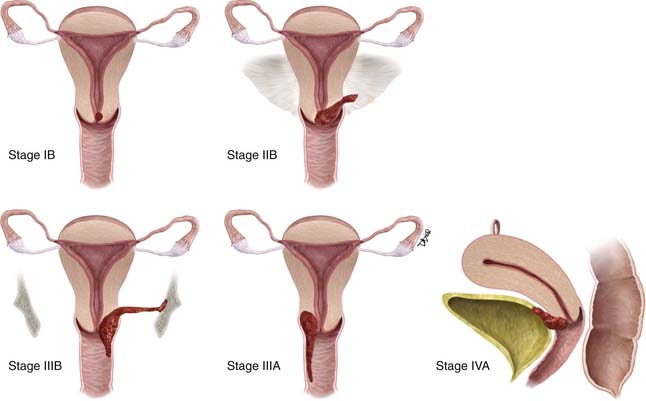
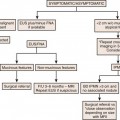
Unlike most other tumors, the staging of cervical cancer is primarily clinical, using the classification by the Federation of Gynecology and Obstetrics (FIGO) committee. This is because, in most countries where this disease is prevalent, elaborate cross-sectional staging techniques such as MRI, CT, and positron-emission tomography (PET)/CT are not available. Consequently, to maintain some uniformity between clinical trials across nations, the basis for initial staging is the clinical stage. The classification has undergone numerous revisions. This chapter references the most recent 2009 revision (Figure 25-6). Some important facts about the FIGO staging is that it applies only to squamous carcinoma; the clinical stage is determined prior to the start of therapy and cannot be changed because of subsequent findings once treatment is started. For evaluation of the T (tumor) stage, the following examinations are recommended: physical examination, preferably as an examination under anesthesia; colposcopy; cystoscopy; proctoscopy; intravenous urography; and chest x-ray. Suspected involvement of the rectal or bladder mucosa must be confirmed by biopsy. The tumor size plays an important prognostic role; consequently, subgroups have been created in both stages I and II for tumors less than 4 cm (T1a&b1 and T2a1) or greater than 4 cm (T1b2 and T2a2) in the tumor-node-metastasis (TNM) and FIGO classifications. It is noted that the definition of T categories corresponds to stages accepted by the FIGO classification (Table 25-1).7 A limitation of the clinical system of staging is that, compared with surgical staging, it can be erroneous in up to 32% of patients with stage IB and 65% of patients with stage III disease.8,9
Table 25-1 TNM & FIGO Staging of Cervical Cancer
| TNM CATEGORIES | FIGO STAGES | |
|---|---|---|
| Primary Tumor (T) | ||
| TX | Primary tumor cannot be assessed | |
| T0 | No evidence of primary tumor | |
| Tis* | Carcinoma in situ (preinvasive carcinoma) | |
| T1 | I | Cervical carcinoma confined to uterus (extension to corpus should be disregarded) |
| T1a† | IA | Invasive carcinoma diagnosed only by microscopy. Stromal invasion with a maximum depth of 5.0 mm measured from the base of the epithelium and a horizontal spread of 7.0 mm or less. Vascular space involvement, venous or lymphatic, does not affect classification |
| T1a1 | IA1 | Measured stromal invasion 3.0 mm or less in depth and 7.0 mm or less in horizontal spread |
| T1a2 | IA2 | Measured stromal invasion more than 3.0 mm and not more than 5.0 mm with a horizontal spread 7.0 mm or less |
| T1b | IB | Clinically visible lesion confined to the cervix or microscopic lesion greater than T1a/IA2 |
| T1b1 | IB1 | Clinically visible lesion 4.0 cm or less in greatest dimension |
| T1b2 | IB2 | Clinically visible lesion more than 4.0 cm in greatest dimension |
| T2 | II | Cervical carcinoma invades beyond uterus but not to pelvic wall or to lower third of vagina |
| T2a | IIA | Tumor without parametrial invasion |
| T2a1 | IIA1 | Clinically visible lesion 4.0 cm or less in greatest dimension |
| T2a2 | IIA2 | Clinically visible lesion more than 4.0 cm in greatest dimension |
| T2b | IIB | Tumor with parametrial invasion |
| T3 | III | Tumor extends to pelvic wall and/or involves lower third of vagina, and/or causes hydronephrosis or nonfunctioning kidney |
| T3a | IIIA | Tumor involves lower third of vagina, no extension to pelvic wall |
| T3b | IIIB | Tumor extends to pelvic wall and/or causes hydronephrosis or nonfunctioning kidney |
| T4 | IVA IV any T/any N/M1 disease | Tumor invades mucosa of bladder or rectum, and/or extends beyond true pelvis (bullous edema is not sufficient to classify a tumor as T4) |
* FIGO no longer includes stage 0 (Tis).
† All macroscopically visible lesions—even with superficial invasion—are T1b/IB.
From Cervix uteri. In: Edge SB, Byrd DR, Compton CC, et al, eds. AJCC Cancer Staging Manual. 7th ed. New York: Springer; 2010:395–402.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree