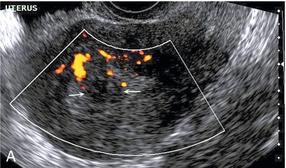
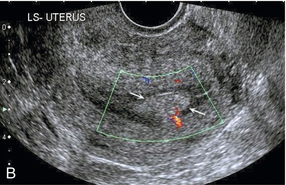
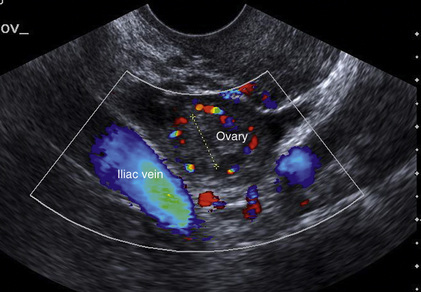
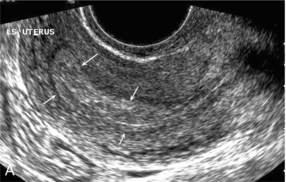
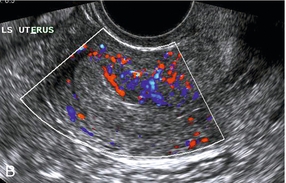
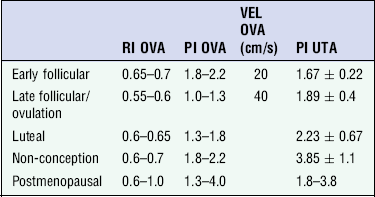
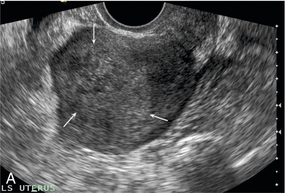
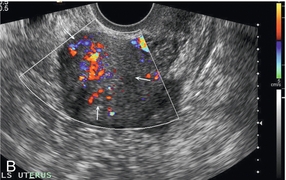
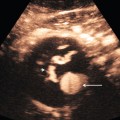
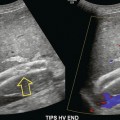
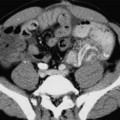
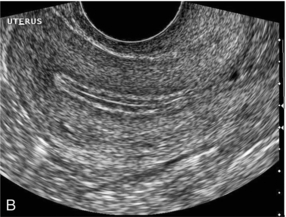
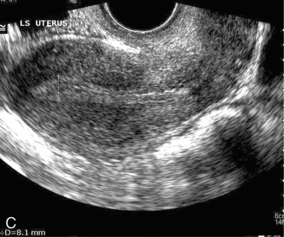
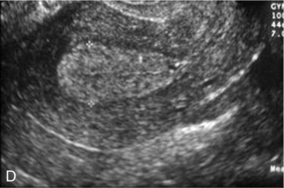
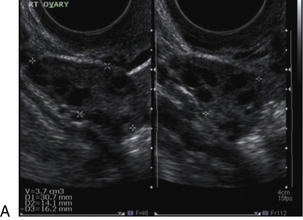
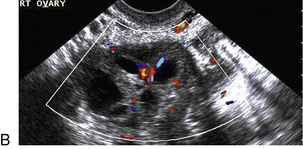
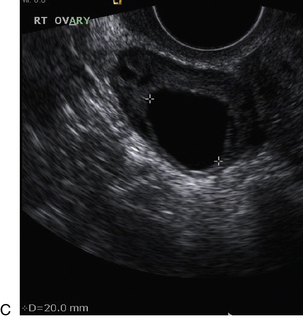
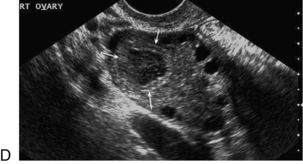
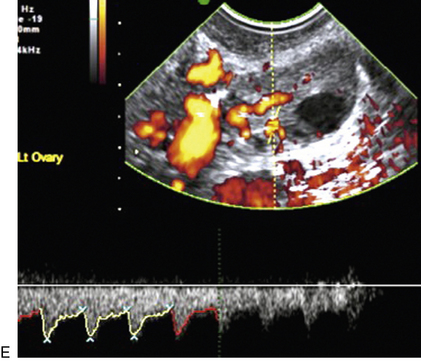
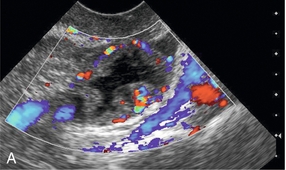
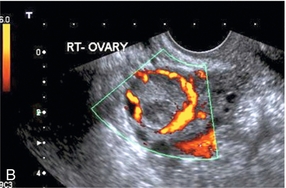
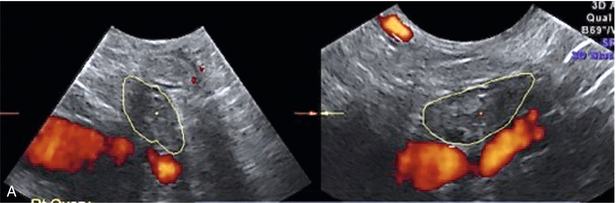
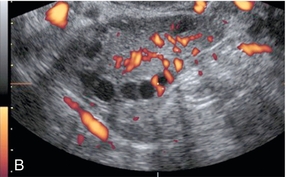
Chapter 14 The iliac artery arises at the aortic bifurcation and courses inferiorly and laterally to emerge at the groin as the common femoral artery. The approximate surface markings for this course are given by a line from the umbilicus to the point of maximum pulsation in each groin. The common and external iliac veins run posterior and medial to their accompanying artery. The paired artery and vein often form a lateral boundary to the ovaries (Fig. 14-1). The internal iliac artery arises medially from the common iliac artery approximately 4 cm from the aortic bifurcation. The internal iliac artery divides into anterior and posterior branches; the uterine artery arises from the anterior branch. The uterine artery runs in the base of the broad ligament medially to the cervix, where it gives branches to the upper vagina and cervix. It ascends in the broad ligament giving off branches to the myometrium until it reaches the cornual region. Here it turns laterally to supply the fallopian tube and ovaries and anastomoses with branches from the ovarian artery. The uterine vein mirrors the artery’s course and eventually drains into the internal iliac vein. The following generalisations apply: Dysfunctional uterine bleeding is one of the most common gynaecological complaints. Many cases will have an underlying hormonal cause but in up to half of these cases ultrasound may reveal an underlying structural defect – the most frequent being submucosal fibroids, adenomyosis or endometrial polyps. The Doppler characteristics of each of these will be discussed later. However, Doppler also has a more general role to play in these cases as it has been shown that women with pain on menstruation have increased myometrial vascularisation during the early menstrual phase,1 and that those with irregular bleeding are also more likely to show increased perfusion of uterine and sub-endometrial blood vessels (Fig. 14-2).2 The pathophysiology underlying these observations is not yet fully understood. Endometrial ablation is a recognised treatment of dysfunctional bleeding but measurement of the Doppler indices in the uterine arteries does not help determine who will show a good response to ablation; although assessment one year after ablation helps predict the duration of amenorrhoea or eumenorrhoea.3 The normal menstrual cycle starts on day one with bleeding heralding the shedding of the endometrium. Ultrasound shows the endometrium reduces to a minimum basal layer and it may occasionally show blood and clot in the cavity. The endometrium then enters the proliferative phase, displaying a distinct trilaminar appearance. As the time of ovulation approaches, the basal endometrium becomes more echogenic; this echogenicity spreading towards the central echogenic line until, in the secretory phase after ovulation, the whole width of the endometrium becomes of equal increased echogenicity (Fig. 14-3). During the proliferative phase, the ovary shows enlargement of a few of the antral follicles, until one becomes dominant (Fig. 14-4). This continues to enlarge until it is about 2.5 cm in diameter at which point the follicle ruptures and ovulation occurs (usually around day 14). The follicle then forms the corpus luteum during the secretory phase. 3D power Doppler has been used to measure the vascular flow index during the normal cycle; the vascular indices in the dominant ovary and the dominant follicle/corpus luteum show an increase in the proliferative phase such that they are 1.7 times greater than in the basal state. After ovulation there is a continued rise in the vascularity with the corpus luteal flow peaking about 7 days after ovulation with an index three times greater than in the basal state. The contralateral, non-dominant ovary in that cycle shows no changes in blood flow.4 Hormonally suppressed ovaries show a low vascular flow index throughout the cycle.5 The resistance index (RI) of the corpus luteum measured by TV colour and spectral Doppler shows that the pre-ovulatory follicle has a high RI, which decreases greatly on ovulation as the corpus luteum is formed. The increased vascularity around the corpus luteum has been likened to a ring of fire (Fig. 14-5). The RI decreases further in the early luteal (secretory) phase but then increases in the late luteal phase. This late increase does not occur if pregnancy has become established, as the RI remains at the low midluteal level until 7 weeks of pregnancy.6 Fertility specialists can use the peri-ovulatory follicular volume and subfollicular vascularisation as predictors of successful pregnancy in intra-uterine insemination techniques – too large a follicle is likely to be anovulatory.7 Furthermore, in oocyte retrieval techniques, it has been suggested pregnancy rates are higher in those whose embryo transfer cohort contains at least one embryo from a highly vascular follicle8 (Box 14-1). FIGURE 14-3 Normal appearances of cyclical endometrium. (A) Menstrual phase showing the endometrium as a thin line; (B) follicular/proliferative phase showing a trilaminar appearance of the endometrium; (C) periovulatory phase showing the echogenicity of the basal layer of endometrium has extended to the midline echo; (D) luteal/secretory phase showing thickened uniform echogenicity. FIGURE 14-4 Normal ovarian cycle. (A) Early phase ovary showing several immature antral follicles of equal size; (B) one follicle has increased flow around it and will start to grow; (C) mid-cycle showing a dominant follicle of 2 cm diameter; (D) post-ovulation ovary with a corpus luteum; (E) spectral trace of ‘active’ ovary showing low-resistance flow with good diastolic flow. FIGURE 14-5 Corpus luteum showing a ‘ring of fire’ of increased colour Doppler flow around it (A) and another with a ring of power Doppler flow (B). There is also cyclical variation in the supplying ovarian and uterine arteries. An increase in volume flow and a reduction in the RI occur in the luteal (secretory) phase compared with the follicular (proliferative) phase. It is the presence of this variation that appears to correlate with fertility rather than any absolute value. Normal ranges of values are given in Table 14-1. The pre-pubertal uterine artery has a high impedance pattern with absent diastolic flow. The change to a lower impedance pattern and the development of diastolic flow indicates the onset of the menarche. The age at which some women seek to start a family has been increasing. Fertility units in particular, need to determine the ovarian reserve to help evaluate if an in vitro fertilisation (IVF) cycle is likely to be successful. Commonly, an antral follicle count of less than four, or an ovarian volume of < 3 cc measured in the first few days of a menstrual cycle, are associated with non-pregnancy or IVF cycle cancellation.9 More recently, it has been suggested that assessing the basal ovarian stromal blood flow may help.10 Being unable to detect any stromal flow in at least one ovary is not just a technical issue but it is more probably related to low ovarian reserve (Fig. 14-6). FIGURE 14-6 (A) Minimal or no stromal flow in a post-menopausal ovary; compared with a pre-menopausal ovary (B). The use of ultrasound contrast agent instilled directly into the uterine cavity to assess patency of the Fallopian tubes has become an accepted precursor to other tests of tubal patency, such as hysterosalpingography, or laparoscopy and dye injection. Intriguingly, loss of sub-endometrial flow on colour Doppler can also indicate tubal blockage,11 although the mechanism for this is not fully understood. Fibroid vascularity is very variable. The attenuation of sound by some fibroids may make any assessment of vascularity impossible. Detection of flow into a fibroid may help confirm its nature; for instance, demonstrating a vascular connection between the uterus and the fibroid may help distinguish a sub-serosal or pedunculated fibroid from an adnexal mass. Likewise, differentiation of an endometrial polyp from a sub-mucosal fibroid (Fig. 14-7) may be helped by finding more than one feeding vessel as polyps should have only a single vessel (Box 14-2). FIGURE 14-7 (A) A submucosal fibroid (arrows) with predominantly reduced echoity and more than one feeding vessel; compare (B) an endometrial polyp (arrows) with a generally increased echoity and only one feeding vessel. Fibroids are often a cause of dysfunctional uterine bleeding. Fibroids are hormonally driven, being more common during menstrual years and tending to regress after the menopause. Visibly vascular fibroids are thought more likely to respond to hormonal manipulation in the suppression of heavy bleeding. Uterine artery embolisation is an alternative to surgery for fibroid-related bleeding, although magnetic resonance imaging is better than ultrasound at assessing suitability and response. A new technique of transvaginal Doppler ultrasound-guided uterine artery occlusion using a temporary clamp has been proposed as a further alternative with some good results in pilot studies.12 Nearly all sarcomatous fibroids are diagnosed incidentally on histology following fibroid removal for other reasons.13 It has long been a goal to detect some vascular feature that would allow diagnosis of a leiomyosarcoma; factors such as the larger a lesion, the more peripheral and central vascularity that is found and lower impedance values have all been put forward as predictive of sarcoma (Fig. 14-8). None has been found useful in practice. Currently, the use of 3D ultrasound and finding increased vascular density have been linked to higher cellular activity scores14 but the imaging diagnosis of sarcoma remains elusive. The most useful sign remains rapid growth on sequential scans.
![]() Doppler Ultrasound of the Female Pelvis
Doppler Ultrasound of the Female Pelvis
Anatomy
Technique
Applications
MENSTRUAL DISORDERS
FERTILITY
Normal Cycle
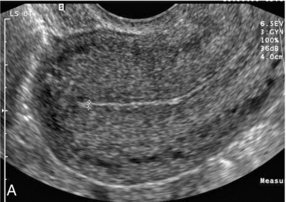
Ovarian Reserve
Tubal Patency and Colour Doppler
DISEASES OF THE UTERINE BODY
Fibroids