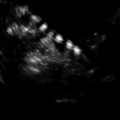
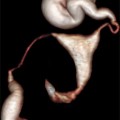
Fig. 11.1
Endometrial polyps. A single polyp located in a lateral wall at midcorpus, shown in two dimensional transvaginal ultrasonographic view (a) and in 3D imaging (b). Multiple polyps and submucosal fibroids (by Pathology) shown by 2D US (c) and by 3D US (d)
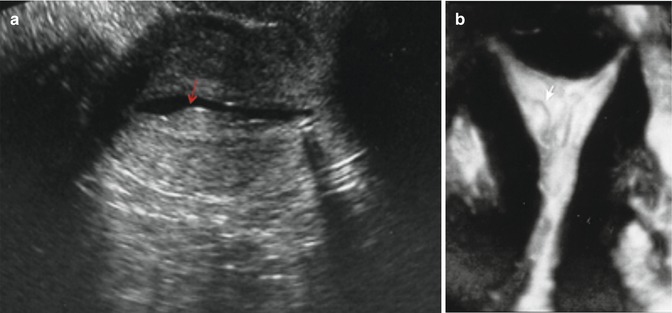
Fig. 11.2
(a) Sessile polyps (arrow) shown in a transverse 2D view of the uterus distended by saline infusion. (b) Coronal view (3D) of the uterus showing a pedunculated polyp (arrow)
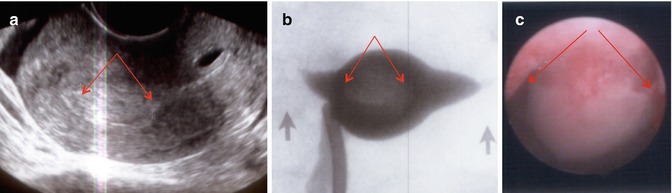
Fig. 11.3
Large polyp (arrows) occupying the entire fundal area shown in a sagittal 2D view (a), in an HSG view (b) giving a globular appearance of the uterus, and in a hysteroscopic view (c)
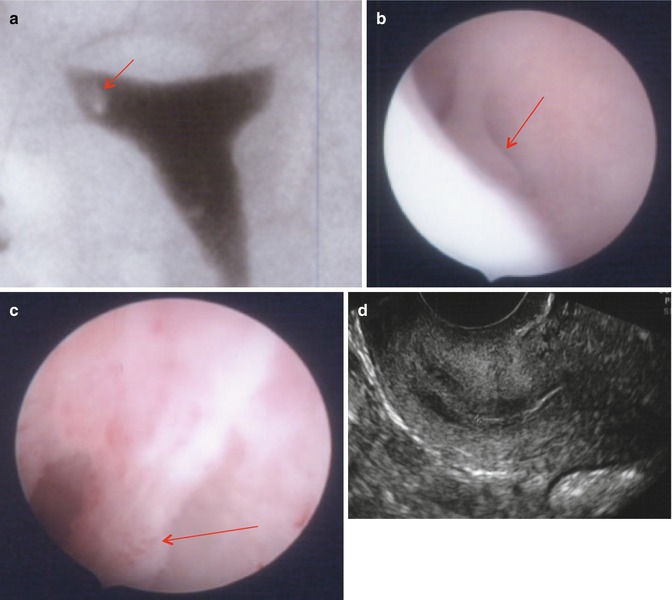
Fig. 11.4
Cornual polyps (arrows) clearly seen in HSG (a) and hysteroscopy (b, c) but not visualized in 2D US (d)
Endometrial polyps are rare among women younger than 20 years of age. The incidence rises steadily with increasing age, peaks in the fifth decade of life, and gradually declines after menopause. The prevalence of polyps can range from 10 to 24 % among women undergoing endometrial biopsy or hysterectomy [1] to 8 to 36 % in postmenopausal women on tamoxifen therapy [2]. Polyps in these women may be large (>2 cm), multiple, or show molecular alterations [2–5]. Also women with Lynch syndrome may have an increased incidence of endometrial polyps compared to the general population [6]. There are several theories on the molecular mechanisms playing a role in the development of endometrial polyps: monoclonal endometrial hyperplasia [7], gene mutations [8], overexpression of endometrial aromatase [9, 10], and, like in leiomyomas, cytogenetic rearrangements and rearrangements in the HMG family of transcription factors [3, 11, 12].
Although endometrial polyps are responsible for approximately one-fourth of cases of abnormal genital bleeding (menorrhagia, postmenopausal bleeding, prolapse through the cervical os, and breakthrough bleeding during hormonal therapy) in both premenopausal and postmenopausal women [1], many polyps are asymptomatic [13].
The natural course of polyps is variable. A prospective study [14] to evaluate this performed two saline infusion sonograms 2.5 years apart on 64 initially asymptomatic women (mean age 44 years). Seven women had polyps on the first examination. Four of these women had spontaneous regression of their polyps at the second scan, while seven women developed new polyps over the 2.5-year interval. Polyps larger than 1 cm were least likely to regress, and hormone use did not appear to affect the natural history of the polyps. In rare cases, endometrial polyps continue to recur multiple times after removal. There are no data regarding management of this clinical situation. If polyps continue to recur, one option is an empiric trial of progestin treatment (oral progestin such as medroxyprogesterone acetate 10 mg daily for 3–6 months, a levonorgestrel-releasing device), and another option is endometrial ablation for women who have completed their childbearing.
The great majority of endometrial polyps are benign, but malignancy occurs in some women. A systematic review of 17 observational studies including over 10,000 women reported that the incidence of malignant or premalignant (simple hyperplasia and atypia, endometrial polyps with complex hyperplasia and atypia) polyps was significantly higher in postmenopausal compared with premenopausal women (5.4 versus 1.7 %; RR 3.86; 95 % CI 2.905.1) and those with bleeding compared to those without bleeding (4.2 % versus 2.2 %, RR 2.0; 95 % CI 1.2–3.1) [15]. Data were inconsistent regarding whether large polyp size was associated with malignancy. Malignant transformation appears to occur more frequently in women on tamoxifen (3–11 %) than in other women [2] regardless of polyp size or duration of tamoxifen therapy. The benefit of using progestagens for treatment of endometrial hyperplasia as well as to reduce the occurrence of de novo endometrial polyps in women treated with tamoxifen has been demonstrated by Chan et al. [16].
For patients with cervical polyps, Stamatellos et al. [17] reported that 25 % of them will also have concomitant endometrial polyps, making hysteroscopy a worthwhile process for their treatment, in contrast to insufficient D&C or blind endometrial biopsies.
Diagnosis
Abnormal uterine bleeding occurs in 9–14 % of women between menarche and menopause, significantly impacting quality of life and imposing financial burden. The American College of Obstetrics and Gynecology (ACOG) [18] recommends endometrial tissue assessment to rule out cancer in adolescents and in women younger than 35 years or older with suspected anovulatory bleeding and women unresponsive to medical therapy. Neither ultrasonography nor hysteroscopy can reliably distinguish between benign and malignant polyps [19, 20].
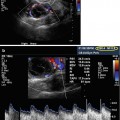
Transvaginal ultrasound (TVUS) is the initial imaging of choice, with MRI reserved for indeterminate cases or where sampling is difficult [21]. On US, endometrial polyps appear as ovoid echogenic masses that project into the endometrial lumen with Doppler US showing a feeding vessel (Figs. 11.1 and 11.5). The following guidelines have been proposed in 2012 by the American Association of Gynecologic Laparoscopists (AAGL) [22] for the diagnosis of endometrial polyps: (1) TVUS provides reliable information for the detection of endometrial polyps and should be the investigation of choice where available; (2) the addition of color or power Doppler increases the capacity of TVUS to diagnose endometrial polyps; (3) adding intrauterine contrast sonography (with or without 3D imaging) improves the diagnostic capacity for endometrial polyps; (4) blind dilatation and curettage or biopsy should not be used for diagnosis of endometrial polyps.
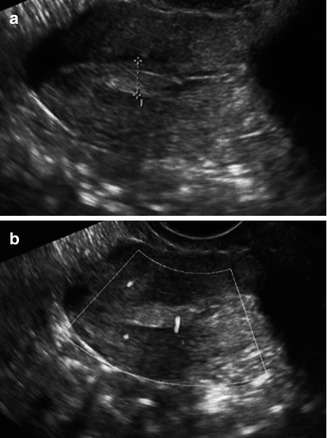

Fig. 11.5
Transvaginal ultrasonographic view of an endometrial polyp (a, cursor) appearing as an echogenic ovoid structure containing a feeding vessel visualized by Doppler (b)
Transvaginal Ultrasonography
Salim et al. [23] performed a review of the literature on the diagnosis and management of endometrial polyps. On TVUS, an endometrial polyp typically appears as a hyperechoic lesion with regular contours within the uterine lumen surrounded by a thin hyperechoic halo [24], occasionally cystic spaces corresponding to dilated glands filled with proteinaceous fluid may be seen within the polyp [25], or the polyp may appear as a nonspecific endometrial thickening or focal mass within the endometrial cavity [26]. None of these findings can reliably distinguish among polyps, submucous fibroids, adenomyosis, and neoplastic change.
In premenopausal women, the TVUS examination should be performed early in the proliferative phase when the endometrium is at its thinnest (4–8 mm) [27, 28] to minimize false-positive and negative findings [29]. Endometrial thickness is associated with risk of endometrial cancer in menopausal women but not in premenopausal women [30, 31]. Endometrial thickening (Fig. 11.6) is a nonspecific finding of endometrial hyperplasia as well as other causes such as polyp, endometrial cancer, trophoblastic disease (Fig. 11.6), retained products of conception (Fig. 11.6), or submucosal leiomyoma (Fig. 11.1) [21]. If structural abnormalities are suspected on ultrasound examination, then these abnormalities can be further evaluated by saline infusion sonography or hysteroscopy. In a retrospective review of multiple studies, Salim and his group [23] report that for TVUS the sensitivity varies between 19 and 96 %, specificity of 53 and 100 %, positive predictive value (PPV) of 75 and 100 %, and negative predictive value (NPV) of 87 and 97 %, when compared with hysteroscopy with guided biopsy. The ranges were tighter in a single-large prospective study evaluating the causes of menorrhagia: 86 % sensitivity, 94 % specificity, 91 % PPV, and 90 % NPV [32].
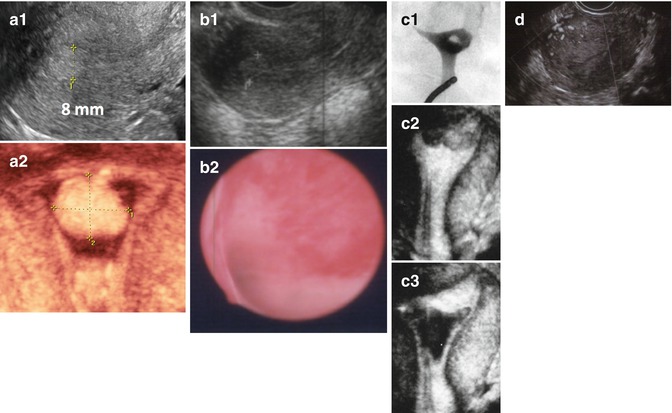

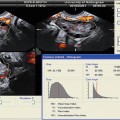
Fig. 11.6
Examples of different endometrial pathologies presenting as endometrial thickening in TVUS. (a) Endometrial polyp (a1 2D US, a2 3D SIS); (b) complex hyperplasia without atypia (b1 2D US, b2 flat lesions in hysteroscopy); (c) retained products of conception (c1 HSG, c2 3D US, c3 3D SIS); (d) trophoblastic disease (multivessel signal)
There are limited data to support color-flow or power Doppler aiding in the differentiation of hyperplasia and malignancy in polyps [33–35]. Color-flow Doppler scanning identifies a single feeding vessel (Fig. 11.5) [11]. The use of power Doppler sonography with identification of a single-vessel pattern improves diagnostic sensitivity to 89 % and specificity to 87 % for an endometrial polyp [36], in comparison to the multivessel (Fig. 11.6) or scattered pattern seen in malignant lesions or endometrial hyperplasia [34]. At this time, sonographic examination either with or without color-flow or power Doppler sonography is not a substitute for pathologic evaluation after surgical removal.
Sonohysterography
The use of saline infusion sonography (SIS), sonohysterography, or hydrosonogram involves injection of sterile saline into the endometrial cavity followed by a transvaginal ultrasound examination (Figs. 11.2 and 11.7). This technique increases sonographic contrast of the endometrial cavity, enabling delineation of the size, number, and location of polyps that could have been missed on gray-scale TVUS, and is likely to improve diagnostic accuracy [37, 38]. With SIS, polyps appear as echogenic, smooth, intracavitary masses with either broad bases or thin stalks outlined by fluid [26]. Differentiating endometrial polyps from submucosal fibroids can be difficult (Fig. 11.1), but examination of lesion echotexture and identification of overlying echogenic endometrium are useful features to distinguish the two [39]. SIS has relatively minor disadvantages: it cannot provide definitive diagnosis of endometrial disease [40], a longer learning curve compared with non-contrast TVUS [41], and patient discomfort caused by fluid leakage or pain with the use of a balloon catheter [42]. Bingol et al. [43] selected a total of 346 patients for operative hysteroscopy, following SIS after TVUS. SIS seems to be superior to TVS, for uterine pathologies, with respect to hysteroscopy as the gold standard.
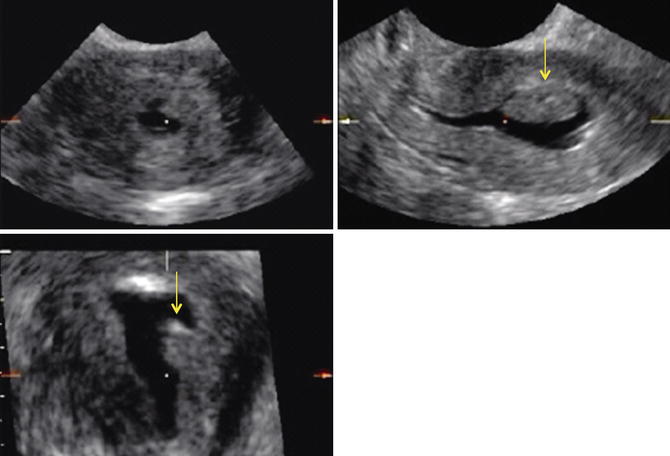

Fig. 11.7
3D-rendered view of the uterus during sonohysterography. The arrow points to an endometrial polyp in the left midcorpus
Kamel et al. [44] reported on 106 women with menometrorrhagia where sonohysterography was significantly more accurate than ultrasound alone in making a diagnosis, with a higher sensitivity (93 % versus 65 %) and specificity (94 % versus 76 %) than transvaginal ultrasonography.
Three-Dimensional TVUS and Three-Dimensional SIS
Three-dimensional ultrasonography (3D US) is a noninvasive imaging technique with the ability to generate multiplanar reconstructed images (Fig. 11.8) through the uterus and its external contours. Coronal views of the uterus allow more accurate visualization between the endometrium and myometrium at the fundus and cornual angles, providing superior diagnostic accuracy in detecting endometrial polyps compared to 2D TVUS (Figs. 11.1, 11.2, and 11.7).
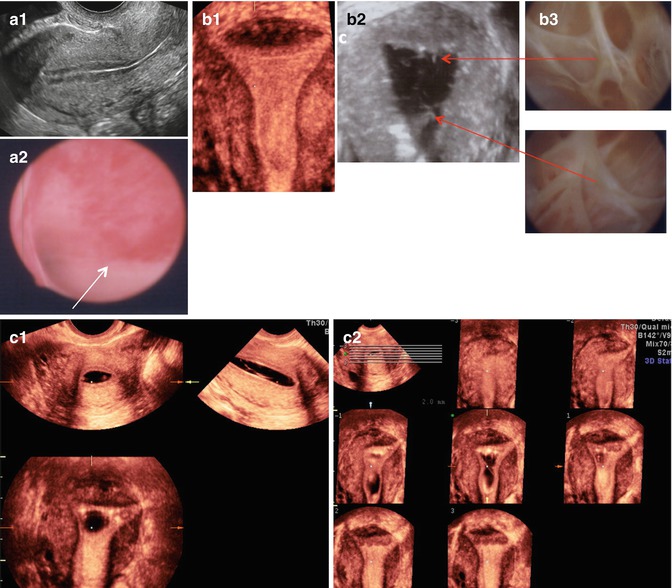

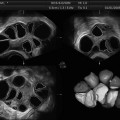
Fig. 11.8
Intrauterine lesions that may not be easily detected by TVUS. (a1) Apparently normal 2D sagittal view of the uterus. (a2) Same uterus as in (a1), containing flat hyperemic lesions visualized directly by hysteroscopy (benign polyp on Pathology). (b1) Apparently normal 3D coronal view of the uterus. (b2) Thin, though multiple, bands of synechiae seen on 3D SIS and hysteroscopy (b3). (c1) Synechiae not clearly visualized on 3D-SIS but more clearly identified upon evaluation of the multiplanar views (c2) of the uterus
In a study of 3,850 consecutive, Kupesic et al. [45] reported 3D US to have sensitivity of 100 %, specificity of 99 %, PPV of 99 %, and NPV of 100 % in diagnosing endometrial polyps when compared to hysteroscopy with biopsy. Adding saline solution contrast into the endometrial cavity to perform 3D SIS may provide additional information in the diagnosis of endometrial polyps (Figs. 11.6 and 11.7). However, studies report only slightly higher specificity (88–99 %) and PPV (97–100 %) for endometrial polyps than those of 3D US, with sensitivity of 92–95 % and NPV of 97 % [46, 47].
Three-dimensional sonography is simple, quick, and noninvasive for detecting most congenital and acquired uterine anomalies. We demonstrated that physicians who learn the Z technique [48] are able to retrieve the mid-coronal plane of the uterus faster and improve its image quality in volume sonography. In a prospective blinded study to evaluate the costs, accuracy, risks, and benefits of 3D TV sonography compared to hysterosalpingography [49], we enrolled 101 women aged 26–44 years with evidence of uterine anomalies (congenital and acquired). We concluded that 3D TV sonography provides visualization and evaluation of the uterine cavity with similar or better accuracy than standard hysterosalpingography (HSG) in the office setting, without radiation exposure, with lower cost and morbidity. Despite the multiple advantages of performing 3D US, including having diagnostic accuracy comparable to MRI or combined laparoscopy and hysteroscopy, it is still not widely available and accepted as a diagnostic tool, and multiple insurance carriers deny its reimbursement.
Other Imaging Modalities
Hysterosalpingography may define an endometrial polyp as pedunculated (Fig. 11.4), or as a nonspecific filing defects within the endometrial cavity (Figs. 11.3 and 11.6), with high sensitivity (98 %) but low specificity (34.6 %) compared with hysteroscopy [50]. It has the advantage of allowing for assessment of tubal patency in infertile women, but due to the disadvantages of using ionizing radiation, iodinated contrast materials, and patient discomfort, routine use of HSG for diagnosis of an endometrial polyp cannot be recommended.
T2-weighted MRI and computed tomography scanning are very high-cost, limited availability techniques with limited advantages when compared with TVUS, even with contrast enhancement [51]. Appearance on MR is not diagnostic, and endometrial polyp, secretory endometrium, stage IA endometrial cancer, and endometrial hyperplasia can demonstrate similar findings.
Hysteroscopy is an outpatient surgical procedure usually requiring local or intravenous anesthesia that provides direct visualization of the endometrial cavity (Figs. 11.3, 11.4, 11.6, 11.8, and 11.9), thereby allowing targeted biopsy or excision of lesions identified during the procedure [52]. Although considered the gold standard for the diagnosis of abnormal uterine bleeding, hysteroscopy requires advanced training and is more costly and invasive than imaging modalities for endometrial assessment [27]. Grimbizis et al. [53] studied a total of 105 consecutive women presenting in an outpatient clinic with symptoms of menorrhagia, postmenopausal bleeding, and infertility. They found diagnostic hysteroscopy to be the most accurate diagnostic technique on any endometrial pathology compared with TVUS and SIS, whereas there was no statistically significant difference in the diagnostic performances of SIS and TVUS.
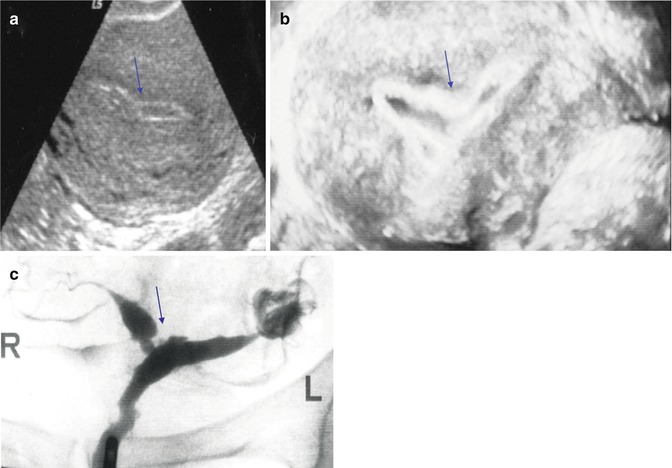

Fig. 11.9
Nonspecific endometrial US findings. An area of endometrial constrictions is shown by 2D US (a), by 3D SIS (b), and by HSG (c). Arrows point to the area of narrowing representing synechiae
False-Positive, False-Negative, and Artifacts
There is not a single imaging technique that can accurately diagnose all possible intrauterine pathologies. Ultrasonography may not distinguish very small polyps, flat endometrial anomalies (Figs. 11.6 and 11.8), cornual polyps (Fig. 11.4), or thin bands of synechiae (Figs. 11.8 and 11.9) even when combined with saline infusion sonography [54].
To the contrary, there could be transient endometrial changes detected by ultrasonography as a possible structural defect, such as an intrauterine blood clot (Fig. 11.6) or presence of mucus especially in hyperestrogenic states such as during controlled ovarian hyperstimulation for IVF (Fig. 11.10) that may spontaneously resolve. Our group [55] reported on early postoperative intrauterine changes by 3D US in patients undergoing hysteroscopic correction of various uterine anomalies. Postoperative changes 1 month after hysteroscopy were detected by 3D US in 20 % (6/30) of these patients. The changes detected consisted of intrauterine cystic loculations (Fig. 11.10) in three patients and endometrial irregularities/possible mucus accumulations in the other 3. These loculations and irregularities were sometimes larger and even more complex in nature than the original lesions and always coincided with the resection site. These changes resolved spontaneously after the second postoperative month and did not interfere with embryo implantation or cause pregnancy loss in patients attempting to conceive during the study period.
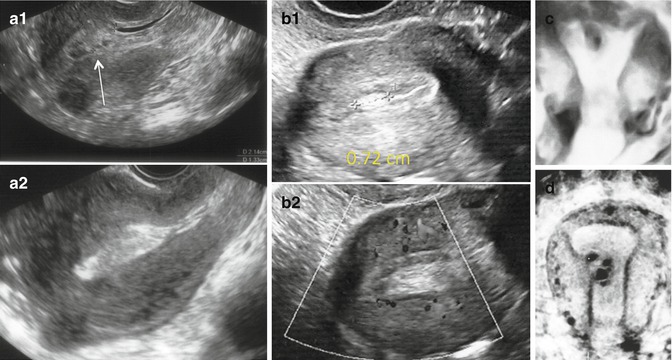

Fig. 11.10
Transient endometrial changes detected as morphological anomalies by US. Endometrial clots and fluid seen on menstrual day 4 (a1) that spontaneously disappeared 1 day later (a2). (b1) Endometrial mucus seen during ovarian stimulation for IVF. Notice lack of internal blood flow in the hyperechoic mucus accumulation (b2). Monoloculated (c) and multiloculated (d) cystic lesions observed by 3D US a few weeks after hysteroscopy that spontaneously resolved within 2 months post op
Grimbizis et al. [53] reported that diagnostic hysteroscopy, on the other hand, can misdiagnose normal endometrium for small endometrial polyps or, to the contrary, can misdiagnosed a case of endometrial cancer as an endometrial polyp. As it is well known, TVS, SIS, or diagnostic hysteroscopy cannot replace biopsy in cases where endometrial cancer is suspected. Also, some artifacts can be unintentionally created during SIS that could mimic intrauterine pathology such as catheter tunneling of the endometrium which could be mistaken for a polyp (Fig. 11.11) or injection of air bubbles which could be mistaken for a trophoblastic disease (Fig. 11.11).
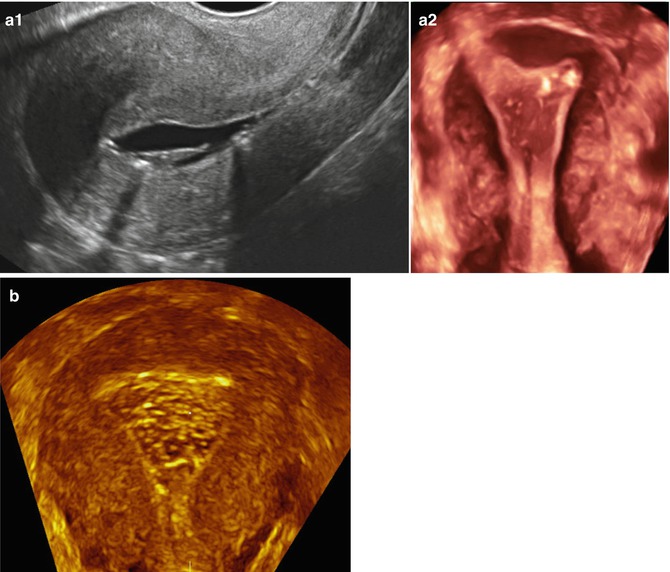

Fig. 11.11
Artifacts created during SIS giving the false impression of intracavitary pathology. Endometrial tunneling created by the catheter used resembling a polyp in a 2D sagittal view (a1) and in a 3D coronal view (a2). Air bubbles injected during SIS may resemble trophoblastic disease (b, 3D coronal view)
Impact of Polyps on Fertility
It is controversial whether endometrial polyps contribute to infertility or miscarriages [56], and because there is no uterine abnormality that is always associated with poor reproductive performance, automatic surgical correction is not indicated when a uterine abnormality is found [57]. However, surgical correction should be considered when a submucous fibroid, endometrial polyp, septate uterus, or uterine synechiae are discovered in the setting of failure to conceive or recurrent pregnancy loss, since there may be a causal association.
Normal endometrial thickness, structure, and texture are crucial for uterine receptivity, and any structural pathology in the uterine cavity may lead to subfertility or implantation failure [58]. Doldi et al. [59] reported endometrial polyps to be the most common of the intrauterine lesions interfering with the endometrial cavity. The exact mechanism by which endometrial polyps cause infertility is not known, but some proposed mechanisms are induction of an inflammatory process similar to an intrauterine device [60], mechanical interference with sperm and embryo transport, impairment of embryo implantation, or altered endometrial receptivity such as abnormalities in the expression of molecular markers such as HOXA 10 and HOXA 11 [61]. Regardless of the mechanism of endometrial disturbance, uterine polyps have been associated with decreased pregnancy rates both in natural conceptions [62–64] and in intrauterine insemination (IUI) cycles [65].
There are no data from randomized trials to guide therapy of asymptomatic polyps, unless there are risks for hyperplasia or carcinoma. For symptomatic patients, polypectomy results in the improvement of symptoms in 75 % of patients in studies with follow-up intervals of 2–52 months [66]. Lieng et al. [67] reported on 150 women in a randomized clinical trial with an endometrial polyp allocated to hysteroscopic removal or observation. Although there was no improvement in the volume of menstrual loss, a significant improvement in other symptoms such as intermenstrual bleeding was significantly improved by removal at follow-up. Hysteroscopically guided polypectomy (with grasping forceps, suction curette, microscissors, a small electrosurgical look, i.e., resectoscope, mechanical morcellation, or a bipolar electric probe [68–70]) is the most effective method of removal since small polyps and other structural abnormalities can be missed by blind curettage [68, 71, 72].
In a retrospective study from 2000 to 2005, Stamatellos et al. [73] reported on 83 subjects with endometrial polyps and no other cause for their infertility subjected to hysteroscopic polypectomy. The mean size of the endometrial polyps was 1.9 +/− 1.4 cm. Following polypectomy, menstrual pattern was normalized in 91.6 % of patients. Spontaneous pregnancy and delivery at term rates, in the total population of the study, increased after the procedure and were 61.4 and 54.2 % respectively. There was no statistical difference in fertility rates between patients having polyps ≤1 cm and patients having >1 cm polyps or multiple polyps. Spontaneous abortion rate in the first trimester of pregnancy was 6 %, and there was no statistical difference between patients with small or bigger/multiple polyps. They concluded that hysteroscopic polypectomy appeared to improve fertility and increase pregnancy rates in previous infertile women with no other reason to explain their infertility, irrespective of the size or number of the polyps.
Polyps and Assisted Reproductive Technology
Most of the data for polypectomy in subfertile patients suggests that removal improves fertility, with reported pregnancy rates varying between 43 and 90 % [23, 62, 68, 74]. Perez-Medina et al. [65




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree