Endovascular stroke therapy has revolutionized the management of patients with acute ischemic stroke in the last decade and has facilitated the development of sophisticated stroke imaging techniques and a multitude of thrombectomy devices. This article reviews the scientific basis and current evidence available to support endovascular revascularization and provides brief technical details of the various methods of endovascular thrombectomy with case examples.
Key points
- •
Less than 1% of patients with acute ischemic stroke in the United States receive intravenous (IV) tissue plasminogen activator (t-PA); recanalization rates associated with IV t-PA for proximal, large vessel arterial occlusions remain poor.
- •
At present, patients who do not meet the eligibility criteria for thrombolytic therapy, who fail to improve neurologically after thrombolytic therapy, or who improve and then worsen (patients with reocclusion) are candidates for endovascular revascularization therapies.
- •
Advanced brain perfusion imaging is increasing the ability to better assess the therapeutic target (the ischemic penumbra) and better select patients who may benefit from revascularization.
- •
Higher rates of recanalization have been shown with endovascular methods, particularly mechanical therapies, and this has been correlated with better outcomes.
- •
Stent-assisted and stent platform–based thrombectomy devices have increased the ability to achieve high recanalization rates in large vessel occlusions.
- •
The rapidity of advances in endovascular stroke therapy has made assessment through large-scale randomized controlled trials difficult and, for many interventionists who see surprising recoveries following endovascular therapy, unethical because of a lack of clinical equipoise.
Introduction
Endovascular stroke therapy has revolutionized the management of patients with acute ischemic stroke in the last decade and has facilitated the development of sophisticated stroke imaging techniques and a multitude of thrombectomy devices. Moreover, it has changed the way in which stroke therapists have used intravenous (IV) tissue plasminogen activator (t-PA), which is the only medical therapy approved by the US Food and Drug Administration (FDA) for acute stroke treatment at present.
Introduction
Endovascular stroke therapy has revolutionized the management of patients with acute ischemic stroke in the last decade and has facilitated the development of sophisticated stroke imaging techniques and a multitude of thrombectomy devices. Moreover, it has changed the way in which stroke therapists have used intravenous (IV) tissue plasminogen activator (t-PA), which is the only medical therapy approved by the US Food and Drug Administration (FDA) for acute stroke treatment at present.
IV thrombolysis is not a panacea for all patients
Less than 1% of acute patients with ischemic stroke in the United States receive t-PA, primarily because of a delay in presentation for treatment, although this number may increase with the extended time window of 4.5 hours. There is also reluctance in emergency physician participation because of perceived risks, particularly intracranial hemorrhage (ICH), and lack of supporting critical care and specialty neurologic services. Even when delivered early, reocclusion after thrombolysis has been shown by transcranial Doppler imaging to occur in 34% of patients receiving IV t-PA and may result in neurologic worsening in many of these patients.
Patients who fail or are not candidates for IV thrombolysis
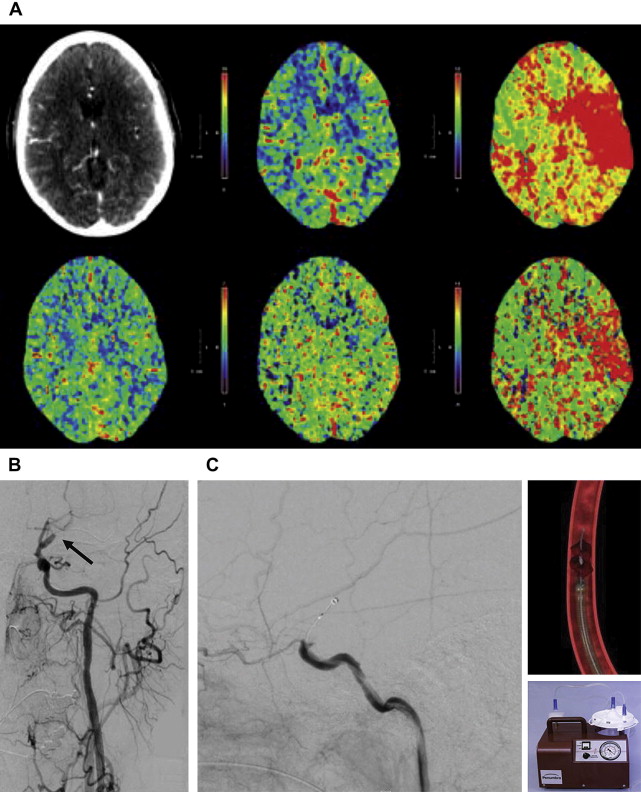
Patients who do not meet the eligibility criteria for thrombolytic therapy, fail to improve neurologically after thrombolytic therapy, or who improve and then worsen (patients with reocclusion) are currently considered candidates for endovascular revascularization therapies. At our center, 94 patients with a mean National Institutes of Health Stroke Scale (NIHSS) score of 14.7 at presentation were treated by endovascular interventions within 3 hours of stroke symptom onset. In these patients, t-PA IV thrombolysis (IVT) was contraindicated or had failed. Partial to complete recanalization (Thrombolysis in Myocardial Infarction [TIMI] score of 2 or 3) was achieved in 62 of 89 (70%) patients presenting with significant occlusion (TIMI 0 or 1). Postprocedure symptomatic intracranial hemorrhage (SICH) occurred in 5 patients (5.3%), which was subarachnoid hemorrhage in 3 of these patients. The total mortality, including procedural mortality, progression of disease, or other comorbidities, was 26.6%. Overall, 36.7% of patients had a modified Rankin scale (mRS) score of less than or equal to 2 at discharge. Mean NIHSS at discharge was 6.5, representing an overall 8-point improvement in NIHSS score.
Atrial fibrillation is a surrogate parameter of cardioembolic stroke with well-known higher bleeding rates in patients who receive IVT. The Interventional Management of Stroke (IMS) I study showed a significant increased risk of SICH in patients with atrial fibrillation. At our center, patients with strokes with atrial fibrillation had high rates (14.3%) of SICH after endovascular therapy that could not be explained by their anticoagulation status. It is important to identify these patients as a separate entity to weigh the risks and benefits associated with treatment.
The concept of clot burden and location
Clot extent and location are important determinants of outcomes in acute stroke and determine the ability to achieve recanalization. The recanalization rates associated with IV t-PA for proximal, large vessel arterial occlusions are poor and range from only 10% for internal carotid artery (ICA) occlusion to 30% for middle cerebral artery (MCA) occlusion. IVT is not as effective in thromboembolic obstruction of these large, proximal vessels as in more distal smaller vessels. The outcome of IVT after large intracranial vessel thromboembolic occlusion currently remains dismal and is associated with high morbidity and mortality. Clinical response to thrombolysis was influenced by the site of occlusion in a multicenter cohort of patients with strokes. Odds ratios (ORs) for complete recanalization (TIMI 3) of vessel occlusions were as follows: distal MCA, 2; proximal MCA, 0.7; ICA terminus, 0.1; tandem cervical ICA/MCA, 0.7; and basilar artery, 0.96. A long, proximal clot is harder to treat, leading to a worse outcome compared with a shorter distal clot.
NIHSS is not a reliable assessment of clot location or burden
Previously, an NIHSS score of greater than 12 was thought to suggest an occlusion of a large, proximal vessel and, therefore, a high thrombus burden. In a prospective cohort study of 699 patients with 377 (54%) large vessel occlusions and 171 (24%) MCA M1 segment occlusions, the median NIHSS score for patients found to have a large vessel occlusion was higher than the overall median score for the cohort (9 vs 5, P <.0001). The median NIHSS score of patients with MCA M1 segment occlusion was 14. An NIHSS score of greater than or equal to 10 had an 81% positive predictive value for large vessel occlusion but only 48% sensitivity, with most patients with proximal occlusions presenting with lower NIHSS scores. These data suggest that all patients with NIHSS scores of greater than or equal to 2 need to have imaging to detect 90% of proximal occlusions. This finding suggests a need to revisit the current practice of administering IV t-PA to patients because of NIHSS scores alone in the absence of any vascular or perfusion imaging studies. Further, many distally located clots (M2, M3, P2, P3) may cause the patient to present with a high NIHSS score; this confounding finding may be explained by intrinsic thrombolysis that results in a large proximal occlusion (the original cause of the high NIHSS score) resolving into smaller distal residual emboli.
Endovascular therapy achieves higher recanalization rates and improved outcomes
In the Mechanical Embolus Removal in Cerebral Ischemia (MERCI) and Multi-MERCI studies and the combined analysis of IMS-I and IMS-II data, functional outcome (measured by an mRS score of ≤2 at 3 months) was significantly better and 3-month mortality was significantly lower in patients who had TIMI 2 or 3 recanalization than in patients in whom vessels failed to recanalize after endovascular therapy. Rha and Saver reviewed 53 studies including 2066 patients and found that good functional outcomes (mRS score ≤2 at 3 months) were more frequent in patients with vessel recanalization than without vessel recanalization. The 3-month mortality was reduced in patients in whom vessels were recanalized. Higher rates of recanalization were achieved with endovascular methods, particularly mechanical therapies, and consequently were associated with better outcomes.
Time window between symptom onset and treatment affects outcomes
The European Cooperative Acute Stroke Study (ECASS) III trial showed the value of extending the time window for IV t-PA to 4.5 hours. A meta-analysis by Lansberg and colleagues included data from patients in the ECASS I, II, III, and Alteplase Thrombolysis for Acute Noninterventional Therapy in Ischemic Stroke (ATLANTIS) trials treated within the 3-hour to 4.5-hour time window, and showed at 3 months that t-PA treatment was associated with an increased probability of favorable outcome and mortality was not significantly different for IVT-treated and placebo-treated patients. A meta-analysis conducted by Wardlaw and colleagues showed higher benefit (compared with risk of being dead or disabled) up to 6 hours after IVT, thus formally providing level 1 evidence, even in patients selected solely from noncontrast computed tomography (CT) imaging.
Endovascular therapy can be used beyond the IVT time window
The Prolyse in Acute Cerebral Thromboembolism (PROACT) trials established a benefit of intra-arterial thrombolysis (IAT) up to 6 hours after stroke symptom onset with an increase in recanalization rates. Mechanical revascularization strategies reestablish flow faster than thrombolytics, and thus may increase the benefit of treatment, even when there is a delay in presentation for treatment. The MERCI, Multi-MERCI, and Penumbra trials show effectiveness of mechanical revascularization therapy up to 8 hours after stroke symptom onset. There is increasing evidence that identification of potentially salvageable brain tissue with advanced magnetic resonance (MR)–based and CT-based perfusion imaging may allow the selection of patients who can be effectively and safely treated more than 8 hours after ictus. In a consecutive series of patients selected for endovascular revascularization from advanced CT perfusion imaging at our center, no difference was found in outcomes in those with known time of symptom onset of less than 8 hours and those with wake-up strokes (with unknown time of onset but suspected to be >8 hours).
The change from time is brain to ischemic penumbra being the therapeutic target
The concept of ischemic penumbra divides tissues in the ischemic territory supplied by the occluded vessel to be comprised of (1) ischemic core, which is tissue that is dead and cannot be salvaged; (2) ischemic penumbra, which is tissue that will rapidly convert to core if the occluded vessel is not recanalized; and (3) area of oligemia, which is the area of the brain that is underperfused but will likely survive even if the vessel is not recanalized (dependent on collateral supply). Compartmentalization of ischemic tissue is dynamic and is changing every minute from symptom onset until tissue perfusion is restored or ischemic penumbra and core are matched. The concepts of identifying the therapeutic target (the ischemic penumbra) and having an estimation of the size and location of the core are driving stroke neuroimaging research and development at a fast pace. The theoretic possibility of being able to manipulate these dynamic variables in the critical time period from patient identification to tissue reperfusion has provided a second overlay over the time of stroke onset to decide on reperfusion strategies and search for adjunctive measures to improve outcomes after stroke revascularization.
Importance of collaterals in stroke outcomes
Collateral supply is one of the main determinants of the dynamic balance between the relative amounts of tissue classified as ischemic penumbra and oligemia. The ability to quantify the presence and extent of collateral support to ischemic tissue and augment collateral support before recanalization may widen the time window for recanalization therapy and improve ability to prognosticate stroke outcomes. It may also further the understanding of the physiology of ischemic tissue and aid selection of patients for aggressive recanalization strategies. The extent of revascularization depends not only on recanalization of the primary arterial occlusive lesion but also on reperfusion of the distal vascular bed. The degree of collateral supply through peripheral leptomeningeal sources is important and correlates with the presence of a smaller final infarct volume. The evaluation of collateral supply remains challenging because of the diminutive size and complex routes of these vessels. Collateral failure may be an important cause of delayed deterioration after successful reperfusion, and thus collateral support may need to be monitored after reperfusion to improve outcomes. Several interventions that might augment collateral blood flow are being investigated.
Importance of assessing ischemic core before revascularization
The percentage and location of the ischemic core are important determinants of the risk of SICH after reperfusion. ICH occurs in the core of the infarction in most patients. SICH usually occurs within 24 to 36 hours of reperfusion therapy. The mortality after SICH in the National Institute of Neurologic Disorders and Stroke (NINDS) trial was 47%. When dichotomized Alberta Stroke Program Early CT Scores (ASPECTS) were assessed, patients with low stroke burden (ASPECTS >7) on CT imaging had a lower occurrence of SICH after IVT and a higher chance of gaining independence (mRS score ≥2). The use of CT perfusion (CTP) imaging, rather than noncontrast CT imaging, increases prognostic accuracy of the ASPECTS, with the final infarct mirroring cerebral blood volume (CBV) or cerebral blood flow (CBF) deficits when reperfusion is or is not achieved, respectively. The Diffusion-weighted Imaging Evaluation for Understanding Stroke Evolution (DEFUSE) trial was the first study that directly established a strong relationship between MR imaging (based on ischemic core) and tissue parameters and SICH. In this study, early reperfusion was associated with fatal ICH in patients with the malignant profile (baseline diffusion-weighted imaging volume >100 mL and/or perfusion-weighted imaging deficit >100 mL with 8 seconds or longer of T max [time to peak of the deconvoluted curve]). The presence of even small ischemic cores in the basal ganglia region predicts higher rates of SICH and disability after revascularization.
Perfusion imaging for patient selection
Perfusion imaging for patient selection for both IVT and endovascular therapy is being investigated for its potential to identify the ischemic territory (perfusion deficit) and the location and percentage of ischemic core (perfusion mismatch). CT and MR are two perfusion imaging modalities that are being evaluated. The main limitation of both modalities is that the perfusion deficit is a sum of the therapeutic target: the real ischemic penumbra and the oligemic tissue. Any perfusion mismatch calculation is erroneous, because currently there is no definite way to differentiate between these territories. As emphasized earlier, the relative percentage of core, ischemic penumbra, and oligemic tissue is a dynamic phenomenon and has to be assessed as close to the time of the recanalization procedure as possible to achieve meaningful outcomes based on perfusion imaging findings. The assessment of collaterals through perfusion imaging and catheter-based angiography is being investigated at multiple centers.
Perfusion parameters stratify the patient’s risk for hemorrhage after endovascular intervention and the potential benefit of mechanical flow restoration. In general terms, when decreased CBF and CBV (findings that suggest completed infarct) represent less than one-third of the territory exhibiting increased time to peak (TTP) (ie, putative penumbra or perfusion deficit), we have found that the patient benefits from endovascular intervention and has a lower risk of SICH. Early ischemic changes on a noncontrast CT scan that correspond with CBF and CBV deficits solidify the reliability of predicting completed infarct. Reperfusion of a larger core is ineffective and likely increases the risk of hemorrhage. In contrast, patients with hyperacute presentations (ie, <2 hours) had salvageable regions after endovascular therapy, even in the face of a poor CTP profile (ie, with decreased CBV and CBF). In addition, occlusion of proximal M1 perforators and attendant basal ganglionic involvement in the infarct core presage a higher risk of hemorrhage and poor clinical outcome after recanalization. Recent studies suggest that clinical improvement is noted even when endovascular stroke therapy is performed more than 8 hours after ictus in patients with large penumbras identified on CTP imaging.
There has been recent interest in estimating CTP thresholds (thresholds of CBF and CBV) and using quantitative and automated CTP maps to estimate core and penumbra to aid in choosing patients for reperfusion beyond traditional therapeutic windows or when the time of stroke onset is unknown. Differences in CTP hardware and software can affect quantified metrics, and clearly defined thresholds for guiding therapy have yet to be standardized. Some studies suggest the use of CBF thresholds for defining areas of infarct, specifically CBF less than 25 mL/100 g/min. In an analysis of 130 patients with acute stroke, Wintermark and colleagues suggested using absolute CBV less than 2 mL/100 g to define core infarct and increase in relative mean transit time by 145% of normal to define penumbra. Murphy and colleagues studied 30 patients and showed CBF × CBV as the best predictor for differentiating core infarct and penumbra (better than CBF or CBV thresholds alone). CTP thresholds are also specific to the perfusion software platform being used and may not be automatically transferable to other vendors, scanners, and even software versions. Attention should be paid to technical aspects of data acquisition and postprocessing, including placement of regions of interest as well as selecting an appropriate volume of imaging commensurate with the clinical condition or symptoms. Disorders such as chronic infarct, severe microvascular ischemia, and seizure can be mistaken for acute infarct. Vascular stenoses can mimic and overestimate areas of ischemic penumbra; therefore, CTP should always be performed and interpreted in conjunction with CT angiography. At this time, much work remains to standardize quantitative methods of CTP interpretation, which, in the future, may be addressed by a proposed consortium for acute stroke imaging.
Patient selection for endovascular therapy
Because patients considered for revascularization (whether IVT or endovascular therapy) require imaging assessment of the presence of decreased tissue perfusion, location and extent of clot burden, and core location and percentage, every patient with a clinical diagnosis of ischemic stroke at our center is evaluated with stroke CT imaging. The imaging protocol includes (1) noncontrast cranial CT scan to exclude hemorrhagic conversion or other structural abnormality; (2) CTP imaging with special attention to TTP, CBF, and CBV sequences; and (3) CT angiogram from the aortic arch through the intracranial vessels. Perfusion imaging is performed on a 320-slice Aquilon scanner (Toshiba Medical Systems, Tustin, CA).
CT angiography, although not essential when proceeding directly to the performance of a catheter angiogram for diagnostic purposes, provides critical information for endovascular planning. First and foremost, the target vessel occlusion responsible for the patient’s symptoms is confirmed, after clinical evaluation, with the corresponding CTP data. In addition, CT angiography allows the length of the occlusion to be determined, the presence of tandem occlusions to be identified, and access planned from the aortic arch to the lesion.
In the setting of large vessel occlusion (ICA to proximal M2) on CT angiography, the patient is always assessed for consideration of endovascular revascularization either alone or with IVT bridging therapy. We have found that patients with anterior circulation strokes with large vessel occlusions presenting more than 8 hours after symptom onset have done poorly, despite selection based on imaging findings. Patients with wake-up strokes with unknown time of onset and posterior circulation strokes 24 hours, or sometimes even up to 48 hours, after stroke symptom onset are evaluated for endovascular revascularization. For patients more than 75 years of age, a more thorough risk-benefit analysis is warranted. In our series of such patients who were selected from imaging, 72% died or were disabled after endovascular treatment. As mentioned earlier, patients with atrial fibrillation have a higher risk of SICH during stroke interventions, and careful selection of patients and revascularization tools is warranted in such cases.
IAT
In theory, IAT may offer a higher dose of thrombolytic drug delivery to the clot with fewer systemic complications and higher recanalization rates. Intra-arterial (IA) treatment may also facilitate extension of the therapeutic window and provide an option for patients with contraindications for systemic thrombolysis (ie, postoperative stroke) or patients in whom IVT has failed. The advantages of IAT include the ability to perform angiography with gold-standard characterization of the obstructive lesion; imaging of collateral flow anatomy (to the extent possible with current technology); confirmation, exact degree, and timing of recanalization; and capability for combination with mechanical thrombectomy methods. Disadvantages of IAT include delay in treatment, risks of catheter manipulation, increased risk of SICH with pharmacologic thrombolysis, and need for skilled endovascular facilities and personnel.
To perform IAT, a long 6-French (F) or 7-F sheath is placed into the femoral artery, and a 6-F or 7-F guiding catheter is advanced into the ICA or vertebral artery of the affected side. A microcatheter is then navigated to the occlusion site over a microwire, and the thrombolytic agent is injected proximal to or directly into the thrombus.
The PROACT studies evaluated IAT with recombinant prourokinase (pro-UK) in patients within 6 hours of an MCA (M1 or M2 segment) occlusion stroke. The PROACT-II study (a phase III prospective, randomized, placebo-controlled study) enrolled 180 patients with a median NIHSS score of 17 (range, 4–30). Favorable outcome (mRS score of 0–2 at 90 days) was achieved in 40% of patients treated with IA pro-UK (9 mg; plus low-dose heparin) versus only 25% of control subjects (low-dose heparin only) ( P = .04). The recanalization rate was significantly higher in the pro-UK group (66%) than in the control group (18%) ( P <.001). Although the rate of SICH was higher in patients given pro-UK (10%) than in control patients (2%) ( P = .06), no difference in mortality was observed. Although only IA pro-UK was used for thrombolysis in the PROACT trials, t-PA is used for thrombolysis in the United States because of the unavailability of pro-UK here. However, the FDA has not yet approved the use of t-PA for IA thrombolysis.
Two studies in Japan evaluated IA urokinase treatment. From these IA thrombolysis studies, it is apparent that recanalization rates may differ with occlusion site and stroke cause. Lower recanalization rates were observed in patients with thromboembolic carotid terminus occlusion (30%) relative to MCA M1 segment (50%) or M2 segment (90%) occlusions. Therefore, considerations of the nature of the occlusion may inform endovascular planning.
Combined IV and IA thrombolysis
The IMS studies investigated the feasibility and safety of a combined IV and IA approach to thrombolysis. In IMS-I, 80 patients were enrolled within 3 hours of stroke onset; median baseline NIHSS score was 18. The patients received 0.6 mg/kg of IV t-PA followed by 22 mg of IA t-PA via a 2-hour infusion or until thrombolysis had been achieved. The outcome of this study was compared with that for the NINDS IV t-PA stroke trial. IMS subjects had a significantly better outcome at 3 months than NINDS placebo-treated subjects for all outcome measures (OR ≥2) but not beyond the benefit conferred to the NINDS IVT group.
IMS-II was a continuous, nonrandomized, safety and feasibility pilot study to evaluate efficacy and safety of IV t-PA (0.6 mg/kg, followed by IA t-PA [22 mg again]) coupled with low-energy sonography (via the EKOS Primo Micro-Infusion Catheter [EKOS Corporation, Bothell, WA]) to theoretically increase fluid permeation and thrombolytic infusion within the clot. IMS-II subjects had significantly better outcomes at 3 months than NINDS placebo-treated subjects for all end points (OR ≥2.7) and better outcomes than NINDS t-PA-treated subjects.
Pooled IMS-I and IMS-II data showed that partial or complete recanalization occurred in 74.6% of ICA terminus and MCA M1 occlusions, with good reperfusion (TIMI 2 or 3) in 61.3%. Good reperfusion (TIMI 2 or 3) correlated strongly with good outcome ( P = .0004). The 3-month mortality was lower (although not of statistical significance) in IMS-II patients (16%), than in placebo-treated (24%) or t-PA–treated (21%) NINDS trial patients. The rate of SICH was higher in IMS-II (9.9%) but not significantly different from that in the NINDS trial (6.6%). The definitive randomized controlled trial, IMS-III, is ongoing; and the results will likely provide class I evidence for the concept of combined IV-IA therapy.
Mechanical thrombectomy
The most common method for mechanical thrombus disruption is probing the thrombus with a microguidewire. This technique seems to be useful in facilitating pharmacologic thrombolysis. Systems for mechanical recanalization can be divided into 2 major groups according to where they apply force on the thrombus. Proximal devices apply force to the proximal base of the thrombus. This group includes various aspiration catheters. Distal devices approach the thrombus proximally but then are advanced over a guide wire and microcatheter across the thrombus to be unsheathed distally, where force is applied to the distal base of the thrombus. This group includes snarelike, basketlike, or coil-like devices. In an animal model, proximal devices were faster in application and were associated with a low complication rate. The distal devices were more successful at removing thrombotic material, but their method of application and attendant thrombus compaction increased the risk of thromboembolic events and vasospasm. Advantages and disadvantages of mechanical revascularization strategies overall are summarized in Box 1 . Current FDA-approved thrombectomy devices are the Merci retriever (distal device) (Concentric Medical Inc, Mountain View, CA) and the Penumbra aspiration catheter (proximal device) (Penumbra Inc, Alameda, CA).
Advantages
- •
Lessen, and may even preclude, the use of pharmacologic thrombolytics, and thus reduce the incidence of SICH
- •
May extend the treatment window beyond the limit of 6 to 8 hours
- •
Mechanical fragmentation of the clot increases the surface area of the clot available for endogenous and exogenous fibrinolysis
- •
Recanalization time may be faster
- •
May be effective for thrombi or other material resistant to thrombolytics that occlude the vessel
- •
Key option for patients who have a contraindication for pharmacologic thrombolysis, such as recent surgery or abnormal hemostasis, or have a late presentation
Disadvantages
- •
Technical difficulty navigating the devices through the intracranial vasculature
- •
Excessive trauma to the vasculature
- •
Distal embolization from fragmented thrombus
Merci Device
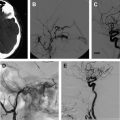
The Merci retriever is a shaped wire constructed of nitinol. The flexible corkscrewlike tip can easily be delivered through a microcatheter into the vessel distal to the occlusion site. When deployed, this device returns to its preformed coiled shape to ensnare the thrombus. The thrombus is bypassed and the retriever deployed from inside the catheter distal to the thrombus. The corkscrewlike tip is pulled back slowly to ensnare the clot as a corkscrew would ensnare a cork. The retriever is then retracted into the guide catheter under proximal flow arrest. Different versions of this device are available. In the first-generation devices (X5 and X6), the nitinol wire was shaped in helical-tapering coil loops. The second-generation devices (L4, L5, and L6) differ from the X devices by the inclusion of a system of arcading filaments attached to a nontapering helical nitinol coil, which has a 90° angle in relation to the proximal wire component. The third-generation devices (V series) have variable-pitch loops in a linear configuration with attached filaments. The retriever device is deployed through a 2.4-F microcatheter (14 X or 18 L, Concentric Medical). The addition of a 4.3-F distal access catheter (DAC) has provided additional coaxial support to the system, resulting in improved delivery ability with the potential for simultaneous thromboaspiration as well. Merci devices are available in various diameters from 1.5 to 3 mm, depending on the caliber of the occluded vessel. These devices are regularly used in combination with proximal balloon occlusion in the ICA, in addition to aspiration from the guiding catheter, to reduce the risk of distal thromboembolism. An illustrative case of Merci retrieval is provided in Fig. 1 .
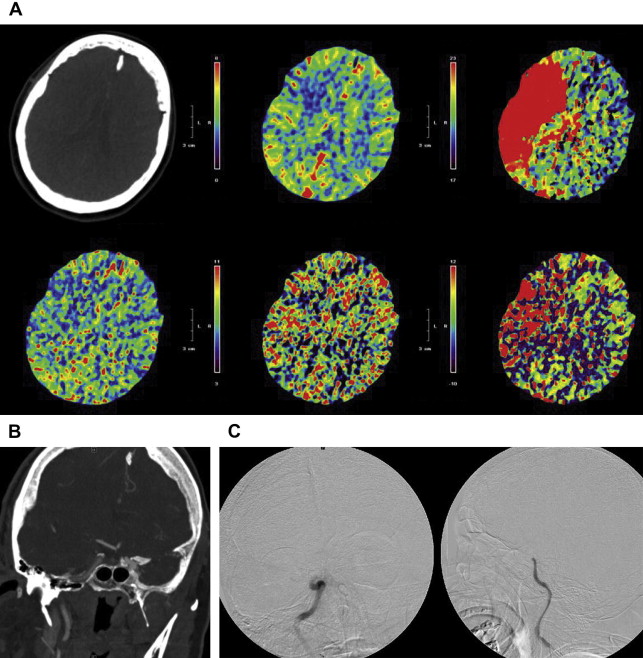
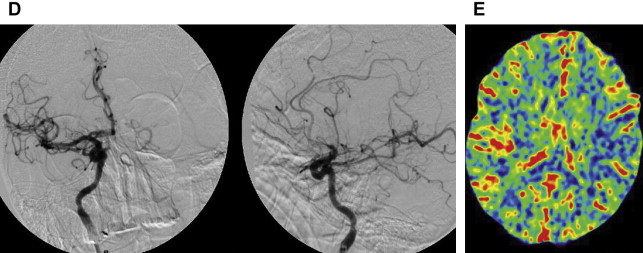
The Merci procedure is performed after femoral artery access is obtained with the Merci balloon guide, which comes in both 8-F and 9-F outer diameters. Once the balloon guide is in the conduit vessel of interest, a medium-sized catheter (available in 4.2-F and 5.3-F outer diameters), the DAC (used for triaxial support), and microcatheter of choice are advanced over the microwire to the clot under direct fluoroscopic guidance. The microwire is then exchanged for the Merci retriever system with placement distal to the clot. The balloon guide is then inflated. Using a slow, steady pulling motion, the retriever engages the clot while the DAC position is maintained. Then, as the clot moves proximally, the DAC, microcatheter, and retriever are moved toward the guide while aggressive aspiration is performed from the guide. The retriever can be resheathed and the steps repeated. The Merci device often requires 3 to 6 passes before flow is restored, which delays the time to recanalization. The aforementioned new DAC available for use with the Merci device allows placement of smaller triple coaxial guide catheter near the lesion of interest to transmit the traction force better in a straighter angle at a shorter distance.
FDA approval of the Merci device in 2004 was based on a review of data obtained in the multicenter Merci trial that involved 141 patients (mean age, 60 years; mean NIHSS score, 20) ineligible for standard thrombolytic therapy. The Multi-MERCI trial was a prospective, multicenter, single-arm registry that included 164 patients (mean age, 68 years; mean NIHSS score 19) treated with different Merci retrieval systems (X5, X6, and L5). Patients with persistent large vessel occlusion after IVT (with t-PA) were also included in the study, and adjunctive IAT (also with t-PA) was allowed. The Merci device has led to an increase in the rates of recanalization of intracranial ICA occlusions. Two ongoing prospective randomized trials are using the device, namely the MR and Recanalization of Stroke Clots Using Embolectomy (MR-RESCUE) trial ( NCT00389467 ) and the IMS-III trial.
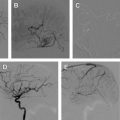
Penumbra Device
From a procedural approach, proximal devices, like the Penumbra, are comparable with IAT. The Penumbra system has 3 main components: a reperfusion catheter, separator, and a thrombus removal ring. A Penumbra procedure is performed after arterial access is obtained and usually after systemic heparinization. All but the largest components of the Penumbra system are deliverable through a 6-F standard guide catheter; a 070 Neuron catheter is the guide designed for the system. The reperfusion catheter is advanced past the guide catheter over a guidewire and placed proximal to the clot. The catheters and separators are available in different sizes for various arterial diameters, including 026, 032, 041, and 057. The 057 aspiration system requires a 6-F long sheath in the access vessel for delivery (shuttle). The guidewire is then removed from the reperfusion catheter, and the penumbra separator is advanced through the reperfusion catheter. The aspiration pump is started and a continuous aspiration, clot disruption-debulking process is performed with the separator. In general, the Penumbra device works better in straight arterial segments than around curves or at branch points, because the separator may cause arterial perforation. In addition, the largest catheter possible should be used to allow for the greatest amount of aspiration, because suction decreases with decreasing vessel diameter. Another advantage of the Penumbra system is that it can reach more distal vessels, such as proximal M3 vessels, compared with other thrombectomy devices that are difficult to manipulate in these distal vessels. The use of the Penumbra system in a patient with acute ischemic stroke is illustrated in Fig. 2 .