Fair Game
Answers
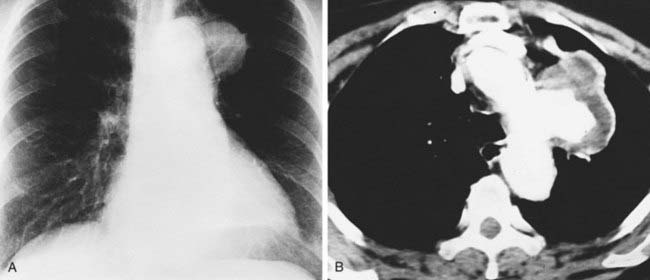
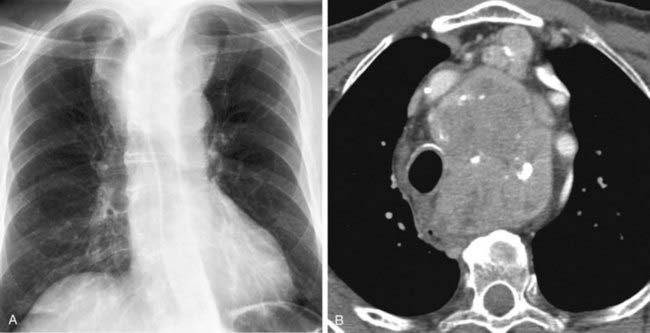
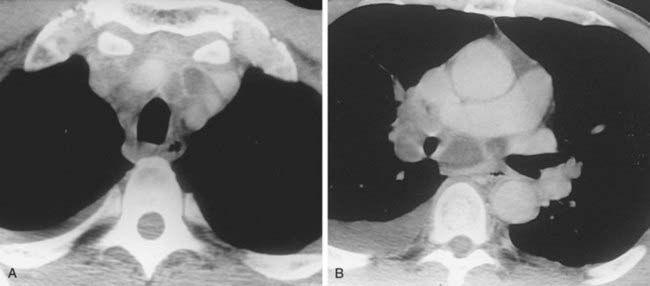
Case 38
3 Name at least three sites of primary neoplasms that commonly result in metastatic intrathoracic lymphadenopathy.
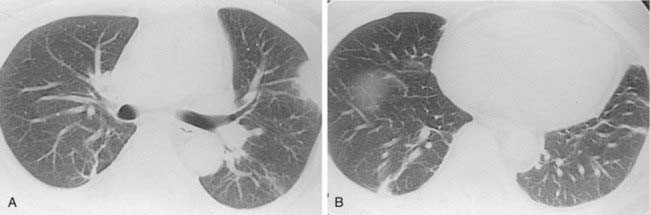
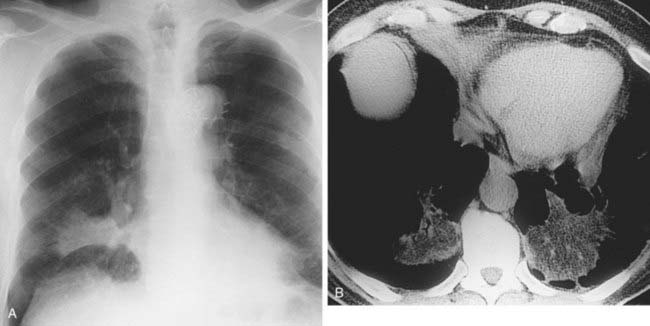
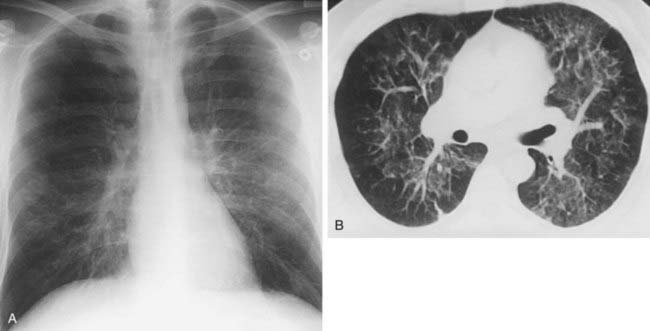
Case 39
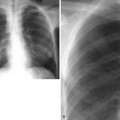
1 What term is used to describe the confluent areas of lung opacification observed in these two patients?
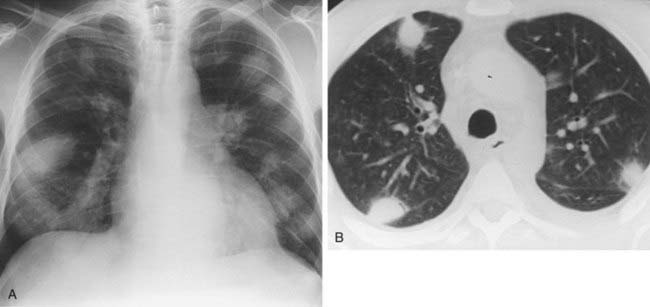
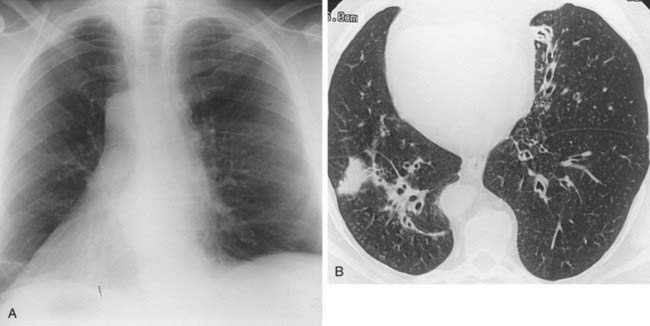
Case 40
2 Name at least three causes of chronic infiltrative lung disease that are associated with a basilar and subpleural distribution of abnormalities.
3 Which is more common among patients with progressive systemic sclerosis—nonspecific interstitial pneumonia (NSIP) or usual interstitial pneumonia (UIP)?
Answers
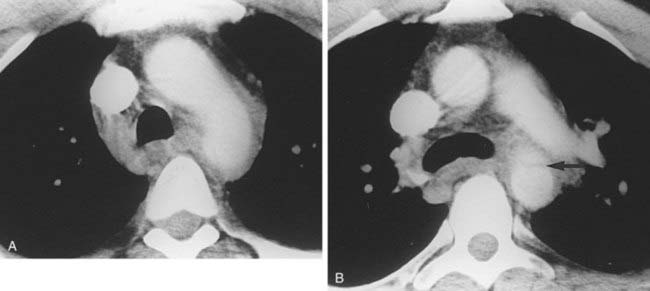
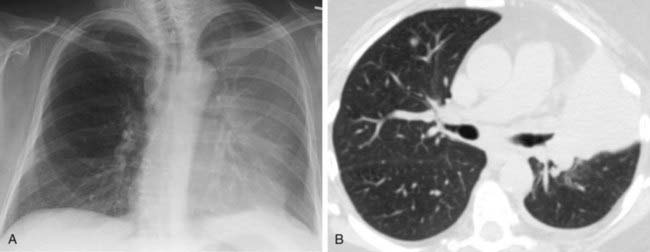
Case 41
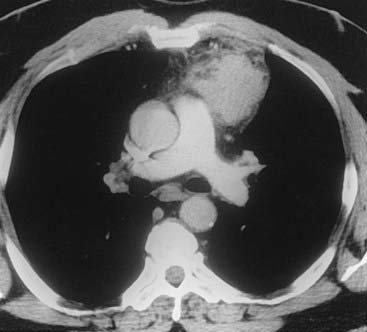
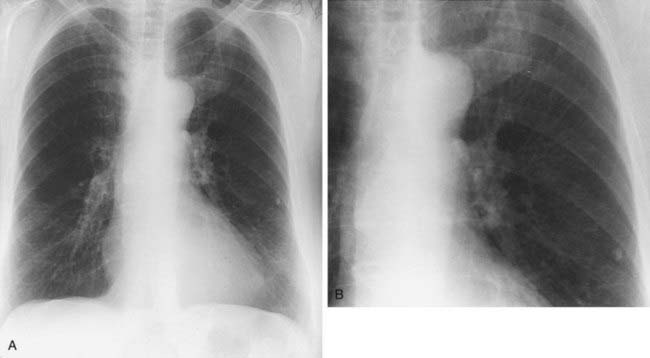
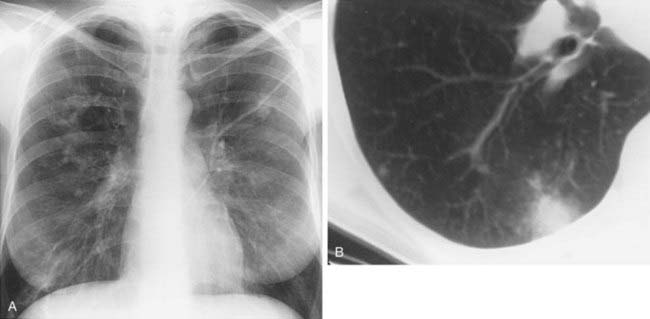
1 What term is used to describe the combination of a calcified lung nodule and calcified lymph nodes?
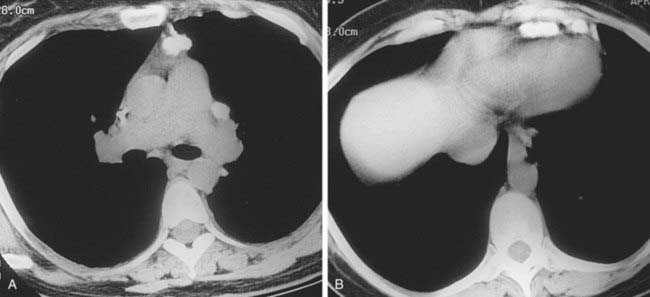
Case 42
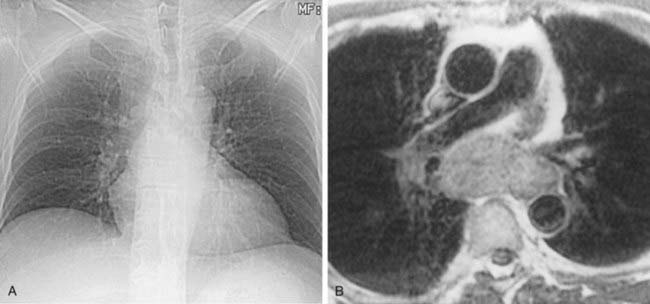
1 What is the most likely cause for the acute interstitial process demonstrated in the second figure?
3 At approximately what pulmonary venous wedge pressure (PVWP) would you expect to detect Kerley lines?
Answers
Case 42
Interstitial Edema
2 Kerley A lines are centrally located, radiate from the hila, and measure 2 to 6 cm in length; Kerley B lines are peripherally located, usually extend to the pleural surface, and measure less than 2 cm in length.
4 Peribronchial cuffing, indistinct pulmonary vessels; interlobular septal thickening (Kerley lines), and thickening of the fissures.
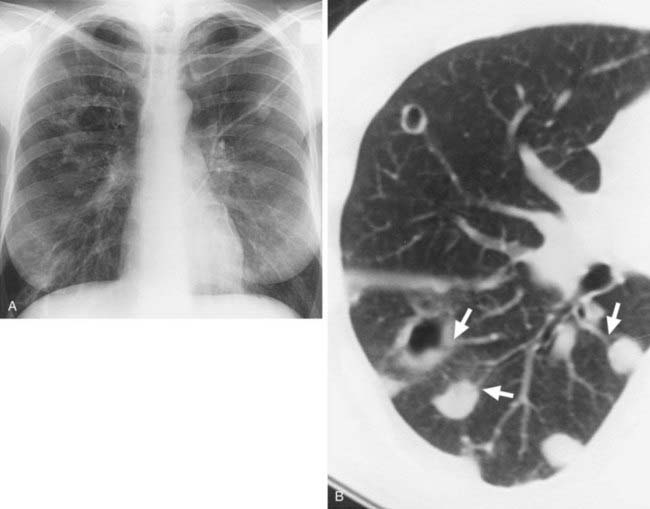
Case 43
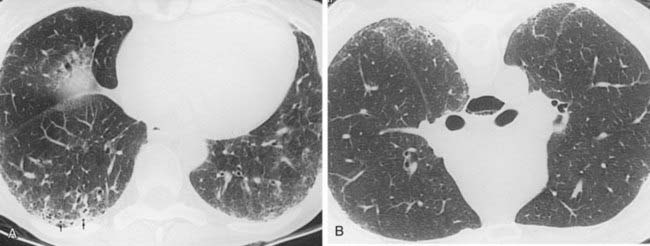
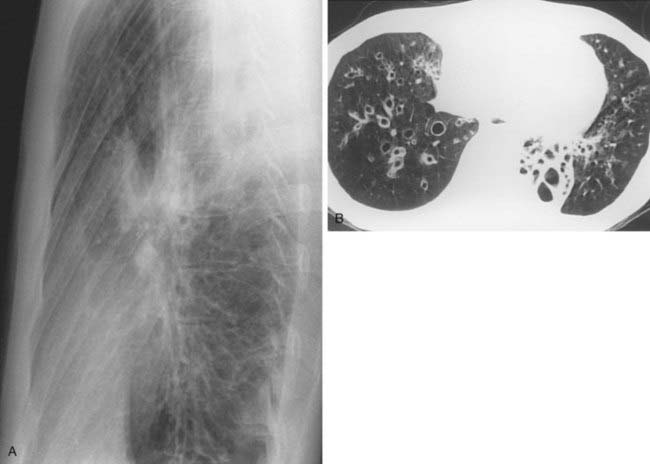
3 What conventional radiographic sign of bronchiectasis is evident in the lung bases in the first figure?
Answers
Answers
Case 44
Arteriovenous Malformation
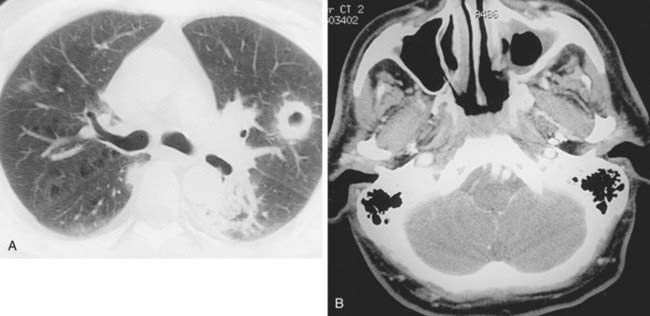
2 Hereditary hemorrhagic telangiectasia (HHT), also known as Osler-Weber-Rendu disease, which is characterized by telangiectasias, AVMs, and aneurysms in multiple organ systems (including pulmonary, gastrointestinal, cutaneous, and central nervous system).
Case 46
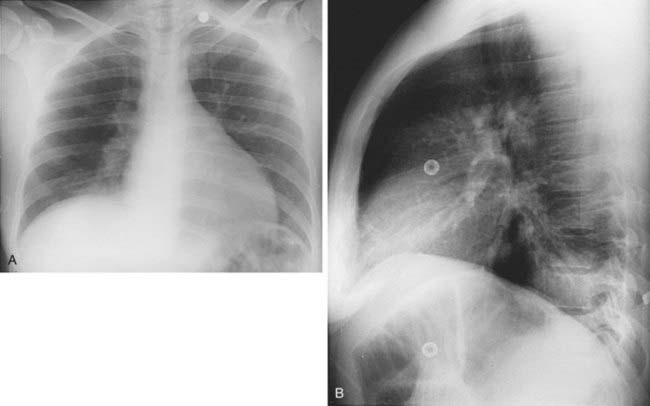
1 What is the differential diagnosis for the wedge-shaped, peripheral consolidation in the left lung in the first figure?
2 What is the significance of the identification of a feeding vessel directed toward the apex of the consolidation?
3 What feature of peripheral consolidations has been described as highly suggestive of pulmonary infarction?
Answers
Case 47
1 What is the differential diagnosis of multiple lung nodules or masses in a patient with acquired immunodeficiency syndrome (AIDS)?
2 In AIDS patients, is the doubling time of nodules a reliable way to differentiate between benign and malignant conditions?
Answers
Answers
Case 52
3 Based solely on the degree of postobstructive atelectasis present, what is the correct T (tumor) stage for this non–small cell lung cancer (NSCLC)?
Case 54
1 Which type of infection most commonly presents with multiple poorly defined lung nodules in an immunosuppressed patient?
2 Is the “CT halo sign” (a “halo” of ground-glass opacification surrounding a nodule) specific for Aspergillus?
Case 55
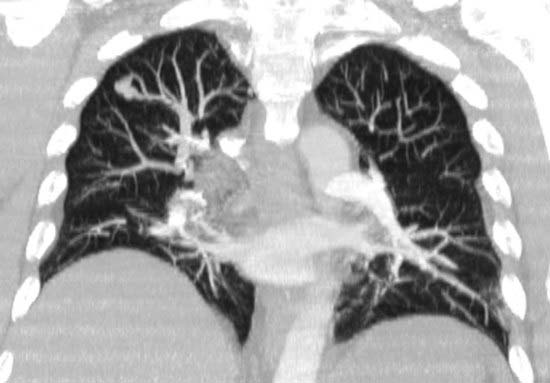
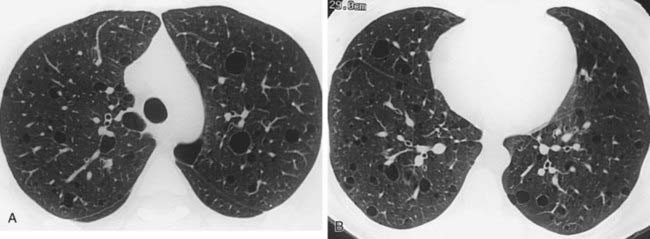
2 Describe the typical demographic features (age, sex) of a patient with lymphangioleiomyomatosis (LAM).
Case 56
1 What is the most likely cause of focal consolidation in a human immunodeficiency virus (HIV)–positive patient?
Case 57
3 In patients with primary TB, is lymph node enlargement more common in pediatric or adult patients?
Case 58
2 Is this infection increasing or decreasing in prevalence among HIV-positive patients in the United States?
Answers
Case 59
2 In patients with lymphoma, are calcified lymph nodes usually seen before or after radiation therapy?
Case 60
1 Name two types of infections that may result in rapidly growing nodules in a non-AIDS immunosuppressed patient.
Answers
Case 60
Case 61
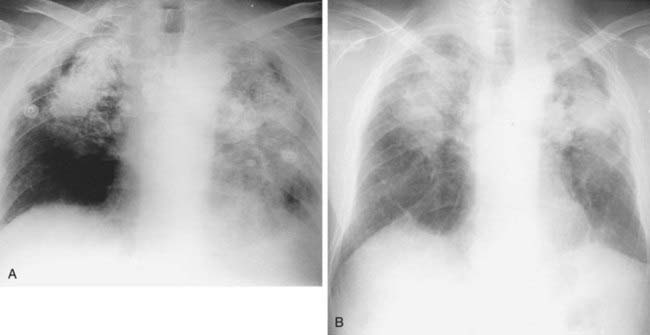
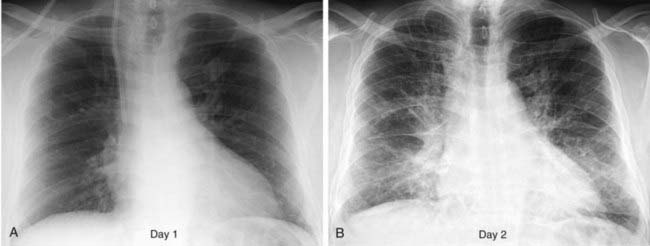
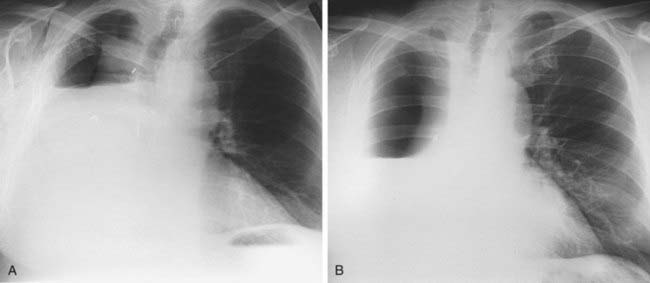
1 What postoperative complication is evident in this case (the first radiograph was obtained prior to the second)?
Answers
Case 61
Bronchopleural Fistula
3 Failure of the pneumonectomy space to fill with fluid; abrupt decrease in the air-fluid level in the pneumonectomy space; contralateral shift of the mediastinum following pneumonectomy; and identification of air in a previously completely opacified pneumonectomy space.