Fig. 1
18F-FDG PET/CT is a sensitive molecular imaging modality for monitoring therapeutic response to chemotherapy in lymphoma, lung cancer etc. Molecular response is noted in the primary tumor and mediastinal lymph node metastases in this 69-year-old male with non small cell lung cancer (a, b, c—pre-chemotherapy; a, MIP; b, transverse fused PET/CT showing lung tumor; c, coronal fused PET/CT demonstrating the mediastinal lymph node and d, e, f—the corresponding 18F-FDG PET/CT images post-chemotherapy)
2 Non-Hodgkin Lymphoma
Currently, two radionuclides have been licensed for radioimmunotherapy of CD20-positive low-grade or transformed B cell lymphomas; 90Y-ibritumomab tiuxetan (Zevalin®, IDEC Pharmaceuticals and Schering AG, Berlin, Germany) and 131I-tositumomab (-Bexxar®, Glaxo Smith Kline, Philadelphia, PA, US). Both these antibodies are effective and induce significant responses in about 75 % of patients, even when the patient has been heavily pretreated with chemotherapy (Fig. 2) (Dancey et al. 2009; Oyen et al. 2007; Larson and Krenning 2005).
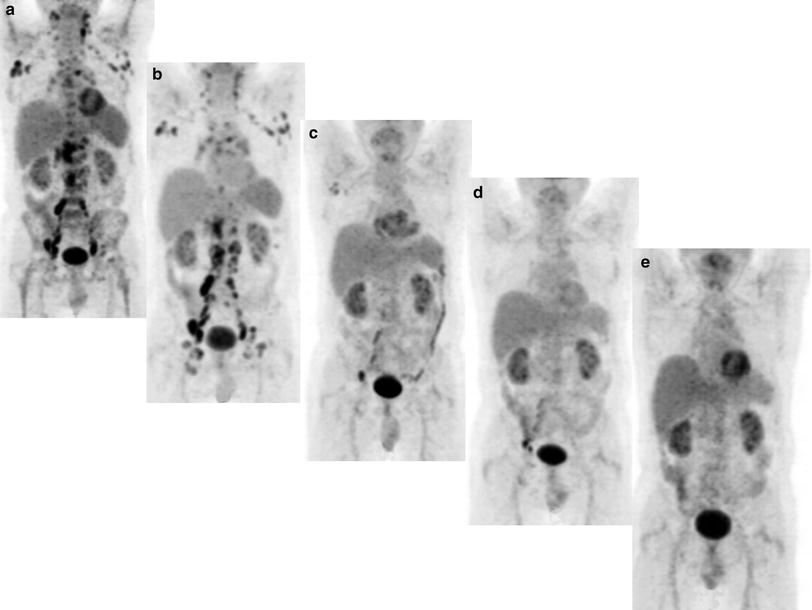

Fig. 2
A 60-year-old male patient with progressive follicular non-Hodgkin’s lymphoma, status post-surgery and chemotherapy, underwent 4 courses of therapy with 90Y-ibritumomab tiuxetan (Zevalin), the cumulative administered activity being 5.4 GBq of 90Y. 18F-FDG PET/CT was performed before, and 2 months after each course of the radioimmunotherapy. Metabolic response to each course of 90Y-Zevalin in the multiple lymph nodes, and metabolic complete remission after the 4th course, can be very well identified on serial 18F-FDG PET (MIP images: a, before therapy; b, after the 1st course; c, after the 2nd course; d, after the 3rd course; and e, and after the 4th course of 90Y-Zevalin therapy)
Most lymphomas, with the exception of extranodal marginal zone lymphoma and small lymphocytic lymphoma, have high 18F-FDG avidity and can thus be monitored with 18F-FDG PET (Weiler-Sagie et al. 2010). Several studies (Torizuka et al. 2000; Ulaner et al. 2008; Bodet-Milin et al. 2008; Jacene et al. 2009; Storto et al. 2010; Lopci et al. 2010; Cazaentre et al. 2010; Lopci et al. 2010) have investigated the utility of 18F-FDG PET for response assessment after radioimmunotherapy in lymphoma. Torizuka et al. (2000) investigated the feasibility of using 18F-FDG PET for the assessment of response to 131I-anti-B1 (CD20) radioimmunotherapy. Fourteen patients with B cell non-Hodgkin lymphoma were first given a tracer dose (i.e. small dose) of 131I-anti-B1 and then the radioimmunotherapeutic dose, each preceded by infusion of unlabeled anti-B1. In 8 of 14 patients, 18F-FDG PET was performed at baseline and 33–70 days after radioimmunotherapy. The other six patients underwent 18F-FDG PET at baseline, 6–7 days after the tracer dose, and 5–7 days after radioimmunotherapy to estimate the early response to tracer dose and radioimmunotherapy. To assess tumor 18F-FDG uptake, standardized uptake value normalized for lean body mass (SUVlean) was measured 1 h after 18F-FDG injection. Evaluation for tumor response, including physical examination, CT (chest, abdomen, and pelvis), bone marrow biopsies (if positive for lymphoma at last evaluation), and blood counts and chemistries, was performed during the tracer studies before radioimmunotherapy, 4–6 and 12 weeks after therapy, and every 2–3 months thereafter. A complete response was defined as a complete disappearance of all detectable disease for at least 1 month or as lack of change in a minimal, residual radiographic abnormality for at least 6 months. A partial response was defined as a reduction of at least 50 % in the sum of the products of the largest perpendicular diameters of all measurable lesions for at least 1 month. After radioimmunotherapy, complete response was observed in six patients, partial response in six, and no response in two. At 33–70 days after radioimmunotherapy, mean SUVlean of six responders markedly declined to 41 % of the baseline value (P < 0.002). Soon after tracer dose and after radioimmunotherapy, mean SUVlean of the other six responders decreased to 79–62 % of the baseline values, respectively (P < 0.05). In two nonresponders, SUVlean did not significantly decline from the baseline value at 37 days after radioimmunotherapy. Torizuka et al. (2000) stated that 18F-FDG PET metabolic data obtained 1–2 months after radioimmunotherapy correlate well with the ultimate best response of non-Hodgkin lymphoma to radioimmunotherapy, more significantly than the early data after tracer dose or radioimmunotherapy. Furthermore, 18F-FDG uptake in non-Hodgkin lymphoma may decline gradually after radioimmunotherapy in responding patients (Torizuka et al. 2000).
Ulaner et al. (2008) retrospectively compared combined 18F-FDG PET/CT with CT alone in the evaluation of 10 patients with relapsed or refractory B cell non-Hodgkin lymphoma who were treated with 90Y-ibritumomab tiuxetan. 18F-FDG PET/CT and CT examinations to evaluate response were performed 4–6 months (mean, 5.6 months) after radioimmunotherapy. Response after treatment at CT was assessed according to the International Workshop Criteria (IWC) (Cheson et al. 1999) and response after treatment at 18F-FDG was assessed according to published criteria of the European Organization for Research and Treatment of Cancer (EORTC) (Young et al. 1999). Interpretation of CT alone resulted in classification of 8 (80 %) of 10 patients as responders to treatment, with 2 patients (20 %) classified as having complete response. At reevaluation with combined 18F-FDG PET/CT, two patients (20 %) had residual lesions at CT that did not show evidence of 18F-FDG avidity. These two patients, classified as partial responders according to CT criteria alone, were classified as complete responders at PET/CT. Both these patients were free of evident disease at 18 or more months of follow-up. The authors of this study concluded that the use of combined 18F-FDG PET/CT may enable superior assessment of response to 90Y-ibritumomab tiuxetan treatment than the use of CT alone, at which one may underestimate 90Y-ibritumomab tiuxetan response by considering inactive residual CT masses to be residual disease (Ulaner et al. 2008).
Bodet-Milin et al. (2008) evaluated 18F-FDG PET imaging for early prediction of response to radioimmunotherapy with the anti-CD22 agent 90Y-epratuzumab tetraxetan in 22 patients with B cell non-Hodgkin lymphoma. Patients underwent 18F-FDG PET and conventional diagnostic methods (including clinical examination, CT, laboratory screening, and bone marrow biopsy if bone marrow was involved at baseline) at baseline and at 6 weeks after each course of radioimmunotherapy. Additional imaging took place every 3 months until progression of disease. Response according to conventional diagnostic methods was assessed using the IWC (Cheson et al. 1999) and response according to 18F-FDG PET was assessed using the Revised IWC (Cheson et al. 2007). Histology and follow-up served as the standard of reference. Of the 31 responses evaluated as unconfirmed complete response by conventional diagnostic methods, 20 (65 %) were classified as negative for disease (complete response) by 18F-FDG PET while the other 11 (35 %) were positive for disease (seven partial response and four progression of disease). Among 22 assessable 18F-FDG PET images acquired at 6 weeks post-radioimmunotherapy, the mean time to progression was 15.6 months when 18F-FDG PET was negative for disease (complete response), compared with 5.4 months when 18F-FDG PET was positive (partial response or progression of disease) (P = 0.008). Sensitivity, specificity, positive predictive value, and negative predictive value of 18F-FDG PET 6 weeks after radioimmunotherapy were 86, 63, 80, and 71 %, respectively, compared with 36, 87, 83, and 44 %, respectively, using conventional diagnostic methods. The authors concluded that their results indicate that radioimmunotherapy responders may be identified with 18F-FDG PET more accurately than with CT, and demonstrate the benefit of using 18F-FDG PET to detect residual disease as soon as 6 weeks post-radioimmunotherapy (Bodet-Milin et al. 2008).
Jacene et al. (2009) retrospectively evaluated 18F-FDG PET/CT for monitoring the response of 33 patients with relapsed or refractory B cell non-Hodgkin lymphoma to 131I-tositumomab (n = 23) or 90Y-ibritumomab tiuxetan (n = 10). 18F-FDG PET/CT was performed before radioimmunotherapy and at 12 weeks after radioimmunotherapy. A third scan was performed in 13 patients at 24 weeks after radioimmunotherapy, 12 of whom did not receive interval therapy. Target lesions up to 10 tumors (per patient) with the most visually intense 18F-FDG uptake, larger than 1 cm, and representative of all involved organs were selected. Tumor metabolic activity was assessed before and after radioimmunotherapy visually and quantitatively by maximum SUVlean (SUVlean max). Response was assessed by the IWC (Cheson et al. 1999) and Revised IWC, which includes 18F-FDG PET (Cheson et al. 2007). Mean lean maximum standardized uptake value (SUVlean max) decreased from baseline in 244 target lesions 12 weeks after radioimmunotherapy (from 6.51 ± 4.05 to 3.94 ± 4.41; P < 0.01), regardless of response at 12 weeks after radioimmunotherapy (P ≤ 0.02). Baseline SUVlean max of target lesions was not predictive of 12 week response. This was also true for the lesion with the highest SUVlean max and for the lesions with the highest 3 SUVs. After radioimmunotherapy, SUVlean max was lower for responders than for nonresponders (P ≤ 0.01). Median percentage change in SUVlean max of target lesions per patient was −51 % (−95 to 97 %). No significant difference in decline in SUVlean max between patients who received 131I-tositumomab and those who received 90Y-ibritumomab tiuxetan was demonstrated (−31 ± 51 % vs. −47 ± 46 %; P = 0.38). Patients with greater than a 52 % decline in SUVlean max tended toward longer survival (P = 0.09) than those with lesser declines. The 12 week overall response rate to radioimmunotherapy based on the IWC was 42 % (14/33); complete response rate was 15 % (5/33). Eleven of twelve patients with progression at 12 weeks had new disease sites, and in four patients, new disease sites were the only sites of progression. Of 108 lesions evaluated at 12 and 24 weeks after radioimmunotherapy, 49 resolved at 12 weeks and remained resolved at 24 weeks, 17 gradually declined in SUV over 24 weeks, and 37 initially decreased at 12 weeks but increased at 24 weeks. 18F-FDG PET/CT showed disease progression at 24 weeks in 10 of 13 patients; seven patients had new lesions and one patient was reclassified from partial response to complete response. Based on these results, Jacene et al. (2009) concluded that in non-Hodgkin lymphoma, 18F-FDG uptake in tumors typically drops significantly after radioimmunotherapy. A continued decline in tumor SUVlean max between 12 and 24 weeks without additional therapy can occur, suggesting a need for delayed-response assessment. In patients who progress after radioimmunotherapy, new sites of disease commonly develop, rather than recurrence or progression at previous disease sites. Large declines in 18F-FDG uptake tend to be seen in those with the longest progression-free survival (Jacene et al. 2009).
Storto et al. (2010) prospectively compared the assessment of metabolic response to 90Y-ibritumomab tiuxetan therapy by using 18F-FDG PET/CT at 2 and 6 months to determine the most appropriate time to detect therapeutic response in refractory B cell non-Hodgkin lymphoma (NHL) patients treated with radioimmunotherapy. Twenty three consecutive patients with relapsed or refractory follicular non-Hodgkin lymphoma underwent 18F-FDG PET/CT at baseline and at 2 and 6 months after radioimmunotherapy. Response was assessed by using the IWC (Cheson et al. 1999) and Revised IWC (Cheson et al. 2007) as well as the criteria of the EORTC (Young et al. 1999). 18F-FDG PET/CT performed at 2 months revealed complete (n = 12) or partial (n = 4) metabolic response in 16 of 23 patients with complete or partial clinical response. These findings were all confirmed at 6 month scanning. 18F-FDG PET/CT indicated refractory or persistent disease at 2 and 6 months in the remaining seven patients. Better overall survival was observed for patients with a reduction in the maximum SUV of 49 % or higher (both at 2 and 6 months after radioimmunotherapy) when compared with those with a decrease of less than 49 % (P < 0.05). The authors concluded that early assessment of response to radioimmunotherapy by using 18F-FDG PET/CT might be useful in the identification of patients needing additional therapeutic strategies (Storto et al. 2010).
Lopci et al. (2010) retrospectively evaluated 18F-FDG PET/CT scans from 59 patients with relapsed or refractory follicular lymphoma who were treated with 90Y-ibritumomab tiuxetan. Response was assessed by using the Revised IWC (Cheson et al. 2007). Post-radioimmunotherapy 18F-FDG PET/CT documented 45.8 % complete responders, 25.4 % partial responders, and 28.8 % nonresponders (i.e. stable disease and progressive disease), with an overall survival of 71.2 % (range 59.5 % −90.9 %). With a median follow-up period of 23 months, multivariate analysis showed that post-radioimmunotherapy 18F-FDG PET/CT remained as the single independent predictor of progression-free survival (P < 0.00001) (Lopci et al. 2010).
Cazaentre et al. (2010) retrospectively evaluated the value of pretreatment 18F-FDG PET in predicting therapy response in 35 patients with B cell non-Hodgkin lymphoma, who were either treated with 90Y-ibritumomab tiuxetan (n = 17) or with 90Y-epratuzumab tetraxetan (n = 18). For each tumor lesion in each patient: maximum and mean SUVs (SUVmax and SUVmean), functional lesion volume (LVol), and total lesion glycolysis (TLG, product of the volume and the SUVmean). For each patient, the highest SUVmax and SUVmean, cumulative TLG (TLGcum), and sum of all LVol (TVol) were determined and their predictive values on response (complete or partial response according to the IWC (Cheson et al. 1999)) were compared with those of conventional prognostic factors, for the 90Y-ibritumomab tiuxetan-treated and 90Y-epratuzumab tetraxetan-treated patients separately. A total of 154 lesions were analyzed. Nineteen patients (54 %) responded to radioimmunotherapy according to the IWC. In the patients treated with 90Y-ibritumomab tiuxetan, response rate was 54, 75 and 75 % in patients with a SUVmax < 20 g/ml, a TVol < 100 ml, and a TLGcum < 1060 g, respectively, while no patient above these thresholds responded (P < 0.005). In the patients treated with 90Y-epratuzumab tetraxetan, response rate was 93 % for patients with an SUVmax < 15 g/ml while no patient above this threshold responded. With TLGcum below 1,360 g, 100 % of patients responded, compared with 37 % of patients whose TLGcum was above this threshold (P < 0.05). By contrast, conventional prognostic factors (including age, gender, Ann Arbor stage, presence of bone marrow involvement, lactate dehydrogenase level, performance status according to the Eastern Cooperative Oncology Group, international prognostic index, and tumor burden) failed to predict response. Cazaentre et al. (2010) concluded that their preliminary results indicate that pretreatment 18F-FDG PET functional parameters such as SUVmax and TLG may help in predicting more accurately the response to single agent 90Y-based radioimmunotherapy. Nevertheless, Cazaentre et al.’s results (2010) are somehow conflicting with those of Jacene et al. (2009) who reported that baseline SUV measurements were not predictive of 12 week response.
Lopci et al. (2010) investigated the value of 18F-FDG PET in 38 patients with relapsed or refractory follicular lymphoma, before and 3 months after treatment with 90Y-ibritumomab tiuxetan. Response according to 18F-FDG PET was assessed using the Revised IWC (Cheson et al. 2007). Final assessment was done 9 months post-radioimmunotherapy, including clinical evaluation, other imaging techniques, and/or biopsy, when necessary. At 18F-FDG PET before treatment, 20 patients out of 38 had a limited disease (nodal involvement on one side of the diaphragm: 7 above and 13 below), 11 patients had nodal findings on both sides of the diaphragm and the remaining seven patients had both nodal and extranodal findings. At 3 months post-radioimmunotherapy, 21 patients (55 %) were in complete remission, 13 patients (34 %) had a partial response, and four patients (11 %) had progressive disease. At final assessment, 21 patients (55 %) were in complete remission, 5 patients (13 %) had a partial response, and 12 patients (32 %) had progressive disease. Out of the 12 patients with progressive disease at final assessment, four were the already known responders at 3 months post-radioimmunotherapy, while the other eight cases were relapsed patients, all belonging to the progressive disease group at 3 months post-radioimmunotherapy. When comparing the disease extent at relapse and the response to treatment, a higher rate of complete response (75 %) was noted in patients with limited disease, while in patients with diffused nodal and/or extranodal findings, it was more frequent a partial response or progressive disease (66 %). Lopci et al. (2010) concluded 18F-FDG PET to be useful in assessing treatment response. In addition, a potential correlation can also be picked out between the disease extent at relapse and the complete response rate, with reasonable 18F-FDG PET predictivity for the final outcome (Lopci et al. 2010).
Other, non-18F-FDG PET radiotracers may also be of use for response assessment after radioimmunotherapy in lymphoma, but have been less intensively investigated. Buck et al. (2007) investigated the utility of the in vivo proliferation marker 3′-deoxy-3′-18F-fluorothymidine (18F-FLT) for early evaluation of the response of lymphoma to antiproliferative treatment in a mouse xenotransplant model. Ten mice served as controls and 10 other mice were treated with 90Y-ibritumomab tiuxetan. Forty eight hours after treatment, antiproliferative effects were assessed with 18F-FLT. Ninety minutes after 18F-FLT administration, mice were sacrificed and radioactivity within the tumor and normal organs was measured using a gamma counter and calculated as % injected dose (ID)/g. In untreated lymphoma, the mean proliferation fraction was 83.6 %. After 90Y-ibritumomab tiuxetan treatment, the mean proliferation fraction decreased to 78.8 % (P = 0.014). In none of the animals was a significant change in tumor size observed. However, tumoral 18F-FLT uptake in untreated lymphoma (5.4 % ID/g) was not significantly different either from that of 90Y-ibritumomab tiuxetan-treated lymphoma (5.8 % ID/g). The mild reduction in the proliferation fraction at 48 h can be explained by the fact that 90Y-ibritumomab tiuxetan can cause relatively later cytotoxic effects. Furthermore, the authors hypothesized that tumor heterogeneity could explain that this reduction in proliferation fraction was not associated with a significant decrease in tumoral 18F-FLT uptake (Buck et al. 2007).
Overall, initial results confirm that 18F-FDG PET has an advantage over anatomy-based imaging alone, and indicate its potential for response assessment at an early stage after initiation of radionuclide therapy in lymphoma. Nevertheless, future prospective well-designed studies are necessary before PET, and in particular 18F-FDG PET, can become an established tool for response assessment after radionuclide therapy in lymphoma. Future studies should also determine at which time points after radionuclide treatment, PET imaging is most effective in assessing response.
3 Neuroendocrine Tumors
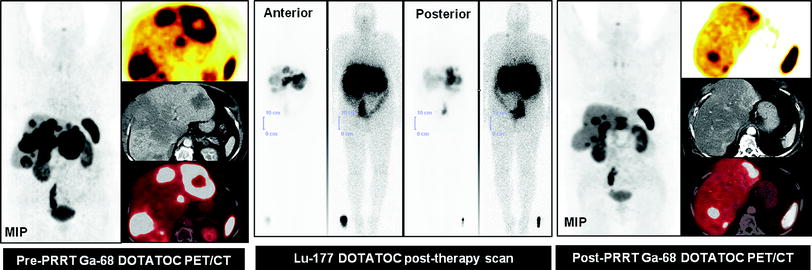
The use of radiolabeled somatostatin (SMS) analogs is a relatively new treatment modality for somatostatin-receptor-positive endocrine tumors. Several different radiolabeled SMS analogs have been applied and an overview can be found in the literature (Kwekkeboom et al. 2009; Van Essen et al. 2009). At present, guidelines recommend to use accepted standards such as WHO or RECIST criteria to assess response of neuroendocrine tumors following radionuclide therapy with radiolabeled SMS analogs (Kwekkeboom et al. 2009). However, guidelines also acknowledge that CT and MRI may underestimate responses in case of cystic or necrotic tumors (Kwekkeboom et al. 2009). In order to more accurately assess therapy response, it may be necessary to shift to a new approach in which functional imaging is integrated with anatomical imaging. Molecular imaging using somatostatin receptor PET/CT (SSTR PET/CT) with 68Ga-labeled SMS analogs and molecular radiotherapy applying Peptide Receptor Radionuclide Therapy (PRRT) with 90Y and/or 177Lu-labeled peptides is an excellent example of THERANOSTICS and personalized medicine (Baum et al. 2012). Utilizing the identical diagnostic SMS analog as for therapeutics, SSTR PET/CT provides quantitative and reproducible data which can be used for evaluation of therapy response to PRRT (Fig. 3).