Outline
Technique, 1001
Normal Anatomy, 1002
Normal Gonadal and Reproductive Tract Development, 1002
Structural Anomalies of the Reproductive Tract, 1004
Ambiguous Genitalia, 1009
Prepubertal Bleeding, 1011
Primary Amenorrhea, 1015
Pelvic Pain, 1018
Adnexal (Ovarian or Tubal) Torsion, 1018
Ovarian Cyst, 1018
Pelvic Inflammatory Disease, 1019
Ectopic Pregnancy, 1019
Acute Appendicitis, 1019
Genital Hernia, 1020
Gynecologic Pelvic Masses, 1020
Conclusion, 1022
Summary of Key Points
- •
Reproductive tract anomalies may present at different stages of life. Most abnormalities of the external genitalia are obvious at birth, whereas obstructive and nonobstructive lesions of the reproductive tract may be evident at birth or present later during childhood, at puberty, in adolescence, or during adulthood.
- •
Sonography is used to determine the presence of a uterus and gonadal location in patients with ambiguous genitalia.
- •
In preadolescent girls with vaginal bleeding, sonography can be used to confirm or exclude a postpubertal appearance of the internal genitalia; to diagnose an estrogen-secreting adrenal or ovarian tumor; and to diagnose a vaginal foreign body or mass.
- •
In girls with primary amenorrhea, sonography is useful in determining the presence and morphologic appearance of the uterus and ovaries.
- •
Sonography is a key tool in evaluating pelvic pain in girls, including those with suspected appendicitis.
- •
In patients with adnexal torsion, the involved ovary is always significantly enlarged, with a median volume 12 times that of the normal contralateral ovary.
- •
Bleeding into an ovarian cyst in utero or after birth has a high association with long-term ovarian loss.
- •
Ovarian tumors in the pediatric population are usually benign, with cystic teratomas accounting for more than 90% of all benign ovarian tumors.
The most frequent indications for gynecologic ultrasound imaging in the pediatric and adolescent patient are ambiguous genitalia, prepubertal vaginal bleeding, primary amenorrhea, pelvic pain, and pelvic mass. Sonography is the primary imaging modality used in the evaluation of these disorders, with magnetic resonance imaging (MRI) and computed tomography (CT) reserved for further delineation of congenital malformations or tumors.
Technique
The pediatric vagina, uterus, and ovaries are best imaged with transabdominal ultrasound when the patient’s bladder is full. All girls are encouraged to drink fluids and not to void in the hour prior to imaging. Teenagers are asked to drink 16 ounces.
Curvilinear, sector, and linear-array ultrasound transducers are usually sufficient for most pelvic ultrasound examinations. In young girls with urogenital malformation, hydrometrocolpos, a labial mass, or anal atresia, a transperineal approach is often useful ( Fig. 34-1 ). A transvaginal approach is used to complement the transabdominal examination in sexually active adolescents. In the patient with a complex congenital anomaly, genitography performed with water-soluble contrast material along with ultrasound examination is frequently helpful in identifying and characterizing the vagina, urogenital sinus, or cloaca.

Normal Anatomy
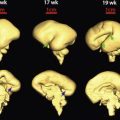
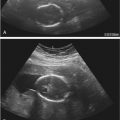
Uterine and ovarian size and shape are age-dependent and under hormonal influence. Maternal and placental hormonal stimulation lead to a relatively large neonatal uterus and ovaries compared to their size in later infancy, at which point they remain relatively stable in size until a growth spurt occurs at approximately 7 to 8 years of age ( Figs. 34-2 and 34-3 ). Mature ovarian follicles can be identified at all ages owing to the secretion of follicle-stimulating hormone (FSH). The prepubertal cervix is greater than or equivalent in thickness to the uterine fundus, and the endometrium is relatively inconspicuous in this age group ( Fig. 34-4 ). Cervical length is approximately twice that of the fundus in the neonate. During childhood, relative cervical length and thickness decrease but uterine fundal length and thickness increase. Average uterine length varies from approximately 2.5 to 4 cm, with thickness less than or equal to 1 cm. Ovarian volume is slightly less than 1 mL. The uterine fundus elongates and thickens during puberty, outstripping the cervix; the endometrium also thickens and undergoes cyclic changes associated with the menstrual cycle.



Normal Gonadal and Reproductive Tract Development
Development of the female reproductive tract is a complex process that involves cellular differentiation, migration, fusion, and canalization with probable apoptosis (programmed cell death). This integrated sequence of events is associated with many opportunities for abnormal development and structural anomalies. Anomalies of the reproductive tract may present at different time points. Most abnormalities of the external genitalia are obvious at birth, whereas obstructive and nonobstructive lesions of the female reproductive tract may be apparent at birth or only later in childhood, at puberty, in adolescence, or during adulthood.
In the first 3 months of embryonic life, the primordia of both the female and male reproductive tracts are present and develop together. The gonads arise after migration of germ cells to the genital ridge, whereas the genital tract evolves from the formation and reshaping of the müllerian (paramesonephric) ducts, urogenital sinus, and vaginal plate.
Biologic differences between males and females are determined genetically during embryonic development. Sex development can be divided into two processes: sex determination , the developmental decision that directs the undifferentiated gonad to develop as a testis or ovary; and sex differentiation , which occurs once the gonad has developed and is induced by the products of the gonad to establish the phenotypic sex. Factors that affect gene expression influence sex determination, whereas factors that influence sex differentiation include hormones and their receptors.
Gonads
In the first and second weeks of development, embryos of both sexes differ only in their sex chromosomes. The gonads arise from the bilateral genital ridges, sites of focal mesothelial thickening of the peritoneum, before 5 to 6 weeks of life. A number of genes are required for development of the bipotential gonad. The bipotential gonad becomes an ovary or testis, depending on differentially expressed genes. Both testicular and ovarian development involve sex-specific pathways that appear to act antagonistically to each other. The normal role of the sex-determining region of the Y chromosome (the SRY gene) in XY gonads is to tip the balance in favor of the testis-specific pathway. If it is absent or abnormal, the gonad differentiates into an ovary. The presence of two X chromosomes appears to be important for the development of a normal, functioning ovary.
The bipotential gonad begins to develop into either a testis or ovary in XY and XX individuals, respectively, at about 6 weeks of life. Differentiation of the gonads leads to testicular and ovarian hormone production and subsequent induction of anatomic and psychological differences. The first sign of gonadal differentiation occurs in the male with the appearance of Sertoli cells at 6 to 7 weeks of life. Leydig cells appear at about 8 weeks. At this stage, the only sign of ovarian differentiation is the absence of Sertoli and Leydig cells. At 9 weeks, primordial germ cells begin to differentiate into oogonia followed by normal ovarian development at 12 to 123 weeks of life. In males, testicular cords will form in the absence of germ cells. However, in females, the ovary will not develop in the absence of germ cells.
Testosterone and antimüllerian hormone are secreted by the Leydig and Sertoli cells, respectively, resulting in the differentiation of the genital primordia into the male phenotype. In the absence of a functioning testis, regardless of whether or not an ovary is present, the genital primordia will differentiate into the female phenotype. The presence of a functioning testis is necessary but not sufficient for the development of a male phenotype; normal androgen metabolism is also required.
In adolescence, an increase in adrenal androgen secretion is associated with the appearance of pubic and axillary hair. Estrogen is responsible for breast development; for maturation of the uterus, vagina, and external genitalia; and for the initiation of the menstrual cycle. Excess androgens of either adrenal or ovarian origin may cause acne, hirsutism, clitoromegaly, increased muscle mass, and deepening of the voice.
External Genitalia and Reproductive Tract
External genital development in the male occurs between the 8th and 12th weeks of life and requires high levels of circulating testosterone, the conversion of testosterone to dihydrotestosterone (DHT) by 5α-reductase in the target organs, and functional androgen receptors. Under the influence of DHT, in the male the urogenital sinus gives rise to the prostate gland; the genital tubercle forms the glans penis; the labiourethral folds form the urethra and ventral shaft of the penis; and the labioscrotal folds fuse to form the scrotum. In the female, or in the absence of testicular tissue secreting biologically active testosterone, functioning androgen receptors, or 5α-reductase, the genital tubercle forms the clitoris; the labiourethral folds form the labia minora; and the labioscrotal folds form the labia majora ( Fig. 34-5 ).

The cell layers involved in the formation of the female reproductive tract include the mesoderm, endoderm, and ectoderm. The mesoderm gives rise to the mesonephros and metanephros. In normal development, the mesonephros involutes, leaving only the wolffian ducts, and the metanephros eventually matures into the kidney. A defect within or insult to these mesodermal structures may ultimately result in congenital anomalies of the gonads, kidneys, and associated ducts. The urogenital sinus arises from the endoderm and gives rise to the bladder and urethra in males and females. In females it also forms the vestibule. The urethral and paraurethral glands in girls and the prostate gland in boys develop as outpouchings of the urethra. Nervous tissue, including sensory epithelium, arises from the ectoderm. Fusion of endoderm and ectoderm is involved in the canalization process; defects lead to fusion failure or obstructive lesions.
During the “indifferent” stage of development, two pairs of genital ducts develop from mesodermal tissue in both males and females: the mesonephric (wolffian) and paramesonephric (müllerian) ducts. The paired wolffian ducts connect the mesonephric kidney to the cloaca. The ureteric bud arises from the wolffian duct at about the 5th week of life and induces differentiation of the metanephros, which eventually becomes the functioning kidney; the mesonephric kidney involutes at 10 weeks. The paramesonephric (müllerian) ducts are identified in both sexes at 6 weeks of life. As they grow, the müllerian ducts lie lateral to the wolffian ducts until they reach the caudal end of the mesonephros where they extend medially to nearly touch in the midline near the cloaca. The urorectal septum forms by the 7th week, separating the urogenital sinus from the rectum. In males, regression of the müllerian ducts begins at 8 weeks of life and is almost complete by 10 weeks. In females, the müllerian ducts extend caudally, reaching the urogenital sinus by 9 weeks to form the uterovaginal canal, which inserts into the urogenital sinus at the müllerian tubercle. By the 12th week, the two ducts have completely fused into a single tube, the uterovaginal canal. Two solid evaginations of urogenital sinus origin, the sinovaginal bulbs, grow from the distal müllerian tubercle. Proximal to the sinovaginal bulbs, outgrowths from the distal müllerian ducts result in formation of the vaginal plate. The upper portion of the vagina is formed by vacuolization of the vaginal plate, whereas the lower portion is formed by vacuolization of the sinovaginal bulbs. Canalization begins caudally and proceeds proximally; it is complete by the 5th month of gestation. The distal-most portions of the sinovaginal bulbs form the hymenal tissue which perforates prior to birth ( Figs. 34-5 and 34-6 ). The upper portions of the müllerian ducts form the fallopian tubes.

Structural Anomalies of the Reproductive Tract
Abnormalities related to lateral fusion, vertical fusion, or resorption, agenesis and/or hypoplasia result in structural anomalies of the reproductive tract. A number of classifications of these anomalies have been proposed, although none is complete. The American Society for Reproductive Medicine (ASRM) system identifies groups with similar clinical manifestations and prognosis, although vaginal anomalies are not included ( Table 34-1 , Fig. 34-7 ). The Vagina Cervix Uterus Adnexa–Associated Malformations classification describes abnormalities not covered by the ASRM classification ( Table 34-2 ).
| Type I | Müllerian Agenesis or Hypoplasia |
| |
| Type II | Unicornuate Uterus |
| |
| Type III | Uterus Didelphys |
| Type IV | Uterus Bicornuate |
| |
| Type V | Septate Uterus |
| |
| Type VI | Arcuate |
| Type VII | DES-Related Anomalies |
|

| Vagina (V) | 0 | Normal |
| 1a | Partial | |
| 1b | Complete hymenal atresia | |
| 2a | Incomplete septate vagina <50% | |
| 2b | Complete septate vagina | |
| 3 | Stenosis of the introitus | |
| 4 | Hypoplasia | |
| 5a | Unilateral atresia | |
| 5b | Complete atresia | |
| S1 | Sinus urogenitalis (deep confluence) | |
| S2 | Sinus urogenitalis (middle confluence) | |
| S3 | Sinus urogenitalis (high confluence) | |
| C | Cloacae | |
| + | Other | |
| # | Unknown | |
| Cervix (C) | 0 | Normal |
| 1 | Duplex cervix | |
| 2a | Unilateral atresia/aplasia | |
| 2b | Bilateral atresia/aplasia | |
| + | Other | |
| # | Unknown | |
| Uterus (U) | 0 | Normal |
| 1 | Arcuate | |
| 1b | Septate <50% of the uterine cavity | |
| 1c | Septate >50% of the uterine cavity | |
| 2 | Bicornuate | |
| 3 | Hypoplastic uterus | |
| 4a | Unilaterally rudimentary or aplastic | |
| 4b | Bilaterally rudimentary or aplastic | |
| + | Other | |
| # | Unknown | |
| Adnexa (A) | 0 | Normal |
| 1a | Unilateral tubal malformation, ovaries normal | |
| 1b | Bilateral tubal malformation, ovaries normal | |
| 2a | Unilateral hypoplasia/gonadal streak (including tubal malformation if appropriate) | |
| 2b | Bilateral hypoplasia/gonadal streak (including tubal malformation if appropriate) | |
| 3a | Unilateral aplasia | |
| 3b | Bilateral aplasia | |
| + | Other | |
| # | Unknown | |
| Associated malformation (M) | 0 | None |
| R | Renal system | |
| S | Skeleton | |
| C | Cardiac | |
| N | Neurologic | |
| + | Other | |
| # | Unknown |
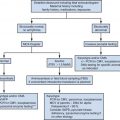
Vaginal and uterine anomalies may be detected in the neonatal period in children undergoing investigation of multiple congenital anomalies; or in adolescents, during workup of amenorrhea, pelvic or abdominal pain, or mass. An initial ultrasound study is usually sufficient in early childhood, with MRI performed at puberty for a more detailed evaluation. In the patient who first presents in adolescence, MRI is usually done following an abnormal ultrasound examination for a comprehensive assessment of the genitourinary tract.
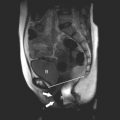
During development of the uterus, failure of septal resorption between the two embryologic müllerian ducts leads to a septate uterus with two endometrial cavities. The septum can be partial or complete, with extension to the internal cervical os. A single cervix is usually present. The outer fundal contour of the uterus is either normal ( Figs. 34-7 and 34-8 ) or flat and broad. A bicornuate uterus results from incomplete fusion of the müllerian ducts. Leave out the part about incomplete development of the uterine horns. The central myometrium may extend to the level of the internal cervical os (bicornuate unicollis) or external cervical os (bicornuate bicollis), and the external surface is indented, typically by more than 1 cm ( Fig. 34-9 ). Complete nonfusion of both müllerian ducts leads to a didelphys uterus ( Fig. 34-10 ). Both horns are fully developed and nearly normal in size. There are always two cervices. There may be a transverse or longitudinal vaginal septum. Although a transverse vaginal septum may occur in association with all of the müllerian duplication anomalies, the greatest association is with didelphys uteri. When there is complete or near-complete arrested development of one müllerian duct, a unicornuate uterus develops with a single horn, an ipsilateral round ligament, and fallopian tube. The unicornuate uterus communicates with a single cervix and a normal vagina. In most patients, arrest is incomplete, and a contralateral rudimentary horn with or without functioning endometrium is present ( Fig. 34-11 ). The rudimentary horn may or may not communicate with the developed uterine horn. Sonography may demonstrate two uterine horns of different sizes. Unless functional endometrium within a noncommunicating horn leads to hematometra or endometriosis, no treatment is generally required. However, because of the potential risk of an ectopic pregnancy developing within a rudimentary noncommunicating horn, some advocate surgical resection.




Failure of fusion and canalization of the müllerian ducts and embryologic urogenital sinus lead to a transverse vaginal septum. Septa are generally less than 1 cm in thickness and extend completely or incompletely across the vagina ( Fig. 34-12 ). There is often a small central or eccentric perforation. A complete vaginal septum may occur in the upper (46%), middle (40%), or lower (14%) vagina. The vagina is short or is a blind-ending pouch. The external genitalia appear normal. Girls may present with mucocolpos in infancy or childhood, hematocolpos in adolescence, or pyohematocolpos from ascending infection through the small perforation. On imaging studies, it is critical to document the presence of a cervix in order to differentiate between a high vaginal septum and cervical atresia, because treatment and prognosis are very different.