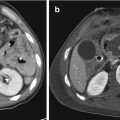
Fig. 3.1
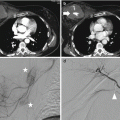
(a, b) Three-dimensional CT reconstructions. Comminute cranial vault fracture, well delineated on 3D reconstructions
The use of contrast media is reserved to evaluate the vascular structures at submillimeter resolution. It should be performed using a rapid contrast bolus injection with vessel tracking; the classic imaging parameters are a slice thickness of 1.25 mm with a 0.625-mm overlap and a bolus injection rate between 3 and 4 mL/s. Its use is required when a vascular injury is suspected, such as pseudoaneurysm, carotid–cavernous fistula, laceration–extravasation, and dissection. It is performed in cases of penetrating head injury or skull base fractures traversing the carotid canal or a venous sinus also (Fig. 3.2). When these findings are positive, a catheter angiography must be performed to confirm and to treat the vascular injuries [24–28].
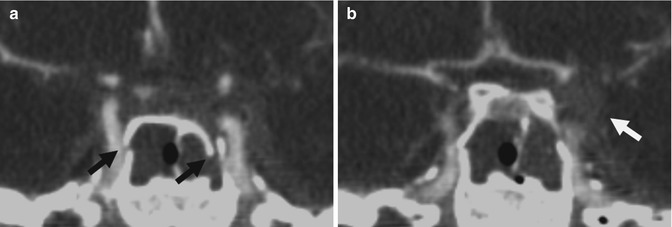

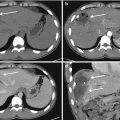
Fig. 3.2
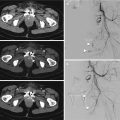
(a, b) CT angiography (coronal reconstructions). Presence of bilateral sphenoid fractures (black arrows); the left internal carotid artery is interrupted at the supraclinoid segment from dissection, associated with a small hemorrhagic collection (white arrows)
In some diagnostic dilemma not resolved by CT scans, magnetic resonance imaging (MRI) can serve as a problem-solving tool, since, owing to its higher contrast resolution for soft tissues, it is by far more capable than CT to detect all the stages of the hemorrhage, diffuse axonal injury, small subdural collections (the only indicator of child abuse), and ischemia.
Nevertheless, MRI has some downsides in the emergency settings, owing to long imaging times, incompatibility of many medical devices, insensitivity to acute subarachnoid hemorrhage, and excessive sensitivity to patient’s motion. The key sequences are fluid-attenuated inversion recovery (FLAIR), gradient-recalled echo (GRE) or better susceptibility-weighted images (SWIs), and diffusion-weighted imaging (DWI). FLAIR is a T2-weighted sequence with suppression of the cerebrospinal fluid, improving detection of lesions in periventricular regions and cerebral cortex; it is very sensitive to cortical contusions, diffuse axonal damage, and subarachnoid hemorrhage. GRE sequences are particularly sensitive to blood breakdown products and can easily depict small areas of micro-hemorrhage not visible on CT. The blood products result in areas of signal loss because of the alteration of local magnetic susceptibility. This sequence is particularly indicated to evaluate subacute and chronic TBI, since hemosiderin can persist indefinitely. SWI is the most sensitive sequence for evaluating petechial hemorrhage (Fig. 3.3); this is a high-spatial-resolution three-dimensional gradient echo technique involving phase post-processing that enhances the paramagnetic properties blood products. By using this sequence, a mask is created to enhance the phase difference between susceptibility artifacts and surrounding tissue, and the contrast-to-noise ratio is optimized by increasing the mask and magnitude images; the phase images are sensitive to regions of local alteration of the magnetic field caused by some substances, such as hemorrhage, iron, and so on. SWI is three to six times more sensitive than GRE to detect hemorrhagic diffuse axonal injuries. A potential drawback of this sequence is the signal loss within the veins because of the magnetic properties of the intravascular deoxygenated hemoglobin and the resultant phase differences with adjacent tissues; to avoid misinterpretation and to minimize partial volume effects, it is auspicable to adjust the thickness of the minimal intensity projection to the brain size [29–36].
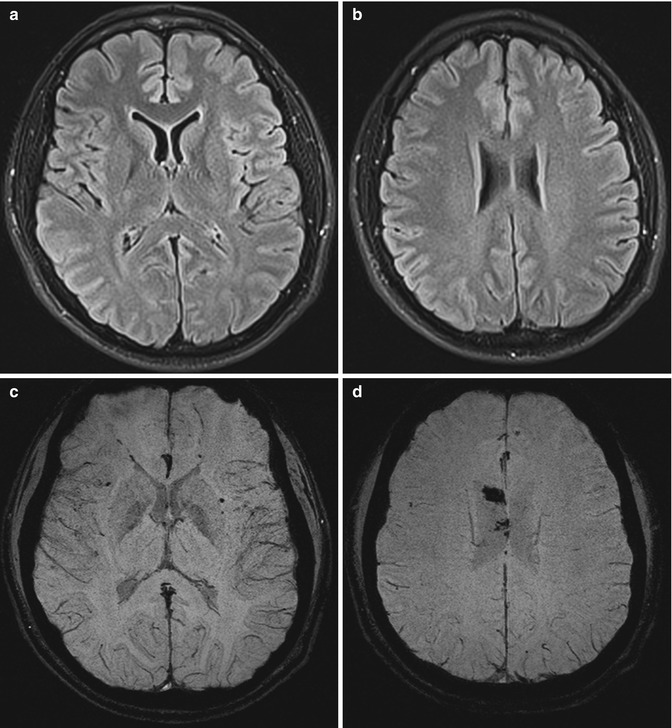

Fig. 3.3
(a, b) FLAIR axial sections; (c, d) SWI axial sections. Presence of small hemorrhagic foci in the right portion of the corpus callosum, the bilateral external capsulae, and the left fronto-mesial region due to axonal damage (c, d), not identifiable on FLAIR (a, b)
DWI is the sequence of choice when an acute diffuse axonal injury (Fig. 3.4) or ischemia is suspected. DWI is based on the random motion of water molecules (Brownian motion), which is affected by the intrinsic speed of water displacement based on tissue properties and type (gray matter, white matter, cerebrospinal fluid). In the nervous system, water diffusion is impeded by cell membranes, myelin sheaths, proteins, and all the subcompartment structures within the living nervous cell; therefore water diffusion is faster in the extracellular than in the intracellular space. When water molecules move from the extracellular space to the intracellular one, the overall diffusion is restricted; this restriction correlates with cytotoxic edema, allowing the early election of ischemia [37, 38].
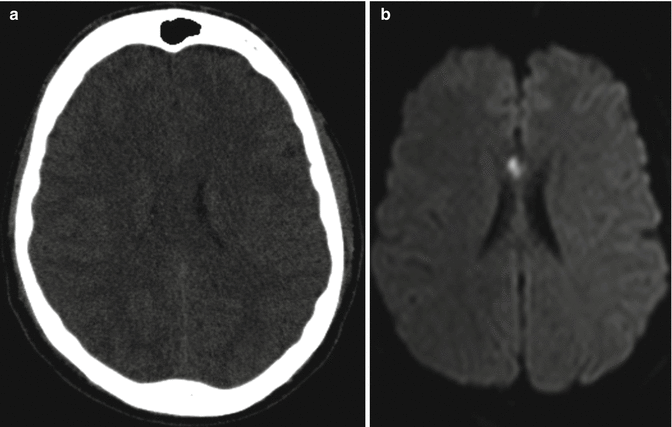

Fig. 3.4
(a) CT scan; (b) DWI on axial plane. Presence of a restricted diffusion focal area located in the right portion of the corpus callosum, not identifiable on CT
Diffusion tensor imaging (DTI) is sensitive to the spatial orientation of water diffusion and allows the virtual reconstruction of axonal networks within the brain by observing restricted water diffusion; in healthy white matter, there is a greater diffusion along the long axis of the axonal bundles rather than the radial ones. The concept behind DTI is that water diffusion in the white matter tracts occurs in one direction rather than randomly, which is called anisotropy. The key indices of DTI are fractional anisotropy (FA) and ADC. FA is sensitive to changes in white matter integrity providing information about the degree of white matter damage; therefore, in the presence of an acute TBI in which a huge damage of white matter occurs, FA values are expected to be very low.
On the other hand, ADC correlates with the average magnitude of water movement in a voxel; so, it increases in the presence of vasogenic edema and decreases with cytotoxic edema and when diffusion is impeded by injured swollen cells [39–41].
Magnetic resonance spectroscopy (MRS) evaluates noninvasively brain chemical environment, revealing posttraumatic neurometabolite abnormalities when conventional neuroimaging is normal. The most common brain metabolites measured are sensitive to hypoxia, neuronal injury, inflammation, and energy dysfunction; they include N-acetylaspartate (NAA), creatine (Cr), choline (Cho), glutamate (Glu), lactate, and myoinositol. Basically, a reduction of NAA (indicating either neuronal loss or neuronal/mitochondrial dysfunction) occurs as a dynamic process after TBI: it remains low in patients with poor prognosis and returns normal in patients with good outcomes.
Cho is a marker for membrane synthesis or repair, inflammation, or demyelination and is typically increased in TBI.
Several studies performed on the brain of injured patients found a great increase in Cho levels and decrease in NAA levels, indicating membrane disruption and increased cell membranes turnover on one hand and a permanent neuro-axonal damage or reversible axonal dysfunction on the other hand, respectively [42–47]. Cr is a marker for intact brain metabolism, and an increased Cr level may be an expression of repair mechanism associated with increased mitochondrial function in areas of injury. After a TBI an alteration of levels of Glu, the brain’s major neurotransmitter, as well as an increase in lactate may be observed.
Cr peak is the most stable index in a wide spectrum of conditions, so allowing a ratio with the other metabolites.
3.3 Extra-axial Injuries
3.3.1 Fractures
A close examination of the scalp should be one of the first steps to perform while interpreting a trauma head CT, since the identification of a soft tissue lesion helps to identify the coup site, which must be carefully inspected to recognize any underlying skull fracture, a soft tissue laceration, and the eventual presence of foreign bodies.
The three main types of fractures are the linear (the most common), depressed, and basal fractures. The linear one is the less commonly associated with parenchymal injuries, although it has a high clinical impact when involving bones close to the vascular structures. Therefore, temporal bone fractures are more prone to injure the middle meningeal artery, with a following arterial epidural hematoma, while occipital ones can damage venous sinuses, with a venous epidural hematoma.
On axial plane they can be easily missed, and a close examination of the “scout” may help to recognize them.
Depressed fractures are more often associated with brain injuries, and they are usually surgically explored to evaluate a potential dural tear, to remove some fragments, and to reduce the risk of infections.
Basal fractures are the most dangerous from a prognostic point of view, since they can be complicated by injuries to the cranial nerves and vessels of the base of the skull (the petrous and cavernous portions of the internal carotid artery (ICA), the transverse and sigmoid sinuses).
Temporal bone fractures can be classified as “longitudinal” versus “transverse” with respect to the long axis of the petrous temporal bone or as “otic capsule sparing” versus “otic capsule involving” fractures. Longitudinal TBF is the most commonly (95%) encountered and is often associated with a fracture extending to the squamosal portion of the temporal bone. It typically spares the otic capsule but may involve the ossicles, resulting in conductive hearing loss. It is usually associated with facial paresis due to the facial nerve injury.
Transverse fracture is much less common and results from an occipital impact. It is more prone to injure the otic capsule, resulting in sensorineural hearing loss and/or facial nerve injury.
3.3.2 Vascular Injuries
Blunt cerebrovascular injuries (BCVIs) include carotid and vertebral artery injuries, occurring in the setting of either generalized multitrauma or direct craniocervical trauma.
Some recent studies report a prevalence ranging from 1.1 to 1.6% among all blunt traumas, while among severely injured patients, the prevalence may be as high as 2.7%. The morbidity and mortality associated with vascular injuries are usually attributed to cerebral or cerebellar infarction. Blunt carotid artery injuries are associated with morbidity and mortality rates of 32%–67% and 17%–38%, respectively. The morbidity rate for blunt vertebral artery injuries is 14–24%, and the mortality rate is 8–18%. Considering only BCVI-specific deaths, the mortality rate for untreated BCVI ranges from 25 to 38% [52–55].
Several studies advocate an early treatment to prevent or limit the infarction development after a vascular injury; however, some authors prefer to adopt a nonoperative management with anticoagulants or antiplatelet therapy. In selected patients the endovascular treatment can be performed [56, 57].
The gold standard examination to diagnose a vascular injury is the four-vessel digital subtraction angiography, which is characterized by some drawbacks, being invasive and associated with some rare complication such as stroke. On the other hand, CT scans with contrast media injection are widely accepted because of its rapidity, safety, and noninvasivity.
The mechanism responsible for cerebrovascular injury seems to be related with the longitudinal stretching of the artery beyond its mechanical limits, with partial or complete loss of mural integrity; vertebral artery injuries occur when spinal subluxation or dislocation stretches the relatively fixed vertebral artery over the adjacent bone structures, or against the dural margins in cases of craniocervical junction injury [58]. Displacement of bone fragments from fractures of the skull base or cervical spine can lead to direct injury to the carotid (Fig. 3.2) or vertebral arteries, respectively. An ICA injury may occur also with a strong hyperflexion of the head or a posterior displacement of mandible fragments, crushing the ICA between the mandible and the spine [59–61] (Fig. 3.5).
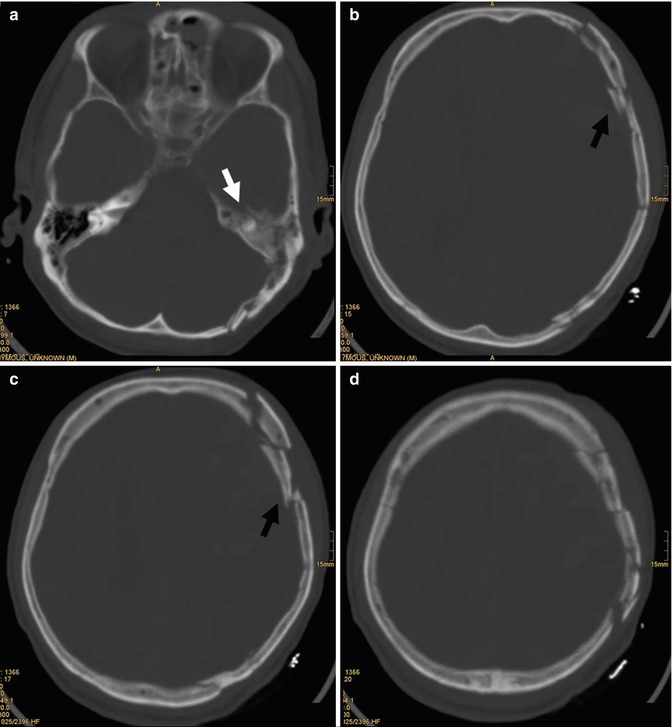

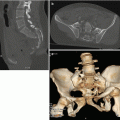
Fig. 3.5
(a–d) CT scan. Presence of comminuted fractures in the left occipital and frontal regions with depressed bone fragments (black arrows); a longitudinal left temporal fracture is associated (white arrow)
In the acute setting, a vascular damage is suspected in the presence of fixed lateralizing or central neurologic deficits that are incongruent with the results of brain imaging or when imaging signs of unexplained cerebral or cerebellar ischemic stroke are present.
Additional clinical signs include active hemorrhage from the mouth, nose, ears, or open wounds, an expanding neck hematoma, cervical bruit in a patient younger than 50 years, transient ischemic attack or amaurosis fugax, pulsatile tinnitus, and Horner syndrome.
Signs and symptoms suggestive of posttraumatic carotid cavernous fistula include orbital pain; pulsatile exophthalmos; chemosis; proptosis; palsies of cranial nerves III, IV, or VI; and blurred vision.
It is a shared opinion that patients with clinical or imaging evidence of severe craniofacial or neck trauma are considered to suffer from brain vascular injury. In fact some studies reported that central skull base fractures and those involving the carotid canal are risk factors for blunt carotid artery injury and that severe facial fractures (Le Fort II and III injuries) and diffuse axonal injury have been identified as independent risk factors for carotid artery injury.
Several studies have identified cervical spine injuries as predictors of blunt vertebral artery injury, especially those involving C1–C3, the transverse foramen or other lateral vertebral elements, and subluxation–dislocation [62]. The so-called Denver criteria group together the most common risk factors for vascular injuries, including Le Fort II or III facial fractures, basilar skull fractures involving the carotid canal, diffuse axonal injury with a Glasgow Coma Scale score under 6, cervical spine fracture patterns that involve C1–C3, cervical spine fracture extending into a transverse foramen, cervical spine subluxation, and near hanging with anoxic brain injury. In addition, a major chest trauma (injury scale score > 3) has also been associated with vascular injuries [63, 64].
The subtypes of traumatic cerebrovascular injury can range from minimal intimal injury, raised intimal flap, separation of the intima from the media resulting in dissection, and medial and adventitial perforation with the consequent pseudoaneurysm to complete vascular transection or occlusion. The minimal intimal injuries appear as areas of luminal irregularity best appreciated on multiplanar or three-dimensional reformatted images.
Raised intimal flaps appear as linear intraluminal filling defects rising from the arterial wall [64, 65].
Dissection with intramural hematoma can either be focal or involve a long arterial segment. The hematoma causes mural thickening which may be eccentric or circumferential. Luminal narrowing may be minimal to nearly occlusive and is usually eccentric [66–70].
ICA dissection occurs more commonly in the cervical and petrous segments of the artery, and it is highly suspected in the presence of a skull base fracture near the carotid canal. Also the distal cervical vertebral artery is very prone to dissection in blunt trauma. An interesting study reported that 35% of patients with a carotid canal fracture were found at angiography to have an ICA dissection, but 40% of the patients with an ICA dissection did not have a carotid canal fracture.
The diagnosis can be performed using CT angiography, MR imaging or MR angiography, and catheter angiography. The advances in multidetector CT have made this technique a first-line tool to assess a neurovascular injury, showing mural irregularity, narrowing, or occlusion. On MR imaging, the most common finding is a crescent hyperintensity (representative of intramural hematoma) around the lumen of the dissected vessel. Unenhanced T1-weighted images using fat saturation technique and proton density and T2-weighted images are particularly sensitive for detecting the intramural hematoma associated with arterial dissection (Fig. 3.6).
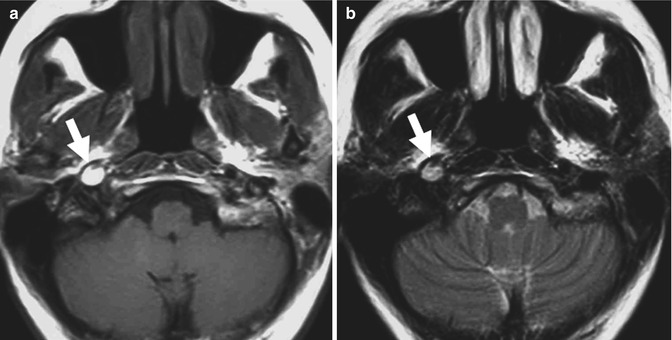
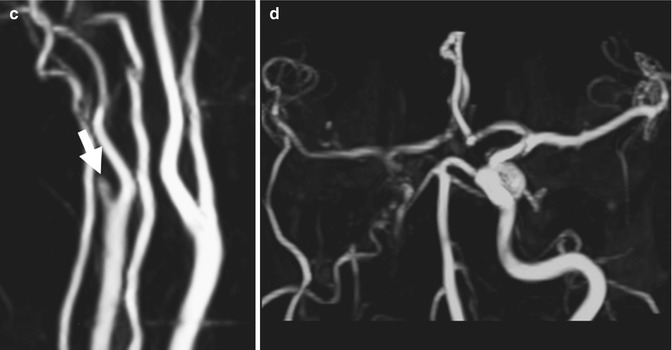


Fig. 3.6
(a) T1-weighted axial section; (b) T2-weighted axial section; (c, d) 3D TOF MR angiography (MIP reconstructions). Internal carotid artery dissection demonstrated by the presence of a hyperintense intramural hematoma with eccentric narrowed lumen (arrows). MR angiography confirms the ICA occlusion after the carotid bifurcation (arrow), with the flow of the terminal branches sustained by the circle of Willis
Vertebral artery dissections are more commonly extracranial in location because of the close proximity to the adjacent bone as the artery courses through the vertebral artery canal or along the sides of the C1–C2 vertebral body complex.
Pseudoaneurysms are characterized by eccentric outpouchings of contrast media from the arterial lumen with an intact adventitia, which provides a little support, being thus more prone to rupture.
On imaging their appearance varies from minimal contour abnormality to large, irregular, and saccular, with a wide neck. At times, a pseudoaneurysm can narrow the native arterial lumen, with potentially significant compromise of arterial flow (Fig. 3.7). At MRI, in the absence of thrombosis or turbulent flow, the pseudoaneurysm appears as a round area of signal void on both T1- and T2-weighted images [64, 71].
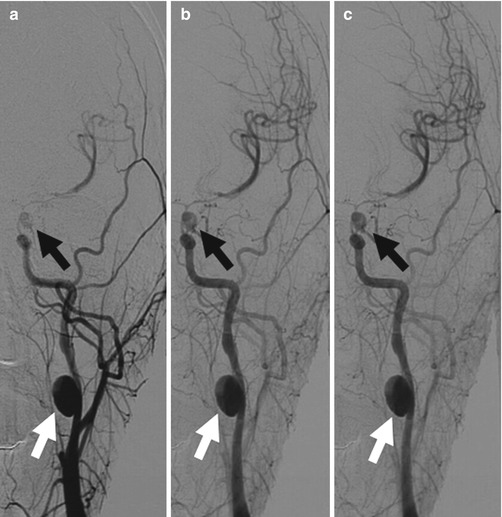

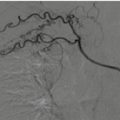
Fig. 3.7
Common carotid artery digital angiography (anteroposterior view). Presence of an extracranial ICA pseudoaneurysm (white arrows), associated with another syphon pseudoaneurysm and narrowing of the arterial lumen, involving the homolateral terminal branches (black arrows)
Active hemorrhage appears as an irregular collection of extravascular contrast material that surrounds the parent vessel, although the exact site of arterial transection may be impossible to identify.
The traumatic arteriovenous fistula (AVF) appears when laceration of an artery occurs adjacent to a vein, resulting in communication between the two systems [72].
The archetypal traumatic arteriovenous fistula is the carotid cavernous fistula (CCF), which results from a laceration of the ICA with direct communication to the cavernous sinus. It usually occurs in the presence of skull base fractures involving the carotid canal. Symptoms often develop in a delayed fashion, days to weeks after the initial trauma; therefore, these injuries can pass misdiagnosed [73–77].
Clinical findings of CCF include proptosis, chemosis, and ophthalmoplegia, with vision loss and facial pain variably present. The cross-sectional imaging findings of a CCF include early venous enhancement during the arterial phase, enlargement of the involved draining veins (i.e., a dilated superior ophthalmic vein-normal diameter < 4 mm), retrobulbar fat stranding, enlarged extraocular muscles, and proptosis, findings that reflect venous engorgement resulting from increased venous outflow from the intracranial circulation.
On conventional catheter angiography, there is an abnormal early filling of the cavernous sinus during the arterial phase and early venous outflow to the superior ophthalmic vein, inferior petrosal sinus, and/or contralateral cavernous sinus [78].
A traumatic AVF can also result from laceration of the middle meningeal artery with fistulous communication to the middle meningeal veins.
In addition to these traumatic AVFs that may present acutely, venous thrombosis in the setting of head trauma can lead to formation of indirect, generally lower flow dural AVFs in a delayed fashion.
3.3.3 Epidural Hematoma
Epidural hematoma (EH) is usually arterial in its origin and represents a hemorrhagic collection within the inner table of the skull and the outer dural layer. It usually results from a skull fracture, often a temporal fracture, which strips away the dura from the inner table, and injures the middle meningeal artery, with consequent filling of the newly formed epidural space. Being the dura tightly adherent to the skull at cranial suture lines, the EH does not typically cross the cranial sutures (with an exception at the level of the sagittal suture, since dura does not cover the suture due to the presence of the superior sagittal sinus).
The majority of cases occur at the coup site with an underlying fracture being identifiable in the 90% of cases.
The loss of consciousness is often immediate, although many patients may experience the so-called lucent interval, during which time the EH expands until it can impact the patient’s level of consciousness.
EH is a neurosurgical emergency requiring immediate evacuation since it causes mass effect symptoms with shift of the midline structures, compression of the lateral ventricle and subfalcine herniation, and sometimes uncal herniation. Small EHs can be managed conservatively.
Venous EHs can also occur, associated with skull fractures, manifesting typically infra-tentorially or within the anterior aspect of the middle cranial fossa, after injuries to the transverse/sigmoid sinuses or the sphenoparietal sinus, respectively; EHs located in the anterior aspect of the middle cranial fossa have a better prognosis.
On non-contrast CT it appears as a well-defined biconvex, hyperdense, extra-axial collection not crossing sutures (Fig. 3.8). Occasionally, an acute EH may contain heterogeneous foci which appear hypodense at imaging (Fig. 3.9), indicating active extravasation of fresh unclotted blood (swirl sign); these lesions have a worse prognosis as they are more prone to further enlargement than the homogeneous EH, requiring thus an immediate surgical management [79–82].
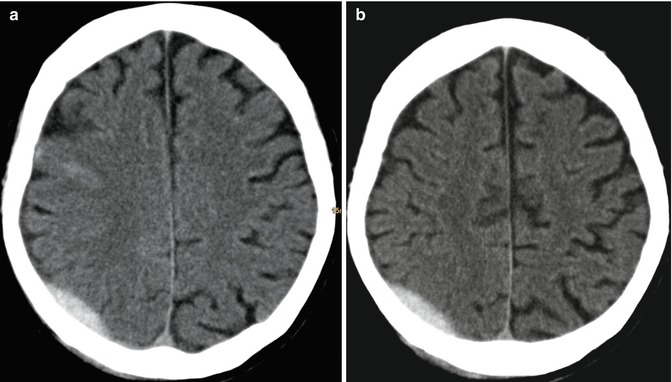
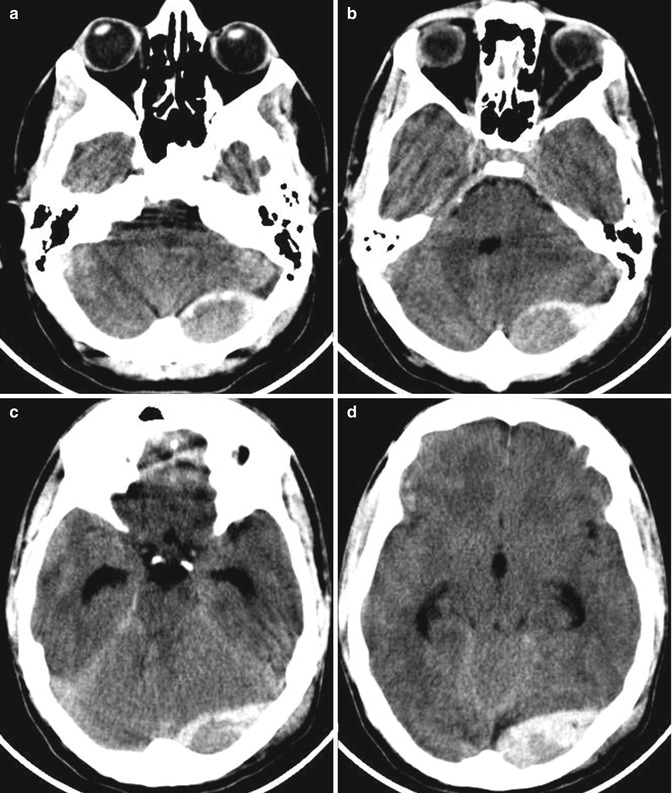

Fig. 3.8
(a, b) CT scan. Epidural hematoma: presence of a well-defined biconvex, hyperdense, extra-axial collection in the right parietal region

Fig. 3.9
(a–d) CT scan. Epidural hematoma of venous origin: presence of a left retrocerebellar biconvex, extra-axial collection, inhomogeneously hyperdense due to the presence of fresh unclotted blood. The fourth ventricle and the aqueduct of Sylvius are compressed, with supratentorial hydrocephalus
Other CT findings associated with an adverse outcome are thickness greater than 1.5 cm, volume greater than 30 mL, location within the lateral aspect of the middle cranial fossa, and midline shift greater than 5 mm.
A rapidly expanding EH can occur also after a decompressive craniectomy, maybe because of the removal of the tamponade effect on a previous extra-axial collection. Some studies suggest performing a postoperative CT scan in patients with a skull fracture remote from the planned craniotomy or a small collection not surgical in nature, remote from the craniectomy.
3.3.4 Subdural Hematoma
Subdural hematoma (SDH) is usually venous in its origin and represents a hemorrhagic collection within the space between the inner meningeal layer of the dura and the arachnoid. Acute SDHs result from a significant head injury causing the disruption of the bridging cortical veins. They can be found both in the coup and in the contrecoup side, the latter one being more common.
They usually occur along the cerebral convexities, the falx cerebri, and the tentorium cerebelli; they can cross the suture lines but not the dural reflections, such as the falx and tentorium.
They can also occur in patients without a history of head trauma, such as elderly patients, those ones on anticoagulant therapy and hemodialysis, and thrombocytopenic patients; thus the symptoms may be very subtle and remain undiscovered for days and weeks until it enters the subacute or chronic stage.
Infratentorial SDHs are rare in adults and are more commonly seen in neonates secondary to perinatal injury [69, 83, 85–88].
They can be divided into three stages, namely, acute, subacute, and chronic, with their own distinct imaging features.
Acute SDH is usually a crescentic, hyperdense extra-axial collection on CT. In some specific conditions, they may be iso- or hypodense due to an underlying anemia or have a mixed density because of active bleeding in the hyperacute setting: in these cases the hypodense areas correspond to the unclotted blood. Alternatively, a preexisting chronic SDH or an arachnoid tear can show a mixed density due to rebleeding, resulting in a mixture of blood with CSF. These “atypical” acute SDHs may be lentiform (biconvex) rather than crescentic in shape, likely secondary to adhesions in the subdural space, as a typical epidural hematoma (Fig. 3.10). Small SDHs may be overlooked on traditional CT “brain windows,” because of the high convexity location, beam hardening artifacts, or narrow window settings; an additional wider “subdural window” (width 200; level 70) should be used in all trauma patients to detect these small SDHs, helping to differentiate between bone and hemorrhage.
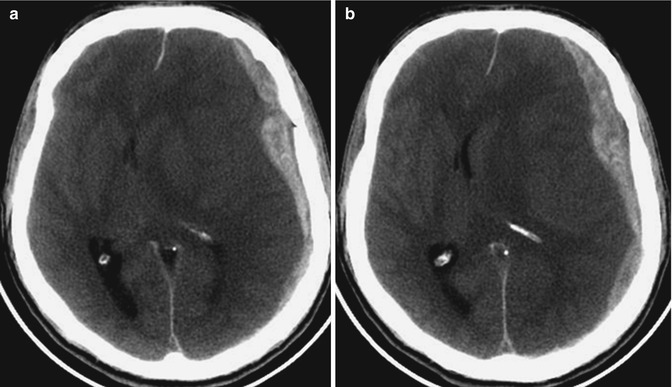

Fig. 3.10
(a, b) CT scan. Acute subdural hematoma: presence of a hyperdense extra-axial collection in the left frontotemporal region, associated with subfalcine herniation; it has a mixed density because of unclotted blood, and a somewhat crescentic/lentiform shape
SDHs occurring at the falx cerebri appear as hyperdense collection between the two hemispheres. The thin SDHs over the tentorium can be very difficult to recognize because of volume averaging effect with the brain, so that thinner axial CT slices, or coronal ones, may help to identify them.
As for as their management, because acute SDHs can rapidly enlarge, they often require surgical intervention; on the contrary small acute SDHs that remain static in size over few days after the trauma are not surgically treated.
Subacute (1–3 weeks) SDHs usually appear isodense to the gray matter on CT, making it difficult to be recognized on CT; possible imaging clues include the so-called abnormally thick cortex, a medial displacement of the gray–white junction from the inner table, white matter buckling, midline shift, compression of the ipsilateral ventricle, and sulcal effacement.
As mentioned before, in the elderly, most SDHs are discovered in the subacute–chronic stage because of the subtle clinical presentation; in particular, brain atrophy seems to play a central role in developing of these lesions, since as the distance between the cortex and the dura increases, small veins extending from the cortex to the dura (the “bridging” veins) become stretched and can be disrupted by violent head motions even in the absence of a high-energy head trauma. SDHs can slowly increase over a few weeks or months, because of repeated small hemorrhages, producing a subdural collection composed of blood products of different stages (Fig. 3.11), often having a thick proteinaceous consistency.
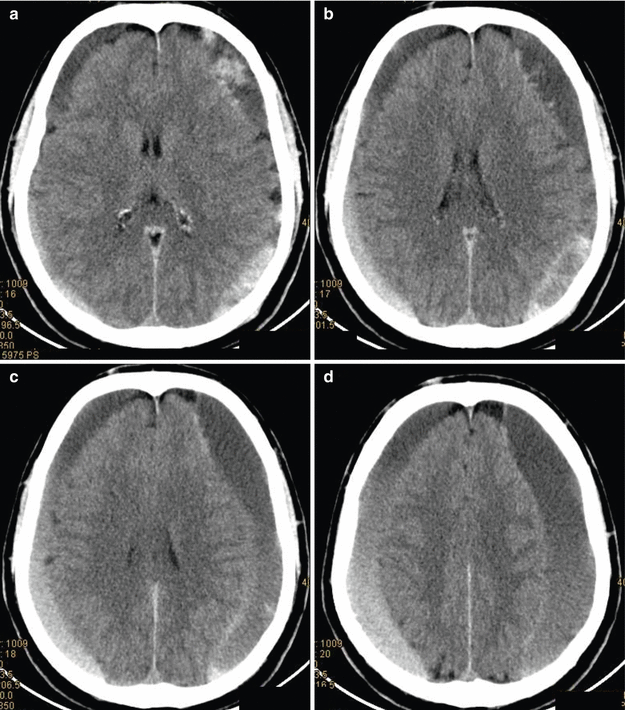

Fig. 3.11
(a–d) CT scan. Subacute–chronic subdural hematomas: bilateral subdural collections composed of blood products in different stages deriving from repeated hemorrhages
Chronic SDHs (>3 weeks) appear as homogeneous hypodense crescentic collection; when a rebleeding occurs, a bilayered appearance may result, with a hypodense layer (the chronic hemorrhage) in the less dependent position and the hyperdense component (the acute hemorrhage) in the dependent position (Fig. 3.12). In this phase multiple adhesions and membranes within the collection can be observed, giving the appearance of multicompartment collection.
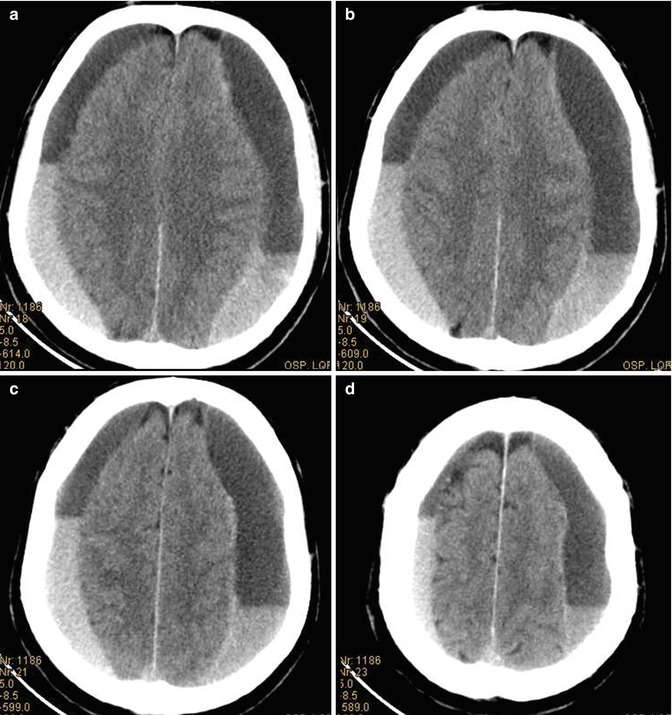

Fig. 3.12
(a–d) CT scan. Chronic subdural hematomas: bilateral bilayered crescentic collections, with a hypodense component (the chronic hemorrhage) in the less dependent position and the hyperdense component (the acute hemorrhage) in the dependent position
MR imaging is more sensitive than CT in the detection of small SDHs and tentorial and interhemispheric SDHs; in addition, MR can easily stage the disease according to the blood signal intensity changes over the time. In the first few days, blood is in the stage of intracellular deoxyhemoglobin; the SDH will be isointense to gray matter on T1-weighted images and hypointense on T2-weighted images. After a few days, being blood in the stage of intracellular methemoglobin, SDHs become hyperintense on T1-weighted images. After the first week, with lysis of red blood cells and production of extracellular methemoglobin, SDHs become hyperintense on both T1- and T2-weighted images, a finding that can persist for many months, associated with hypointense components on T2*-weighted images (Fig. 3.13) [32, 33, 35].
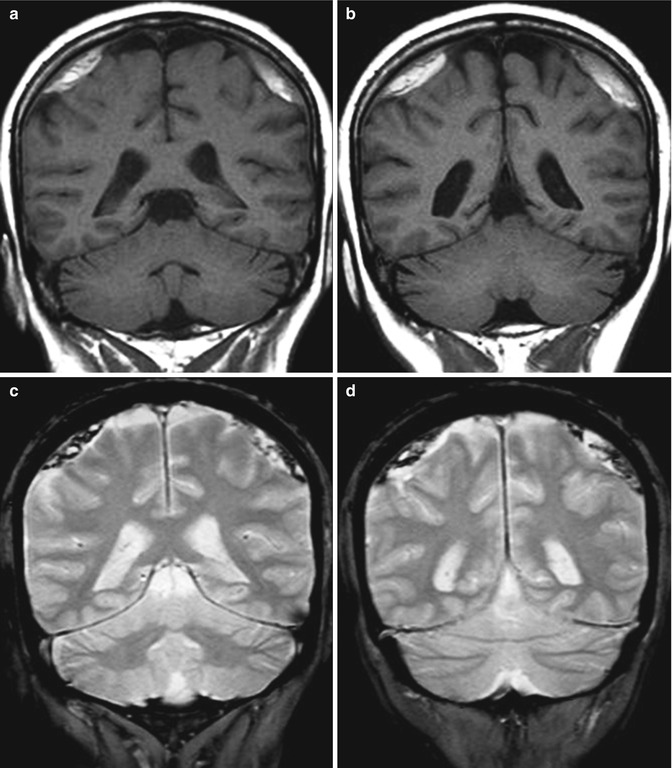

Fig. 3.13
(a, b) T1-weighted coronal sections; (c, d) T2*-weighted coronal sections. Subacute–chronic subdural hematomas: bilateral subdural collections hyperintense on T1-weighted images, with hypointense components on T2*-weighted images
3.3.5 Subarachnoid Hemorrhage
Subarachnoid hemorrhage (SAH) results from injury to the small, bridging, cortical vessels on the pia and arachnoid that cross the subarachnoid space, from extension of interventricular hemorrhage via the fourth ventricular outlet foramina, or from contiguous extension of an intracerebral contusion/hematoma into the subarachnoid space.
The most common locations are the convexity, the fissures (contrecoup Sylvian fissure), and the basal cisterns [69, 89–91].
Aneurysmal SAHs can be differentiated from the traumatic ones basing on the distribution of the hemorrhage [92]; in fact while the aneurysmal SAHs are more centrally located, involving the suprasellar, interpeduncular, and prepontine cisterns, the latter have a peripheral distribution. On CT they appear as hyperdensity within the subarachnoid space (Fig. 3.14); occasionally, effacement of the sulci, secondary to the presence of intrasulcal SAH, can be the only indicator of SAH.
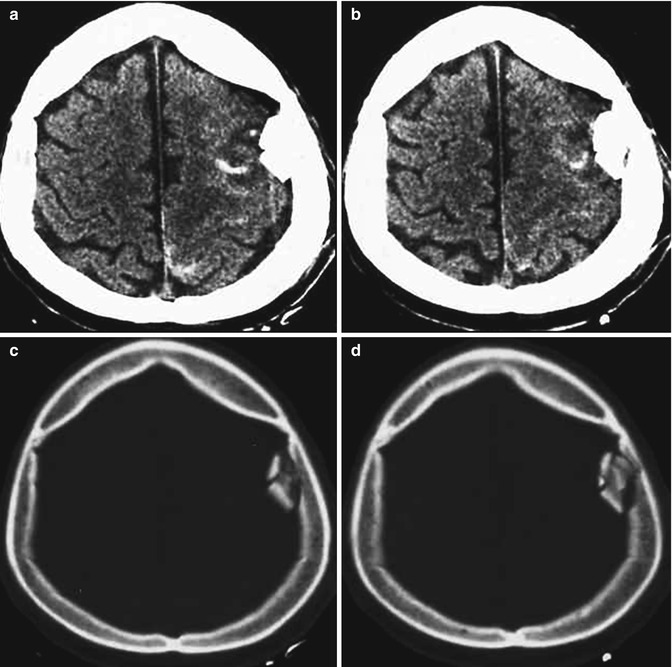

Fig. 3.14




(a–d) CT scan. Subarachnoid hemorrhage: presence of linear hyperdensities within the left frontoparietal subarachnoid spaces, associated with a contiguous depressed fracture
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree