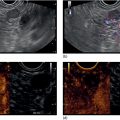
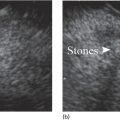
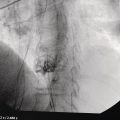
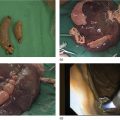
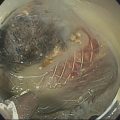
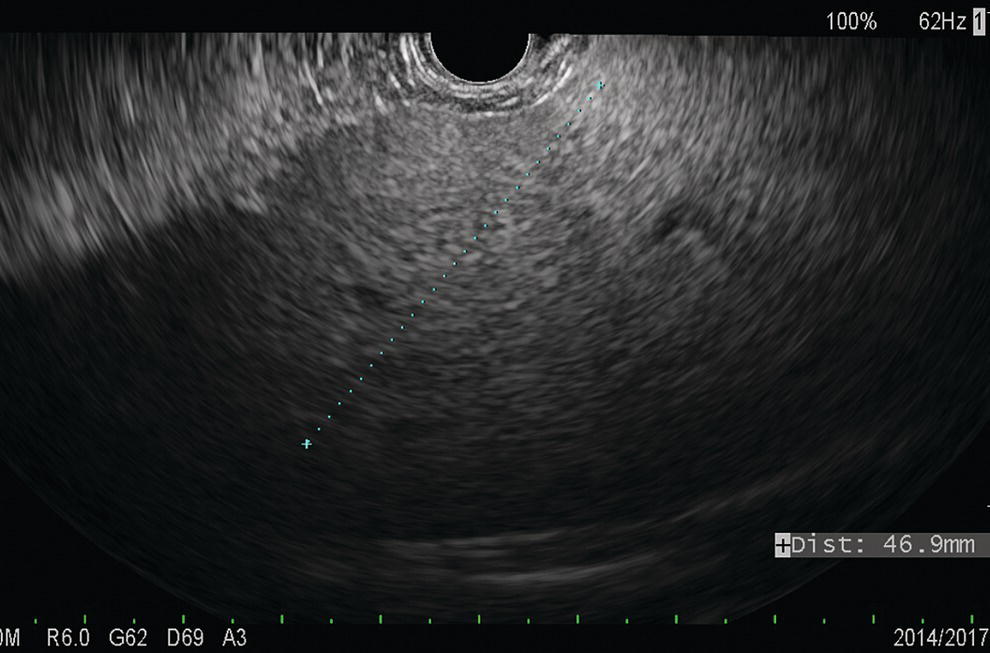
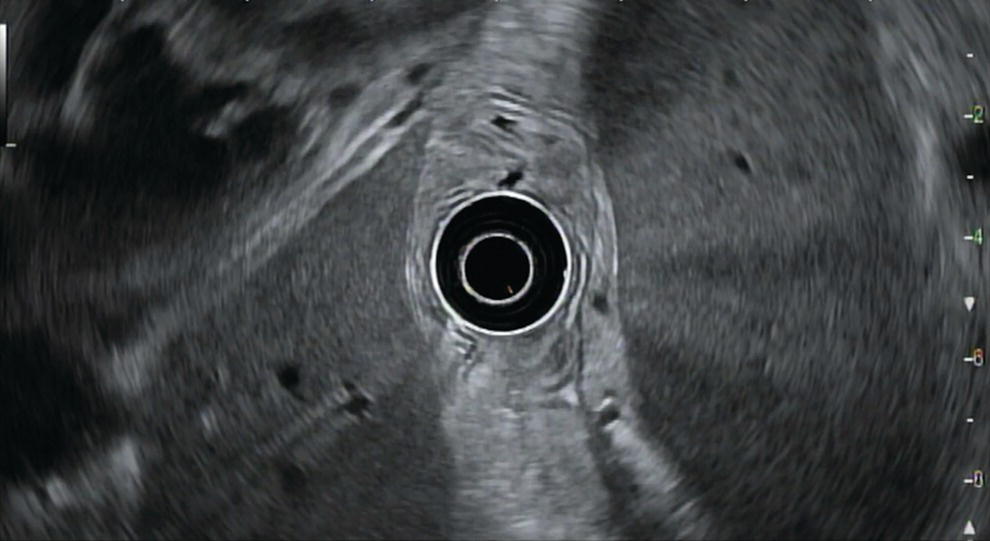
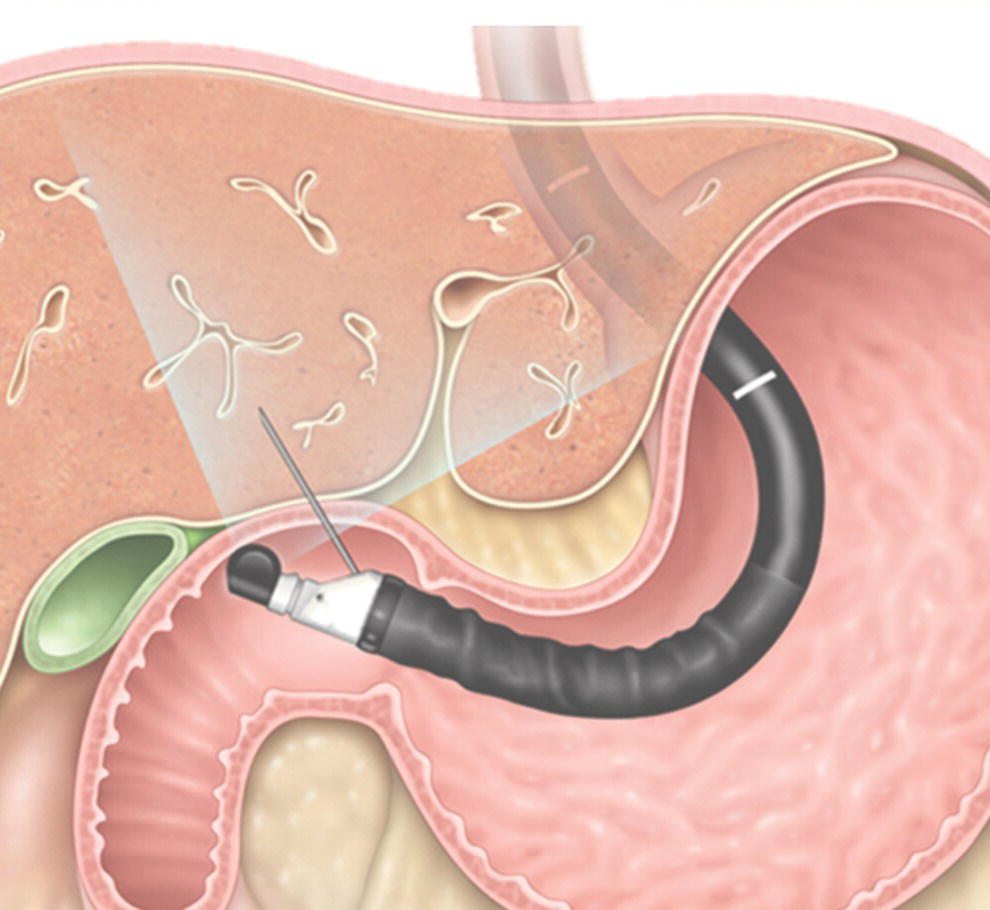
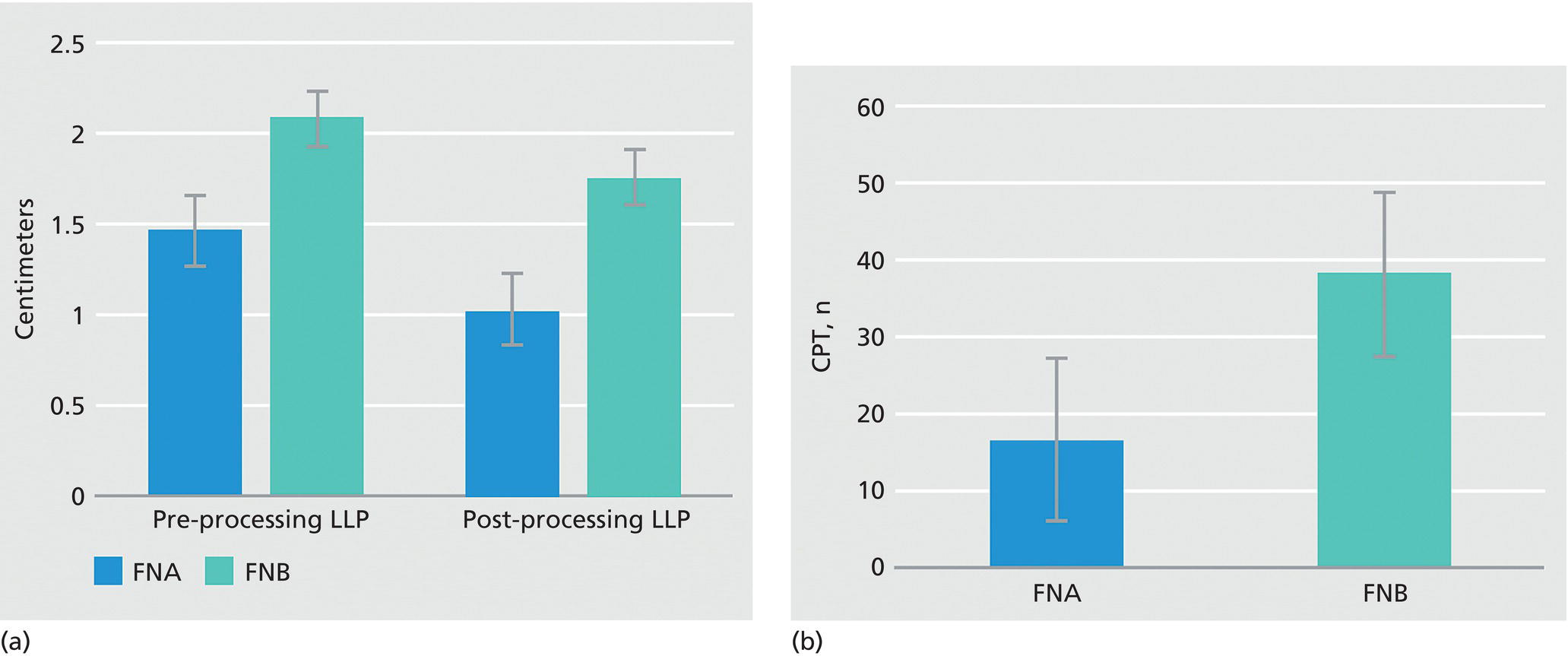
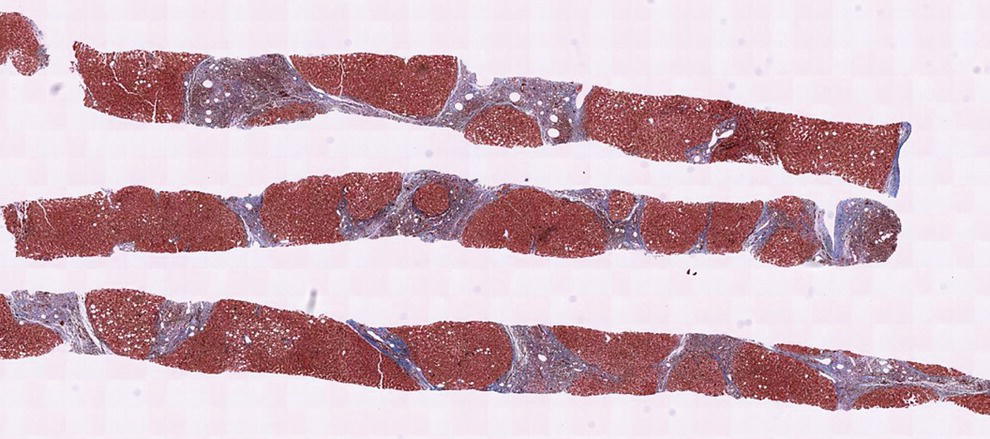
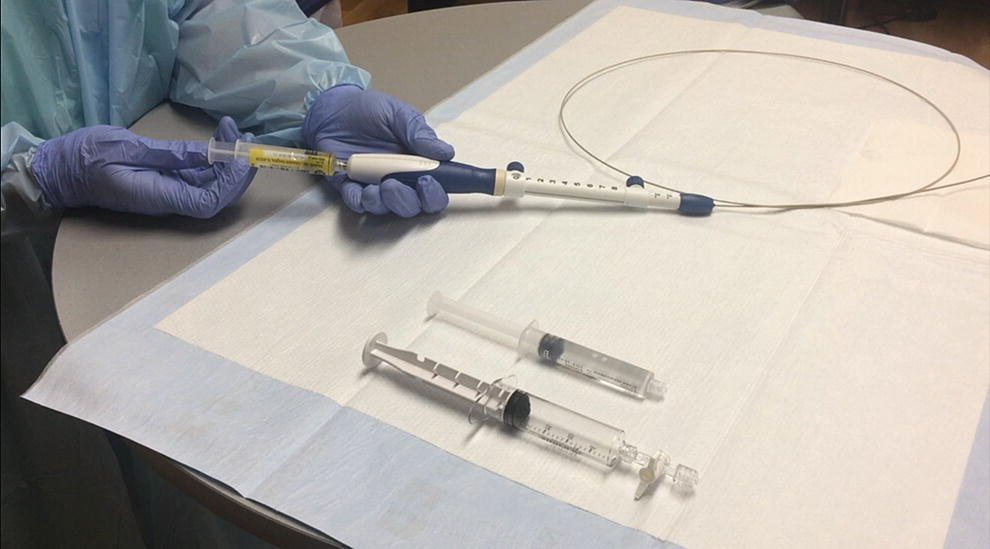
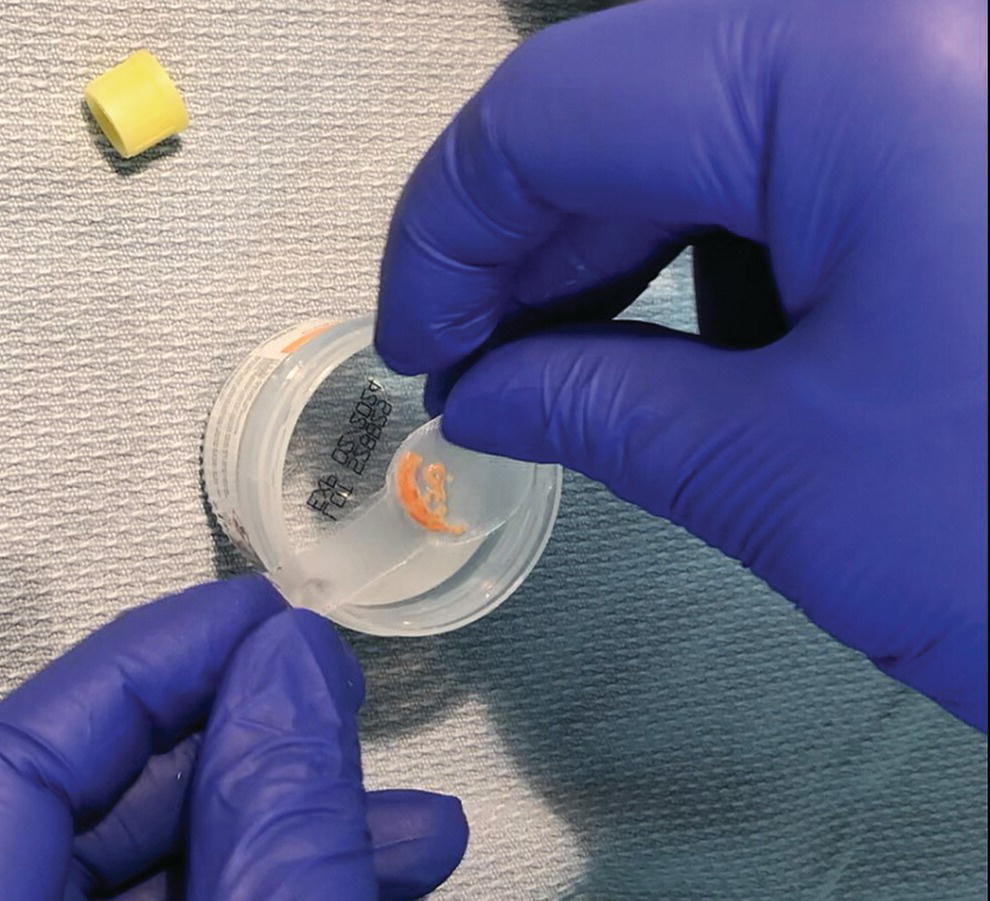
David L. Diehl Geisinger Medical Center and Geisinger Commonwealth School of Medicine, Danville, PA, USA There remains an important role for liver biopsy in the current management of liver diseases despite advancements in non‐invasive hepatic assessment [1–3]. Gastroenterologists are no longer trained in the technique of percutaneous liver biopsy, and this workload has largely gone to interventional radiology. However, use of endoscopic ultrasound‐guided liver biopsy (EUS‐LB) is increasing, and has several advantages. Comparison of tissue yields between percutaneous, transjugular, and EUS‐guided routes has shown comparable tissue yields [4]. Use of newer core needles has further increased the tissue yields for EUS‐LB. Indications for EUS‐LB are broadly any patient who needs a liver biopsy and does not require a transjugular approach. If the patient requires endoscopy (e.g. to exclude varices, Barrett’s surveillance or detection, evaluation of upper gastrointestinal symptoms) or EUS (evaluation of common bile duct, gallbladder, pancreas, or other EUS indication) in addition to a liver biopsy, then EUS‐LB is highly efficient. Some patients require a sedated liver biopsy, for example anxious patients or children [5]. Absolute contraindications include coagulopathy, use of anticoagulants, and obvious cirrhosis, while ascites is a relative contraindication. We require a platelet count of 50 × 109/l or above and an international normalized ratio (INR) of less than 1.5. More recently, thromboelastography (TEG) has been used as a potentially better measure of the interaction of platelets with the entire coagulation cascade, and it is possible that patients with an elevated INR but normal TEG could safely undergo liver biopsy. However, there are no data on this at present. EUS‐LB is performed with the curvilinear echoendoscope, which allows real‐time monitoring of needle entry into the liver. The left hepatic lobe is found by identifying the liver from the proximal stomach (Figure 41.1). It is important to positively identify liver and distinguish it from the spleen, which is found in a similar location (Figure 41.2). In some cases, the echotexture of the liver and spleen can be similar. Confirmation of appropriate targeting of the left lobe of the liver is important to avoid inadvertent splenic puncture [6]. The right hepatic lobe is found by placing the tip of the EUS scope in the duodenal bulb and torquing counterclockwise until the large area of the right lobe is identified (Figure 41.3). For patients after gastric bypass, only the left lobe can be easily sampled. Whether single lobe or bilobar liver biopsy should be done is a matter of debate. Some liver diseases can show heterogeneity in histologic findings between lobes, and bilobar biopsy may be favored to decrease sampling error. For non‐alcoholic steatohepatitis, it has been our practice to sample both lobes [7]. Figure 41.1 Visualization of the left hepatic lobe from the proximal stomach. The cursor shows the expected trajectory of the biopsy needle when obtaining the core specimen. Figure 41.2 View of liver (on the left) and spleen (on the right) from the proximal stomach with the radial echoendoscope. In some cases, the spleen can be very prominent, and care must be taken to positively identify the left lobe of the liver and distinguish it from the spleen. A 19G EUS needle provides the best tissue cores for pathologic interpretation (Figure 41.4). A 19G fine needle biopsy (FNB) needle has been shown to be better than a regular fine needle aspiration (FNA) needle [8] (Figures 41.5 and 41.6). Smaller, 22G FNB needles have been used for EUS‐LB and often provide adequate specimens; however, the diameter of the cores is smaller and specimen fragmentation during processing has been noted [9]. Figure 41.3 Right lobe biopsies are obtained with the echoendoscope tip placed in the duodenal bulb. Source: image used by permission of Boston Scientific. Figure 41.4 Close‐up of the tips of the core needles used for EUS‐LB: (left) SharkCore needle (Medtronic); (right) Acquire needle (Boston Scientific). Source: images used with permission of Boston Scientific and Medtronic. The wet suction technique is superior to dry suction for EUS‐LB [10]. For wet suction, the stylet is removed and the needle lumen primed with saline or heparin flush prior to biopsy (Figure 41.7). The heparin flush decreases clogging of the needle lumen by blood and produces a less bloody tissue specimen [11]. This in turn leads to easier tissue handling by the pathology laboratory. In the chosen lobe, a trajectory for needle passage is identified that does not include larger hepatic or portal vein branches. The longest trajectory of needle travel is sought, which is typically about 3–6 cm. The endoscopy assistant can hold the echoendoscope at the bite block, preventing “recoiling” of the echoendoscope during the needle throw. After a suitable trajectory is defined, the luminal wall is punctured and the needle positioned in the liver parenchyma. The stopcock on the suction syringe is then turned to “on” (full suction). Three to‐and‐fro actuations of the needle are made into the liver [12]. The stopcock on the vacuum syringe is turned off prior to removing the needle from the parenchyma. The needle is then removed from the echoendoscope. The contents of the needle are expressed directly into a formalin cup by either stylet reinsertion or flushing the contents with saline or the heparin flush. Most, if not all, the specimen will be in the needle lumen, although if blood has entered the vacuum syringe, tissue may be found there as well. We are currently utilizing a “tissue sieve” to separate tissue from blood (CoreCatcher; ProAct Ltd, Center Hall, PA) (Figure 41.8). Heparinization of the needle prevents formation of blood clots in the needle, which can be visualized as “blood noodles” in the formalin. When using the tissue filter, the needle contents are expressed first onto the sieve. The specimen is then rinsed gently with saline, which leaves only (or mainly) liver tissue on the sieve. This tissue is then placed into formalin (Figure 41.9). Figure 41.5 Comparison of a 19G FNB needle to 19G FNA needle in terms of (a) length of the longest piece (LLP) before and after histologic processing, and (b) total complete portal triad (CPT) count as shown in a recent prospective randomized trial [8]. The surgical pathology receiving laboratory is typically used to processing small specimens. The endosonographer should discuss EUS‐LB with the pathologist to ensure optimal specimen handling. Excessive handling of the cores should be avoided to limit specimen fragmentation. We use a one‐hour recovery period after EUS‐LB, including for bilobar biopsies. Post‐biopsy abdominal discomfort, if present, is typically easily controlled by a single dose of opiate analgesic (we usually use hydromorphone hydrochloride 0.5–1 mg). Figure 41.6 Excellent core of cirrhotic liver obtained with the 19G FNB needle. Adverse effects after EUS‐LB are uncommon. An adverse effect unique to EUS‐LB is inadvertent splenic puncture [6]. This can occur from misidentification of the left lobe of the liver and confusing it with the spleen (see Figure 41.2). Occasionally, the echotexture of the spleen and left lobe of the liver are remarkably similar. Figure 41.7 Priming the needle with heparin for wet suction technique. Figure 41.8 (a) Use of tissue sieve (CoreCatcher; ProAct Ltd, Center Hall, PA) to collect specimen from biopsy needle. (b) Core sample is washed with saline to remove blood. EUS‐LB is emerging as an important approach for obtaining excellent tissue cores from the liver. It has several advantages over other techniques and obviously is a more “humane” way of performing a liver biopsy. The safety profile is excellent, although inadvertent splenic puncture, potentially leading to severe bleeding, is a complication unique to EUS‐LB. It can be avoided by positive confirmation of the liver as the biopsy target (and not the spleen) prior to the biopsy. Figure 41.9 Long liver core is placed in formalin jar without excessive handling. There are a variety of techniques for accomplishing EUS‐LB, including choice of needles. Use of wet suction provides the best results. Prospective randomized trials have confirmed the superiority of a 19G core needle (FNB) over a 19G FNA needle, and over a 22G FNB needle. However, if the FNB needle is not available, wet suction with the 19G FNA needle is a reasonable option.
41
How to do Endoscopic Ultrasound‐guided Liver Biopsy
Indications and contraindications
EUS‐LB technique
Identification of liver lobes
Needle selection

Needle preparation
Needle technique
Specimen handling
Postprocedure recovery after EUS‐LB
Adverse effects

Conclusions
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree