HYPERSENSITIVITY PNEUMONITIS
Hypersensitivity pneumonitis (HP) represents an immune reaction to inhaled organic antigens. The possible sources of these antigens are diverse and include microbes, animals, plant material, and various chemicals.
There are three possible presentations of HP: acute, subacute, and chronic. Acute HP is rare and involves a large antigen exposure leading to the rapid onset of cough, dyspnea, and fever. Exposure to moldy hay in farmer’s lung is the most typical example of acute HP. Subacute HP is common and demonstrates symptoms that are similar to, but less severe than acute HP. Exposures leading to the development of symptoms are more prolonged, extending over weeks to months. Chronic HP is also common and involves long-term exposure to low levels of antigen over a period of years.
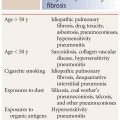
There are striking differences in the prevalence of HP based upon antigen exposure. For instance, up to 15% of pigeon breeders will develop HP. Of note, however, only 50% of patients with HP will have an identifiable exposure. This is one of the major challenges in the diagnosis of HP as most other inhaled diseases, such as pneumoconioses, have an easily identifiable exposure. Smoking is thought to be relatively protective against the development of HP, but it does not preclude this diagnosis.
HRCT Findings
The HRCT findings of HP vary depending upon the clinical presentation.
Acute HP
The findings of acute HP have not been well studied, as most patients are not imaged in this stage. The HRCT findings are likely similar to subacute HP (discussed below), but may be more extensive (Fig. 13.1) and demonstrate an increased incidence of consolidation.
Subacute HP
The diagnosis of subacute HP is usually based on the presence of a combination or constellation of abnormalities, including ground glass opacity (GGO), centrilobular nodules of GGO, mosaic perfusion or air trapping, and the headcheese sign (Table 13.1).
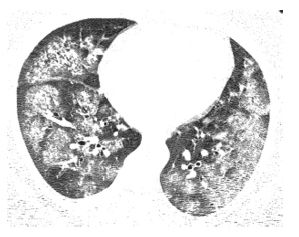
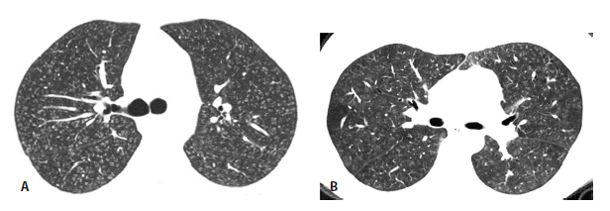
Figure 13.1
Acute hypersensitivity pneumonitis (HP). Extensive bilateral ground glass opacity is seen in a patient with acute HP secondary to mold exposure. These abnormalities are nonspecific and more severe than that typically present with subacute HP.
Patchy GGO
Centrilobular nodules of GGO
Mosaic perfusion and air trapping
Headcheese sign (combination of GGO and mosaic perfusion/air trapping)
Mid–lower lung distribution, spares costophrenic angles
GGO, ground glass opacity.
Ground Glass Opacity
GGO in HP is often patchy and bilateral in distribution. It may be seen in isolation, in which case it is a nonspecific finding (Fig. 13.2). Isolated GGO may be seen with a variety of acute diseases (infections, edema, diffuse alveolar damage, and hemorrhage) and chronic diseases (invasive mucinous adenocarcinoma, interstitial pneumonias, organizing pneumonia, eosinophilic pneumonia, lipoid pneumonia, and alveolar proteinosis). When associated with other findings of subacute HP, such as air trapping, the specificity of GGO for a diagnosis of HP increases.
Centrilobular Nodules of GGO
This is a common finding in subacute HP and reflects peribronchiolar inflammation, cellular infiltration, and poorly marginated granulomas. HP is the most common cause of centrilobular GGO nodules in association with chronic symptoms. The nodules are often diffuse or symmetric (Fig. 13.3). In the setting of an identifiable exposure to an organic antigen, centrilobular GGO nodules are considered diagnostic of subacute HP. The differential diagnosis of centrilobular GGO nodules includes respiratory bronchiolitis, follicular bronchiolitis, atypical infections, and vascular diseases, including pulmonary hemorrhage.
Mosaic Perfusion, Air Trapping, and the Headcheese Sign
Mosaic perfusion and air trapping may be seen in association with other abnormalities (Figs. 13.3B and 13.4) or may be seen as an isolated abnormality (Fig. 13.5). Mosaic perfusion appears as one or more focal areas of decreased lung attenuation associated with reduced size of vessels within the lucent region (see Chapter 5). It reflects the presence of bronchiolitis and bronchiolar obstruction occurring as a result of bronchiolar inflammation. It may persist after treatment of HP and following resolution of GGO or nodules. When seen as an isolated abnormality, the differential diagnosis includes asthma and constrictive bronchiolitis.
The combination of patchy mosaic perfusion and GGO has been termed the headcheese sign; it is highly suggestive of HP (Fig. 13.6). Both GGO and mosaic perfusion must be present. GGO reflects the cellular infiltration typical of HP, while the mosaic perfusion reflects bronchiolar obstruction. It is called the headcheese sign because of its resemblance to a sausage of the same name.
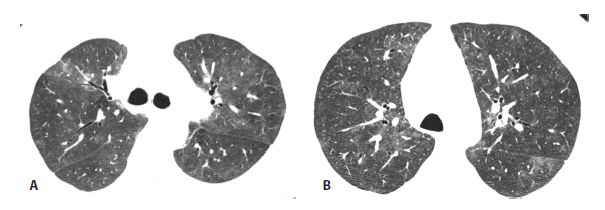
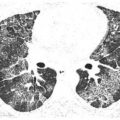
Figure 13.2
Subacute hypersensitivity pneumonitis (HP) with ground glass opacity in two patients. A and B. Patchy ground glass opacity is a nonspecific finding that may be seen in a variety of disorders. It may be the only manifestation of subacute HP, but more commonly is associated with other findings such as mosaic perfusion and centrilobular nodules.
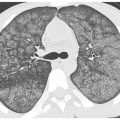
Figure 13.3
Subacute hypersensitivity pneumonitis (HP) with centrilobular ground glass opacity nodules in two patients. A and B. HP is the most common cause of diffuse centrilobular nodules of ground glass opacity. In the setting of an appropriate exposure, such as this patient with a chronic mold exposure, the HRCT should be considered diagnostic of that disease. In the absence of an exposure, biopsy is required for diagnosis. In (B) note that the centrilobular nodules are associated with areas of low-attenuation mosaic perfusion.
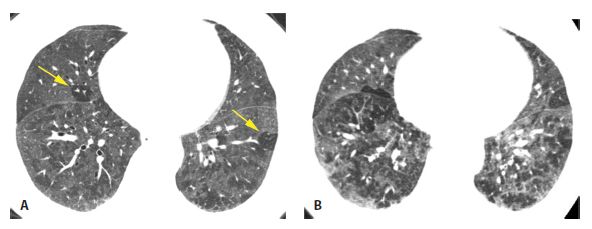
Figure 13.4
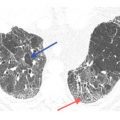
Subacute hypersensitivity pneumonitis (HP) with mosaic perfusion and air trapping. A. HRCT in a 66-year-old bird fancier shows patchy and lobular areas of decreased lung attenuation (arrows) representing mosaic perfusion. Ground glass opacity is also likely present. B. Air trapping within the regions of mosaic perfusion is seen on a dynamic expiratory scan. Mosaic perfusion and/or air trapping may be seen in combination with other abnormalities or as an isolated finding in patients with HP, either at initial presentation or after treatment. Not uncommonly, these are the only abnormalities that persist after treatment of subacute HP.
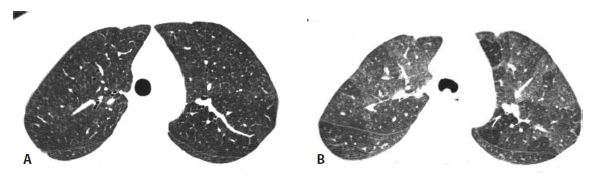
Figure 13.5
Subacute hypersensitivity pneumonitis (HP) with isolated mosaic perfusion and air trapping. A. Other than subtle mosaic perfusion, the inspiratory scan appears nearly normal. B. On expiration, air trapping is clearly seen. This appearance may be seen in patients with subacute HP before or after treatment.
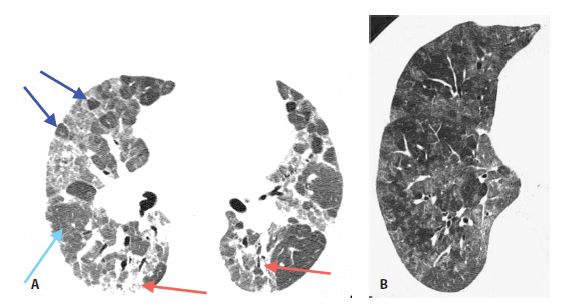
Figure 13.6
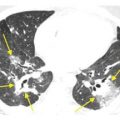
Subacute hypersensitivity pneumonitis (HP) with the “headcheese” sign in two patients. A. The combination of lobular mosaic perfusion (dark blue arrows), geographic areas of ground glass opacity (red arrows), and normal lung (light blue arrow) constitutes the headcheese sign and is highly suggestive of HP. B. In a different patient with HP, geographic areas of mosaic perfusion and ground glass opacity are well demonstrated.
The differential diagnosis of the headcheese sign includes desquamative interstitial pneumonia (DIP)/respiratory bronchiolitis, follicular bronchiolitis/lymphoid interstitial pneumonia, and atypical infections. Of note, mosaic perfusion and air trapping may persist after treatment even when other findings have resolved.
Consolidation
The presence of consolidation is rare in HP. However, it may be the predominant feature in cases in which secondary organizing pneumonia is also present (Fig. 13.7). On HRCT, patchy consolidation in HP may be indistinguishable from cryptogenic organizing pneumonia, eosinophilic pneumonia, invasive mucinous adenocarcinoma, and other causes of chronic consolidation.
Distribution of Abnormalities
In patients with HP, abnormalities typically predominate in the mid-lungs, with sparing of the inferior costophrenic angles (Fig. 13.8). However, an upper lobe distribution is not uncommon. Distribution may help distinguish HP from the interstitial pneumonias (usual interstitial pneumonia [UIP], nonspecific interstitial pneumonia [NSIP], and DIP) that typically predominate in the lung bases, with involvement of the costophrenic angles. Also, in the axial plane, HP is usually diffuse or central in distribution, whereas the interstitial pneumonias are typically peripheral and subpleural.
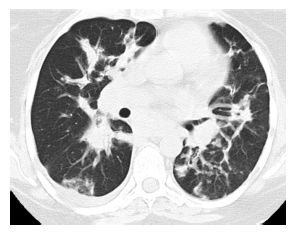
Figure 13.7
Subacute hypersensitivity pneumonitis (HP) with organizing pneumonia. Patchy peribronchovascular consolidation is present in a patient with HP, typical of organizing pneumonia. While organizing pneumonia is commonly seen pathologically in patients with HP, it is only rarely seen as the predominant abnormality on HRCT.
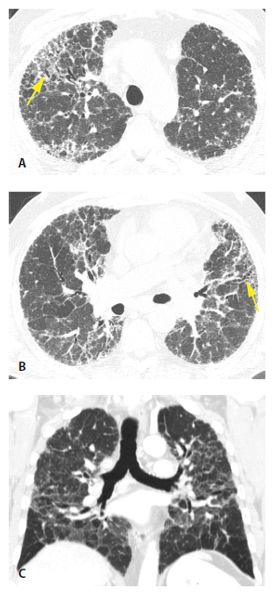
Figure 13.8
Chronic hypersensitivity pneumonitis (HP), distribution. A and B. In a patient with chronic HP with fibrosis, reticulation and traction bronchiectasis (yellow arrows) involve the entire cross section of the lung, both central and subpleural regions. C. Coronal reformatted image shows the mid-lung predominance typical of HP.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree