Key Points
- •
Accurate identification of normal and pathologic lung ultrasound patterns is necessary for appropriate application of diagnostic algorithms.
- •
Misinterpretation of B-lines and Z-lines leads to erroneous diagnosis of interstitial syndromes.
- •
In alveolar consolidation patterns, knowledge of key ancillary findings allows for differentiation between infectious and noninfectious causes.
Background
An examination of discrete points over the thoracic cage allows providers to gather a global, accurate assessment of underlying lung pathology. Interpreting lung ultrasound patterns requires knowledge of unique interactions of ultrasound waves within varying air-fluid compositions in both normal and pathologic states. This chapter reviews the characterization of discrete lung ultrasound patterns that allows immediate clinical application in the management of acutely ill patients.
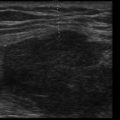
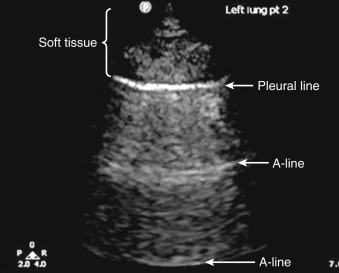
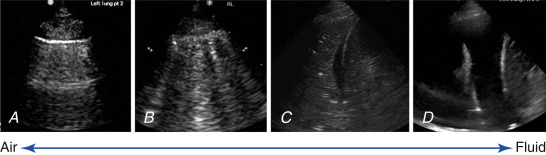
Lungs are predominantly filled with air within a fine architecture of soft tissue lobules surrounding bronchioles. The thin pleural surface is lubricated with a small amount of serous fluid. Air underlying the visceral pleura serves as an acoustic barrier to penetration of ultrasound waves, and fluid facilitates transmission of ultrasound waves. Thus, in normal healthy patients, the pleural surface is readily imaged but underlying lung cannot be seen. At the other extreme, when the lung becomes airless, as in a massive consolidation or atelectasis, it can be visualized like any other solid organ ( Figure 9.1 ). At the opposite extreme, normally aerated lung produces an artifact due to pleural line reverberations called A-lines ( Figure 9.2 ). Between these two extremes, various air/fluid ratios lead to generation of other artifacts, as fluid composition of the lungs varies in different pathologic states ( Figure 9.3 ). Two classification systems, one by Rouby and the other by Lichtenstein, have been described to classify lung ultrasound patterns along the continuum of air/fluid ratios. The nomenclature developed by Lichtenstein is used here ( Table 9.1 ).

| Cause of Respiratory Failure | Ultrasound Pattern |
|---|---|
| Cardiogenic pulmonary edema |
|
| COPD or asthma |
|
| Pulmonary embolism |
|
| Pneumonia |
|
| Pneumothorax |
|
Image Interpretation
A Profile—“A-lines” with “Lung Sliding”
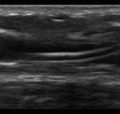
A-lines are pleural line reverberations produced when the subpleural lung is well aerated as in healthy patients or patients with chronic obstructive pulmonary disease (COPD), asthma, or pulmonary emboli ( Figure 9.2 ). In normal patients, the subpleural interlobular septa are so thin that they fall below the resolution of ultrasound; thus the predominance of sub pleural air prevents ultrasound waves from propagating past the pleural line, instead accentuating reflection of the wave back to the transducer. These waves then reflect back and forth between the transducer and the pleural line, causing the reverberation artifact known as A-lines (see Chapter 6 , Figure 6.1 ). A-lines are identical in appearance to the pleural line—horizontal, curvilinear, and occurring at regular intervals deep to the pleural line. The distance between two successive A-lines is the same distance as between the transducer and pleural line.
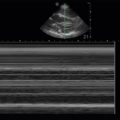
Lung sliding is a dynamic finding seen in healthy lungs when the visceral and parietal pleural surfaces are apposed and the visceral pleura surface moves freely with respirations ( ![]() ).
).
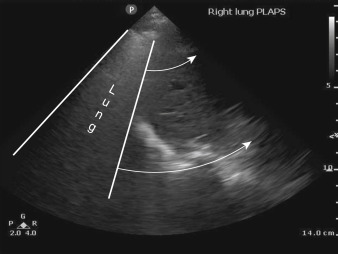
When an A profile is seen at the lung bases, adjacent to the diaphragm, the lung appears as an impenetrable “curtain” that obscures the area where the diaphragm and subdiaphragmatic structures (i.e., liver/spleen) were seen before respiratory descent ( Figure 9.4 and ![]() ).
).
A lung ultrasound exam that reveals an A profile throughout both hemithoraces allows clinicians to conclude that the lung parenchyma contains a normal air/fluid ratio, or is “dry.” This pattern is seen in normal, healthy patients, but when the A profile appears bilaterally in a dyspneic patient, the differential diagnosis is similar to a dyspneic patient with a normal chest radiograph: (1) obstructive airway diseases, such as COPD or asthma, (2) vascular compartment (pulmonary embolism), or (3) nonpulmonary causes (neurologic, neuromuscular, or metabolic causes).
The main utility of a widespread A profile is to effectively rule out presence of pulmonary edema, pneumonia, and pneumothorax. To differentiate between COPD/asthma and pulmonary embolism, the ultrasound approach includes a search for deep venous thrombosis (DVT; see Chapter 26 ). Adding a DVT scan can result in a positive and negative predictive value of 94% and 98%, respectively.
The A′ Profile (“A Prime”)—“A-lines” with “Absence of Lung Sliding”
As discussed previously, lung sliding is caused by independent respiratory movements of the visceral pleural surface when directly apposed against the parietal pleura ( ![]() ). When lung sliding is absent, it is pathologic, and the most worrisome cause is the presence of a pneumothorax (PTX) (
). When lung sliding is absent, it is pathologic, and the most worrisome cause is the presence of a pneumothorax (PTX) ( ![]() ). Unfortunately, PTX is not the only cause of absent lung sliding, given that pleurodesis (chemical pleurodesis, acute infectious or inflammatory states, or fibrotic lung diseases) and lung volume loss (complete atelectasis, mainstem intubation, mucous plug, pneumonectomy) also cause a lack of lung sliding. Thus, absence of lung sliding is not specific to PTX, but presence of lung sliding definitively rules out pneumothorax with 100% specificity at the site of interrogation in a supine patient.
). Unfortunately, PTX is not the only cause of absent lung sliding, given that pleurodesis (chemical pleurodesis, acute infectious or inflammatory states, or fibrotic lung diseases) and lung volume loss (complete atelectasis, mainstem intubation, mucous plug, pneumonectomy) also cause a lack of lung sliding. Thus, absence of lung sliding is not specific to PTX, but presence of lung sliding definitively rules out pneumothorax with 100% specificity at the site of interrogation in a supine patient.
When absence of lung sliding is due to causes other than pneumothorax, the absence of lung sliding is “real”—the visceral pleura is truly not moving because it is adherent to the parietal pleura. However, in pneumothorax, the visceral pleura is moving, but this movement cannot be detected because the air trapped in the pleural space scatters ultrasound waves so the waves do not propagate to reflect movement of the now deeper visceral pleura. Thus, only the immobile parietal pleura is seen, resulting in visualization of absent lung sliding ( ![]() ). Fortunately, there is a sign that is specific to PTX called the lung point . This dynamic sign is caused by an edge of normal aerated lung sliding into view beneath an interspace where lack of lung sliding was initially seen before inspiration. As the visceral pleura expands and slides into the interspace being examined, pleural air from the pneumothorax is pushed away by normal lung and visceral pleura can be seen (
). Fortunately, there is a sign that is specific to PTX called the lung point . This dynamic sign is caused by an edge of normal aerated lung sliding into view beneath an interspace where lack of lung sliding was initially seen before inspiration. As the visceral pleura expands and slides into the interspace being examined, pleural air from the pneumothorax is pushed away by normal lung and visceral pleura can be seen ( ![]() ). It should be noted that lung point is defined as sudden appearance of lung sliding within an area of A-line pattern. This is different from the “curtain sign” where A-lines and sliding lung suddenly obscure the diaphragm, liver, or spleen ( Figure 9.4 and
). It should be noted that lung point is defined as sudden appearance of lung sliding within an area of A-line pattern. This is different from the “curtain sign” where A-lines and sliding lung suddenly obscure the diaphragm, liver, or spleen ( Figure 9.4 and ![]() ). Lung point is 100% specific for PTX, but unfortunately cannot always be localized.
). Lung point is 100% specific for PTX, but unfortunately cannot always be localized.
B Profile—“Multiple B-lines”
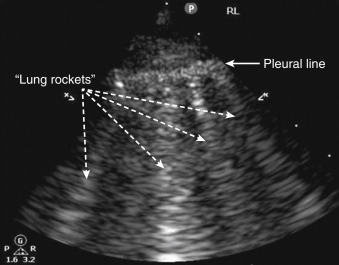
Because normal subpleural interlobular septa fall below the resolution of ultrasound, an A profile results. The utility of lung ultrasound is founded on the fact that parenchymal diseases, such as pulmonary edema, pneumonia, or acute lung injury, cause interlobular septa to widen with fluid accumulation, either due to increased hydrostatic pressure or capillary permeability. These widened, fluid-filled septa now allow propagation of ultrasound waves producing B-lines. ( Figure 9.5 and ![]() ). When B-lines appear acutely, interstitial fluid accumulation is always the cause. Thus, many clinicians have equated B-lines with “wet lung.” However, providers must remember that patients with chronic scarring of the septa from fibrosis, interstitial lung disease, or old infections also have chronically widened septa that create B-lines. Fissures can also produce a single B-line, explaining why isolated comet-tail artifacts are not considered a pathologic finding ( Figure 9.6 ). To be pathologic, three or more B-lines in a single rib interspace must be present ( Figure 9.5 ). Providers must remember that gravity-dependent fluid in the lung bases can widen septa, sometimes producing multiple B-lines in this area in normal patients.
). When B-lines appear acutely, interstitial fluid accumulation is always the cause. Thus, many clinicians have equated B-lines with “wet lung.” However, providers must remember that patients with chronic scarring of the septa from fibrosis, interstitial lung disease, or old infections also have chronically widened septa that create B-lines. Fissures can also produce a single B-line, explaining why isolated comet-tail artifacts are not considered a pathologic finding ( Figure 9.6 ). To be pathologic, three or more B-lines in a single rib interspace must be present ( Figure 9.5 ). Providers must remember that gravity-dependent fluid in the lung bases can widen septa, sometimes producing multiple B-lines in this area in normal patients.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree