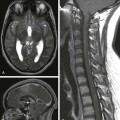
Chapter 30 Adverse events during the neonatal period (the first month of life) account for a large proportion of child deaths and permanent neurologic disability. Preterm neonates are particularly vulnerable to brain injury during the first weeks of life. Imaging has been used widely not only to diagnose and understand brain injury in neonates, but also to predict the neurodevelopmental outcome.1 Because of its portability, cranial ultrasound is usually the first imaging modality to be performed and can be used serially to monitor the evolution of certain injuries. Ultrasound is usually sufficient for evaluation of germinal matrix hemorrhage (GMH) and intraventricular hemorrhage (IVH), hydrocephalus and serial assessment of ventricular size, cystic white matter injury of prematurity, and severe brain malformations. Ultrasound is less sensitive than computed tomography (CT) or magnetic resonance imaging (MRI) in the detection of small calcifications and is less sensitive than MRI in the detection of hypoxic-ischemic injury and subtle brain malformations. MRI also is excellent for the evaluation of punctate white matter lesions, which are seen in the setting of premature white matter. Because of the associated ionizing radiation, the use of CT is mainly restricted to instances in which there is a suspicion of skull fracture or to confirm the presence of intracranial calcifications, lesions that contain fat, and acute intracranial hemorrhage. MRI is the most sophisticated modality to evaluate the neonatal brain, and with advanced imaging techniques such as diffusion-weighted imaging (DWI), functional magnetic resonance imaging (fMRI), and magnetic resonance spectroscopy (MRS), it has the advantage of providing information regarding physiology, function, and metabolism. With the development of arterial spin labeling techniques, it is now possible to assess brain perfusion in the neonate without the use of intravenous contrast material. Dedicated neonatal MR head coils improve image contrast and resolution at the smaller field of view optimal for neonatal imaging.2,3 These dedicated coils improve gray-white matter differentiation and provide better visualization of the brainstem and posterior fossa. Because of the MR environment and length of most MRI examinations, MR safety is a particular concern for neonates. Before the MR examination, any patient, including neonates, should be screened for possible cardiac devices, implants, non–MR-compatible leads, or surgically implanted wires. The compatibility of any device must always be verified with the manufacturer. Additionally, Shellock and Kanal3a provide useful information about the attraction/deflection forces of many items exposed to static magnetic fields. Continuous monitoring and support for respiratory and cardiovascular functions can be achieved with MR-compatible equipment. Thermoregulation, which can be a particular concern for preterm neonates, can be supported through use of MR-compatible incubators and monitored through use of an MR-compatible temperature probe. Hemorrhagic brain injury of prematurity has been classified into four groups. Grade I is hemorrhage confined to the GM; grade II is GMH extending into the ventricles without evidence of ventricular dilatation; grade III is IVH with evidence of ventriculomegaly; and grade IV is IVH with an associated parenchymal infarction, as a result of congestion of the venous outflow (Table 30-1). Grades I and II of IVH have a low morbidity and mortality, whereas grades III and IV have higher mortality rates and a substantial risk of poor neurodevelopmental outcome among survivors (Fig. 30-1). Posthemorrhagic ventricular dilation can be managed with a temporizing neurosurgical procedure, including a ventricular reservoir, a subgaleal shunt, or a ventriculoperitoneal shunt. Table 30-1 Classification of Germinal Matrix Hemorrhage/Intraventricular Hemorrhage Figure 30-1 Ultrasound of germinal matrix hemorrhage. For more than a century, it has been recognized that the developing white matter is exceptionally vulnerable to injury during the second half of fetal gestation (i.e., the period during which preterm infants are born, and in most cases, survive beyond infancy). However, more recently it increasingly has been recognized that injury to the preterm neonate can involve many regions in the central nervous system (CNS), from gray matter (thalamus, cortex, and basal ganglia) to white matter to the brainstem and cerebellum. As a result, it has been argued that more comprehensive terms such as “encephalopathy of prematurity” should be used when referring to injury in the preterm neonate (see Suggested Readings). At the same time, the pattern of injury in preterm neonates has changed during the past several decades in parallel with advances in neonatal intensive care. Historically, cystic periventricular leukomalacia, characterized by multiple areas of cavitary necroses in the periventricular and deep white matter with surrounding astrogliosis, was a frequent observation, particularly among neonates who underwent autopsy. In the modern era, subtle changes in the white matter are more often observed on neuroimaging (e.g., “diffuse excessive high signal intensity”) and at autopsy (e.g., gliosis) with or without accompanying microscopic (1 to 2 mm or less) necroses (visualized on MRI as punctate lesions with high T1-signal intensity in the periventricular and deep white matter).4,5 A low concentration of lactate may be detected in preterm neonates and neonates who are small for gestational age using MRS and is often considered a “normal” finding unless it persists beyond term-equivalency or is associated with other findings (Fig. 30-2). Figure 30-2 White matter injury of prematurity. The central pattern of hypoxic-ischemic encephalopathy (HIE) usually occurs with profound asphyxia, when there is an abrupt interruption of the blood supply, depriving the neonatal brain of oxygen and glucose. Highly metabolic structures such as the thalami, basal ganglia, and brainstem are more vulnerable to hypoxia and ischemia, more specifically the ventral lateral thalami, posterolateral lentiform nuclei, posterior midbrain, hippocampi, lateral geniculate nuclei, and perirolandic cerebral cortex (Fig. 30-3).5 Quadriparesis, choreoathetosis, seizures, mental retardation, and cerebral palsy have been associated with profound asphyxia.6 Ultrasound and CT have low sensitivity to detect early ischemic changes in the deep structures of the brain. The most common pattern on ultrasound is transient or persistent hyperechogenicity, which may progress to cavitation in the basal ganglia and thalami, particularly in the globus pallidus and ventral lateral nuclei of the thalamus. With more severe insults involving the cortex and subcortical white matter, ultrasound and CT may depict indirect evidence of edema, such as effacement of the sulci, loss of gray-white matter differentiation, and compressed lateral ventricles. Figure 30-3 Central hypoxic-ischemic injury. MRI is the imaging modality of choice for neonatal encephalopathy. MRS performed 24 hours after the insult is considered sensitive for hypoxic-ischemic brain injury. Elevated lactate and diminished N-acetyl-aspartate (NAA) are the most common MRS findings in neonates with neurologic and developmental abnormalities (e-Fig. 30-4). Lactate rises after the hypoxic-ischemic event, peaking at 3 to 5 days, whereas NAA starts to decline around the third day. Although a minimally elevated lactate level (lactate/choline ratio <0.15) may be detected in the normal neonatal brain at term, an increased lactate level relative to the total creatine peak in the basal ganglia provides an early indication of brain injury before changes may be apparent on conventional T1- and T2-weighted imaging. It has been suggested that resuscitation may rapidly clear lactate and that a secondary increase in lactate may occur after 12 to 24 hours. The metabolites tend to normalize after about day 5, although in some cases abnormal metabolite ratios persist. Persistently elevated lactate levels in the basal ganglia provide prognostic information about the severity of the brain injury and the subsequent neurodevelopmental outcome. False-negative MRS findings also may occur, with normal spectral findings but abnormal outcomes. e-Figure 30-4 Magnetic resonance spectroscopy (MRS). DWI is a sensitive technique for assessment of acute brain injury. DWI also shows deep gray matter and perirolandic gray matter lesions before they are seen with conventional MRI. If DWI is performed in the first few hours after the injury, it may underestimate the extent of the injury or even show normal results. Paralleling the clinical presentation of HIE, in which neonates may actually transiently improve before demonstrating a more permanent decline in neurologic functioning because of delayed cell death via apoptotic mechanisms, some patients demonstrate mild brain damage for the first few days and then proceed to demonstrate extensive brain involvement around 5 days after the injury. Apparent diffusion coefficient (ADC) values always evolve over time; they decrease initially after the injury, with the nadir around 3 to 5 days, and increase (via facilitated diffusion) later in the chronic phase. As ADC values increase, a point of transient “pseudonormalization” exists in which injured tissue can be misdiagnosed as “normal.” Conventional MRI findings are usually unremarkable during the first few days but then begin to demonstrate first an increased T1-weighted signal and a decreased T2-weighted signal in the subacute period followed by an increased T2-weighted signal in the more chronic period. Evidence suggests that hypothermia therapy delays the onset of MR changes (in metabolic, diffusion, and conventional imaging) associated with injury and, in particular, delays the onset of pseudonormalization in the ADC signal.7 The peripheral pattern of HIE usually results from a period of decreased blood supply to the brain (rather than a near total and abrupt interruption) and is thought to develop as a result of a compensatory shunting of blood to vital brain structures, such as the brainstem, thalami, basal ganglia, hippocampi, and cerebellum, at the expense of less metabolically active structures, namely the cerebral cortex and white matter (Fig. 30-5). Therefore the brainstem, cerebellum, and deep gray matter structures generally are spared from injury in mild to moderate hypoxic-ischemic insults. More prolonged insults result in injury to the intervascular border (watershed) zones, which are relatively hypoperfused as a result of this shunting. Neurologic examination varies depending on the severity of the insult, from asymptomatic in mild cases to proximal extremity weakness or spasticity and cerebral palsy in persons who sustain severe insults.6 Increasing severity of watershed-distribution injury is associated with impaired neurocognitive functioning, including language, visuoperceptual, and executive functioning impairments. Figure 30-5 Peripheral pattern of hypoxic-ischemic injury.
Neonatal Brain Injury
Imaging Techniques
Patient Preparation, Safety, and Hazards
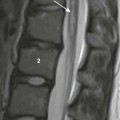
Germinal Matrix and Intraventricular Hemorrhage
Grade
Definition
1
Hemorrhage confined to germinal matrix
2
Intraventricular without ventriculomegaly
3
Intraventricular with ventriculomegaly
4
Parenchymal hemorrhage related to venous infarction (not directly related to grades 1-3)
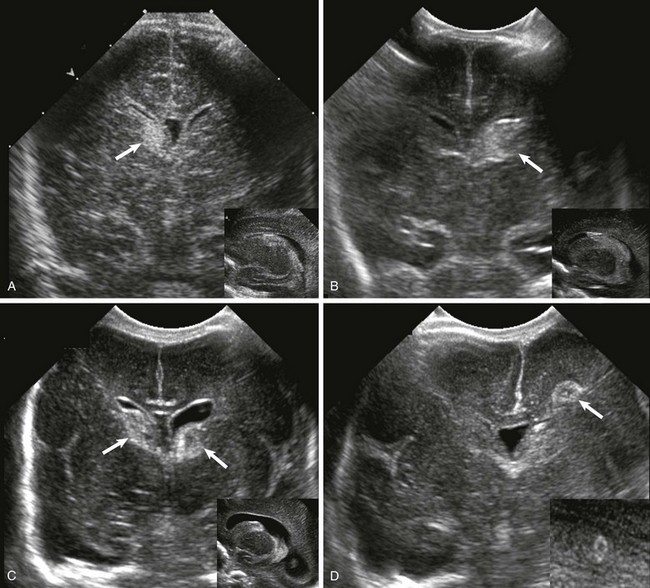
A, Grade 1, hemorrhage confined to the germinal matrix (arrow). B, Grade 2, germinal matrix hemorrhage extending into the ventricles (arrow). C, Grade 3, germinal matrix and intraventricular hemorrhage with ventriculomegaly (arrows). D, Intraparenchymal hemorrhage (arrow).
Premature White Matter Injury
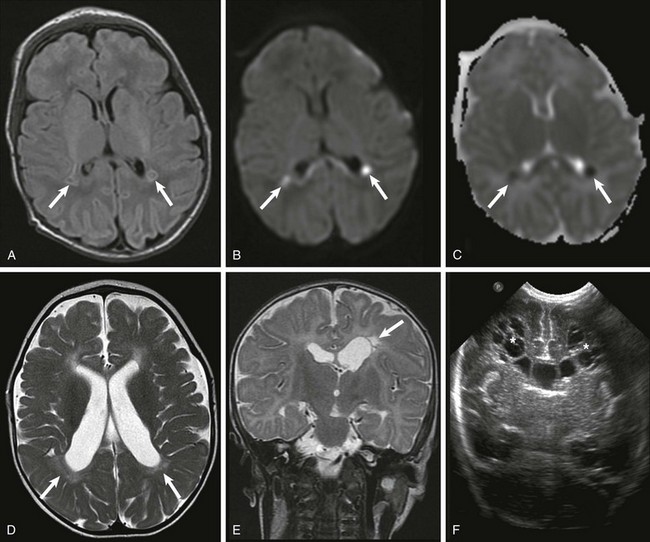
Acutely, focal areas of necrosis with increased signal are seen on T1-weighted images (A), with restricted diffusion (B) and low apparent diffusion coefficient (arrows) (C). D, A different patient at 2 years of age has increased T2 signal and volume loss involving the periventricular white matter bilaterally (arrows). E, A unilateral white matter injury with small cysts (arrow) seen on T2-weighted imaging. F, A bilateral cystic white matter injury (asterisks) on cranial ultrasound.
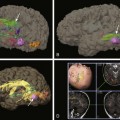
Hypoxic-Ischemic Encephalopathy
Central Pattern of Hypoxic-Ischemic Encephalopathy
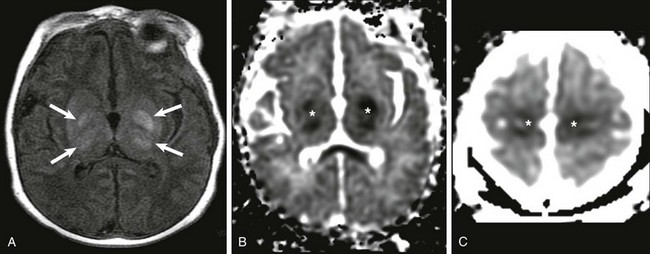
A, Increased T1 signal in the globi pallidi and ventrolateral thalami bilaterally (arrows). B and C, Reduced apparent diffusion coefficient in the globi pallidi and thalami and in the regions of the perirolandic cortex bilaterally (*).
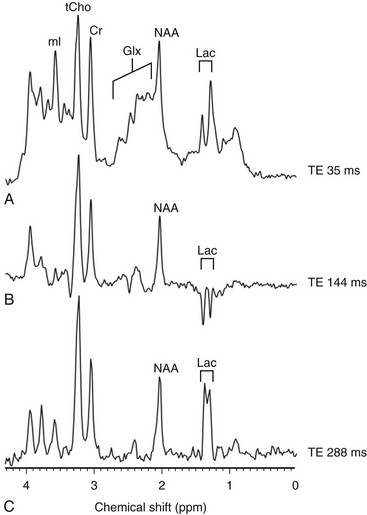
An increased level of lactate is seen on MRS performed with a time echo of 35 ms (A) and 288 ms (C) and inverted with 144 ms (B). Cr, Creatine; Glx, glutamic acid; Lac, lactate; mI, myo-inositol; NAA, N-acetylaspartate; ppm, parts per million; tCho, total choline; TE, echo time.
Peripheral Pattern of Hypoxic-Ischemic Encephalopathy
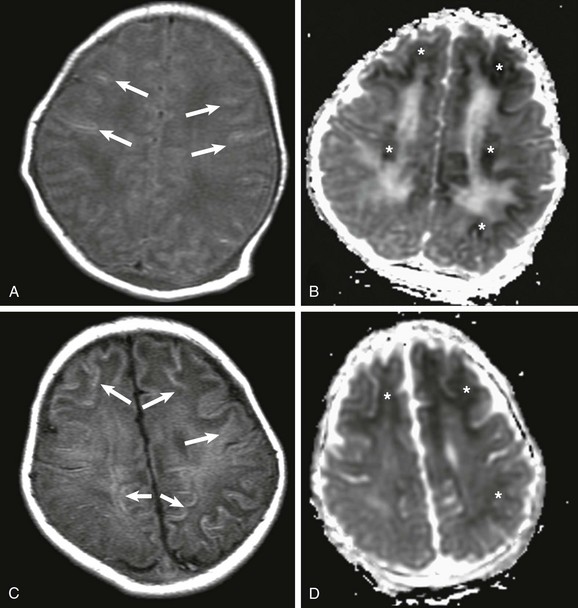
A, At day 2 after the ischemic event, subtle findings are seen on T1-weighted imaging (arrows), but reduced apparent diffusion coefficient (ADC), mainly involving the subcortical white matter, is marked (*) (B). C, At 7 days after the insult, T1-weighted images demonstrate progression on the areas of increased T1 signal, mainly involving the subcortical white matter (arrows), and less conspicuously reduced ADC (*) (D).
Radiology Key
Fastest Radiology Insight Engine