CHAPTER 28 Neuroepithelial Cysts, Porencephaly, and Perivascular Spaces
Many pathologic processes present as cystic lesions of the brain. This chapter focuses on the noninfectious, non-neoplastic cysts of the brain parenchyma and ventricular system in children. These benign fluid spaces most commonly arise in utero, from developmental defects or acquired insults. Infectious cysts, cystic neoplasms, and cystic lesions of the extra-axial spaces are discussed elsewhere in this text.
NEUROEPITHELIAL CYST
These histologically benign, smooth-walled cysts are characterized by a glial wall and an epithelial lining resembling ependyma or choroid plexus (Fig. 28-1). Alternate names include epithelial cyst, ependymal cyst, choroid cyst, choroidal-epithelial cyst, and glioependymal cyst.1
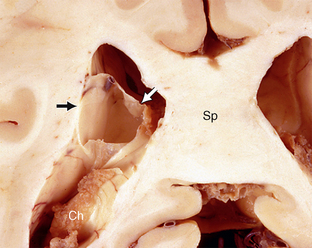
FIGURE 28-1 Neuroepithelial cyst. The smooth, thin-walled cyst (arrows) lies within the atrium of the lateral ventricle superior to and separate from the choroid plexus (Ch). Sp, splenium of the corpus callosum.
(Case courtesy of Dr. John Deck, Toronto.)
Epidemiology
Neuroepithelial cysts are typically first detected in older children but may be found in utero or at birth. There is an uncertain gender ratio, with some series reporting a male predominance2 and others a female predominance.3
Clinical Presentation
Neuroepithelial cysts are usually asymptomatic4 and discovered incidentally at imaging for unrelated conditions. Large cysts may cause symptoms related to mass effect on local structures. Symptomatic neuroepithelial cysts are usually intraventricular and present with obstructive symptoms.
Pathophysiology
Initially, neuroepithelial cysts were thought to arise by evagination and sequestration of ependyma or choroidal epithelium during fetal development. However, these cysts may arise anywhere in the neuraxis, including sites remote from the ependyma and choroid plexus, and they may display diverse immunohistochemical, light, and electron microscopic features. For these reasons, neuroepithelial cysts are now thought to arise from rests of neuroectodermal progenitor cells. The mechanism by which these rests later develop into cysts is unknown. Some of the epithelial cells of the cyst wall do display vacuoles and pinocytic vesicles, suggesting active cellular fluid transport, so the cyst fluid may arise as a secretion of the epithelial cyst wall.1,3
Pathology
Grossly, neuroepithelial cysts are round to ovoid and measure up to several centimeters in size. When large, they may become molded to conform to any rigid adjacent structures. Neuroepithelial cysts typically appear smooth and translucent.4 The cyst fluid is typically clear.
Histologically, the epithelial wall consists of one or more layers of cuboidal or columnar cells that may or may not be ciliated, may or may not contain cytoplasmic vacuoles,3 and may or may not contain a basement membrane.1–3,5–10 The surrounding brain tissue is histologically normal.
Imaging
CT
On CT, neuroepithelial cysts appear as smooth, round to ovoid, nonenhancing lesions with a central low density resembling cerebrospinal fluid (CSF). They may be unilocular or display internal septations. Calcification is very rare.4 Intraparenchymal cysts appear well circumscribed with well-defined cyst margins. Because the cyst wall is typically thin, intraventricular cysts and cysts located within the cisterns may be difficult to distinguish from the surrounding CSF.
MRI
On MRI, the cyst fluid largely resembles CSF on both T1- and T2-weighted (T1W, T2W) images (Fig. 28-2).4 However, the cyst fluid may appear slightly hyperintense to CSF on T1W images, because the cyst fluid has a slightly higher protein content than CSF. The cyst fluid may also appear slightly hyperintense to CSF on T2W images, because the signal from the confined fluid is not dispersed by CSF pulsations (Fig. 28-3). There is little to no restriction of diffusion. The cyst walls do not enhance (Fig. 28-4). The surrounding brain parenchyma displays normal signal intensity with no sign of edema, gliosis, or other reaction to the cyst.
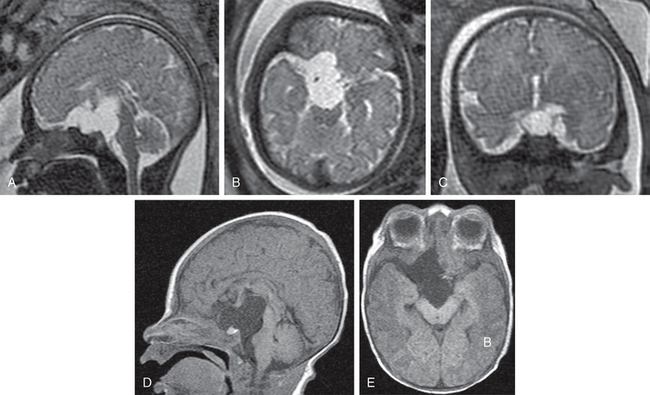
FIGURE 28-2 Pathologically proven neuroepithelial cyst. Fetal maternal T2W MR images in sagittal (A), axial (B), and coronal (C) planes. Postnatal T1W MR images in sagittal (D) and axial (E) planes. The sharply marginated suprasellar cyst shows a uniformly thin cyst wall and homogeneously increased signal intensity just slightly greater than the adjacent CSF. The cyst scallops the skull base including the dorsum sellae, elevates the base of the brain and third ventricle, deviates the basilar artery, midbrain, and pons posterosuperiorly, and extends asymmetrically into the right sylvian fissure. The brain and pituitary gland are otherwise normal.
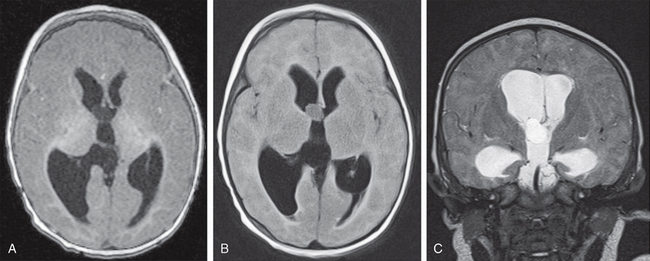
FIGURE 28-3 Neuroepithelial (choroid plexus) cyst, proven. Axial T1W (A), axial T2W FLAIR (B), and coronal T2W (C) MR images show a sharply marginated, thin-walled cyst at the right foramen of Monro. The cyst causes bilateral hydrocephalus, more severe on the right, with mild periventricular edema. The cyst contents have homogeneously increased signal intensity in all three sequences. The signal increase is subtle on T1W images but is readily differentiated from the ventricular CSF on the T2W and T2W FLAIR images (B and C).
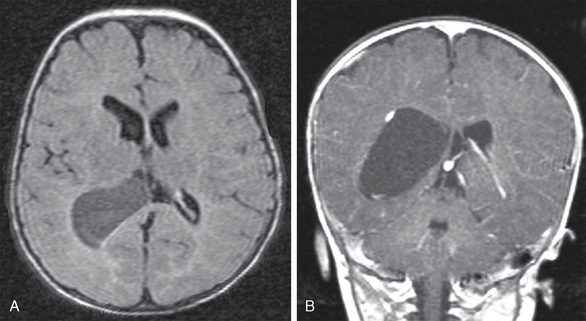
FIGURE 28-4 Neuroepithelial (choroid plexus) cyst. Axial T2W FLAIR (A) and coronal contrast-enhanced T1W (B) MR images show an ovoid, sharply marginated thin-walled homogeneous cyst that expands the atrium and bows the enhancing choroid plexus around its outer wall, but shows no intrinsic contrast enhancement. The brain shows no malformation, destructive lesion, or evidence of parenchymal reaction to the cyst.
Neuroepithelial cysts commonly occur within the choroid fissure (Fig. 28-5). These choroidal fissure cysts may be recognized as CSF-intensity structures situated between the fimbria of the fornix and the diencephalon. They demonstrate a characteristic spindle shape when imaged in the sagittal plane.11 The cyst contents display no restricted diffusion on diffusion-weighted images (see Fig. 28-5).
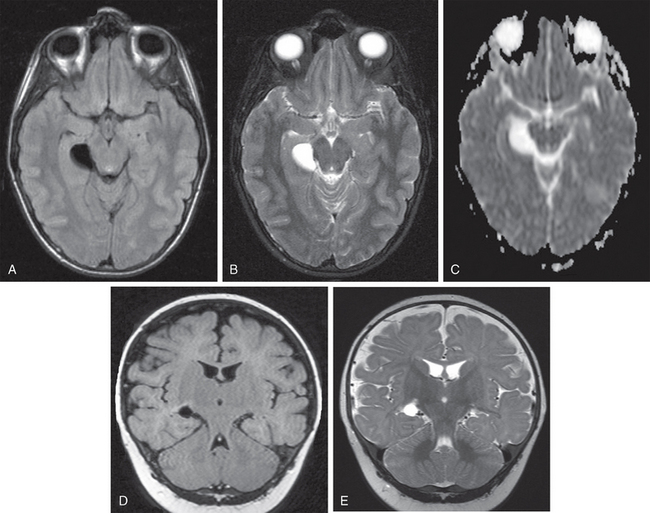
FIGURE 28-5 Pathologically proven neuroepithelial cyst of the choroidal fissure. Axial T1W (A), T2W (B), and apparent diffusion coefficient (ADC) (C) MR images display a sharply marginated lesion with a homogeneous signal that is isointense to CSF on all sequences. The lesion expands the right choroidal fissure, indents the right hippocampal formation laterally, and compresses the right side of the midbrain medially. Coronal T1W (D) and T2W (E) images display a round cyst within the right choroidal fissure. The cyst compresses the hippocampal formation mildly and displaces it inferiorly. There is no edema, gliosis, or other reaction. The right temporal horn is not dilated. No second lesion is evident.
PORENCEPHALY
In the most general definition, porencephaly refers to a CSF-containing cavity that replaces normal brain parenchyma. Heschl coined the term in 1859 (Gr. poros, meaning “opening” or “passage”) to describe full-thickness defects in the cerebral mantle that allowed for communication between the ventricular system and the subarachnoid space. That definition has since been modified to include cysts isolated from one or both of the ventricular and subarachnoid spaces. Loose usage of the term has caused confusion as to its meaning. Some authors classify porencephaly into two types. Porencephaly type I (synonyms: false porencephaly, pseudoporencephaly, and encephaloclastic porencephaly) results from an encephaloclastic insult that occurs in utero, in premature newborns, and in early infancy. The damaged area then evolves over time into a fluid-filled cavity (Fig. 28-6). Porencephaly type II (synonyms: schizencephaly, true porencephaly, and agenetic porencephaly) results from focal injury to the germinal matrix in early fetal life, disrupting the normal migration of subependymal neuroblasts to the surface of the developing hemisphere. This focal disruption creates a focal, often tubular cavity that (1) extends across the full thickness of the brain from the ventricular system to the brain surface and (2) is lined by dysplastic gray matter (Figs. 28-7 and 28-8).
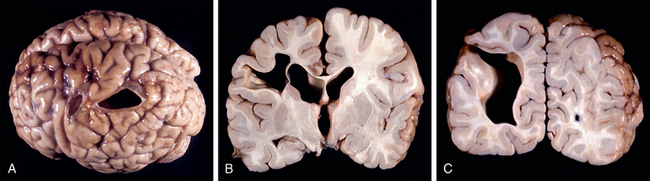
FIGURE 28-6 Porencephaly type I (encephaloclastic porencephaly). Gross pathologic specimens from a 3-year-old boy. A, Superior view of the left parietal lobe. The two cortical defects were originally covered by a pial membrane. There is thinning, discoloration, and secondary change in the adjacent gyri but no cortical malformation. Coronal sections were obtained through the larger cavity. The cyst lies predominantly within the white matter, with sparing of a thin layer of arcuate fibers in one section (B) but extension through the full thickness of white and gray matter in the adjoining section (C). There are no septations within the cyst. The walls of the cyst are lined by white matter. There are no full thickness, transcerebral columns of gray matter lining the cyst cavity. There are no gray matter heterotopias along the adjacent ventricular margins.
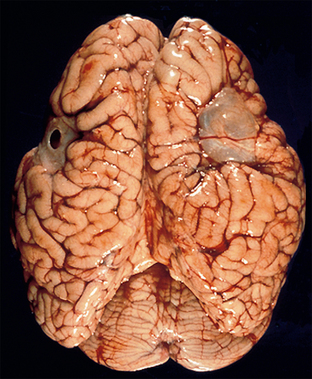
FIGURE 28-7 Porencephaly type II (schizencephaly). Superior view of the unfixed gross brain. Symmetric membrane-covered defects are present in the central regions of both hemispheres. The surrounding cortex appears microgyric. The surrounding sulci converge toward the defect “like rugae toward a stomach ulcer.”
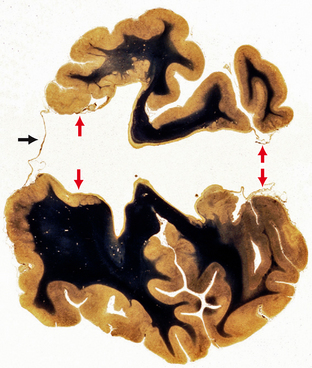
FIGURE 28-8 Porencephaly type II (schizencephaly). Coronal section through bilateral open-lip schizencephaly shows absence of the septum pellucidum, bilateral full-thickness pial-ependymal defects lined by gray matter (two small red arrows at left), the pial-ependymal membrane within the defect (two small arrows at right), and the pial-ependymal membrane (black arrow) that roofs over the schizencephalic defect.
(Image courtesy of the Yakovlev collection, Washington, DC.)
Porencephaly and schizencephaly are closely related. Their differences most probably reflect the different gestational age at which the fetal brain is injured. Injury occurring before approximately 24 weeks of gestation may result in schizencephaly, whereas injury after that time may cause porencephaly.12 The gestational age at which the insult occurs will also determine the reaction in the surrounding brain. More mature brains have a greater capacity to develop a glial scar (gliosis) around the cyst.13 After approximately 30 weeks’ gestation, there is a greater chance of reactive glial scarring.14
Hydranencephaly represents an extreme form of bilateral porencephaly and is due to disruption of the fetal anterior circulation with destruction of the developing cerebral hemispheres. The tissue of each hemisphere is replaced by a large fluid-filled sac covered by a thin membrane representing the leptomeninges. Preservation of the vertebral arteries results in proper development of the brain stem, cerebellum, thalami, and occasionally portions of the temporal and occipital lobes.13 Alternate names include porencephalic cyst and encephaloclastic cyst.
Clinical Presentation
The clinical manifestations of porencephaly depend on the extent of the prenatal encephaloclastic event. The most common presenting signs are hemiparesis and epilepsy.15,16 When the insult is severe, spastic hemiplegia or tetraplegia may be present. There may be developmental delay, especially when the involvement is bilateral. When deeper midline structures are affected, there may be hypoplasia, resulting in visual deficits.17
Pathophysiology
Fetal-maternal ultrasonography and MRI now document the development and evolution of porencephalic cysts in utero. In one case report, for example, the cyst was demonstrated to be the consequence of an intracerebral hemorrhage related to cytomegalovirus infection, with subsequent regression of the hematoma.18
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree