CHAPTER 7 Skull
ANATOMY
The skull consists of multiple flat bones joined together by sutures. These flat bones have rounded margins that permit the formation of a vault that is located superior to the skull base. The skull surrounds the outer surface of the brain, whereas the skull base supports and covers the undersurface of the brain.
The skull vault comprises the neurocranium, which is bordered anteriorly by the facial bones or viscerocranium.1 Multiple bones contribute to the formation of the skull vault. These include, in anterior to posterior direction, the frontal bone, the greater wing of the sphenoid bone, the frontal process of the zygoma, the squamous portion of the temporal bone, the parietal bone, and the occipital bone. These bones are connected by fibrous sutural membranes that initially allow for the expansion of the developing brain in early life. Growth of the brain and adjacent skull are also facilitated by the presence of anterior and posterior fontanelles. The anterior fontanelle is located between the frontal and parietal bones at the junction of the sagittal and coronal sutures. The posterior fontanelle is located between the parietal and occipital bones at the junction of the sagittal and lambdoid sutures.
Each of the skull bones is made up of an outer table of cortical bone, a middle table or diploë that contains bone marrow, and an inner table of cortical bone.2 Periosteum covers the outer table of the skull. This periosteum is actually considered to be part of the deepest layer of the scalp. The periosteum is fairly adherent to the bony cortex and is tightly adherent at sutural junctions between adjacent skull bones. The inner table is lined by the dura mater, the most superficial layer of the meninges. Periosteal dura is located immediately adjacent to the inner table cortex. A second layer of dura, the meningeal dura, is apposed to the periosteal dura except where these two dural leaves diverge to form the major dural venous sinuses, the sagittal and transverse sinuses, and the falx cerebri and cerebelli and tentorium cerebelli. Like the periosteum of the outer table, the periosteal dura is tightly adherent to the inner table at the cranial sutures. Despite this close apposition of these fibrous connective tissue structures, the periosteum and the periosteal dura, to their respective cortical layers, a potential space can be found beneath the periosteum, the subperiosteal space, and above or external to the periosteal dura, the epidural space. The subdural space is located deep to or beneath the meningeal layer of dura.
IMAGING
Special Procedures
Catheter angiography can be used to further characterize suspected vascular lesions within or about the skull. The arterial supply to the lesion, when present, can be defined as can the venous drainage. This can be helpful in preoperative planning. Presurgical embolization of hypervascular lesions can facilitate resection and minimize intraoperative blood loss.
HOW A PATHOLOGIC PROCESS ALTERS NORMAL APPEARANCE
Skull lesions present either as clinically palpable masses or as lesions that are incidentally detected during an imaging study obtained for some other clinical indication.3,4 Skull lesions originate within the calvaria proper. Lesions that arise from the scalp or meninges can invade the skull and rarely can simulate primary skull lesions. Furthermore, certain pathologic entities such as those related to trauma, infection, and inflammation can involve multiple contiguous structures and compartments. For the purposes of this chapter, this discussion will focus on those lesions that either arise directly from the skull or those lesions that are typically associated with the calvaria.
There are several criteria that can be used to analyze skull lesions. The first of these is the patient’s age. Certain masses, such as developmental lesions, tend to present in early childhood and young children whereas other lesions tend to present in adults (Table 7-1). While the approach to image analysis in adults remains the same, the frequency and types of lesions that are encountered with the skull vault are somewhat different. With respect to developmental lesions, in certain instances, the location of the skull lesion may predispose toward a specific diagnosis. It is extremely important to analyze the skull for the presence, or absence, of more than one skull lesion. The presence of multiple skull lesions usually indicates the presence of a multifocal neoplastic process in adults such as metastatic disease or multiple myeloma. In children, on the other hand, lesion multiplicity may not only reflect the presence of metastatic disease but also be due to an inflammatory process such as Langerhans cell histiocytosis. The next criterion that is used in lesion analysis is an assessment of a given lesion’s margins. A lesion with well-defined, sharp margins and evidence of reactive bone formation on CT or plain radiographs is more likely a benign entity because these features suggest a nonaggressive growth pattern. Lesions that possess ill-defined, irregular margins reflect the presence of a more aggressive process such as that which is seen in a neoplasm. The final parameter that should be included in the evaluation of a skull lesion is the assessment of the lesion matrix. The lesion may be characterized as lytic, sclerotic, or mixed on CT or plain radiographs. Lesion contents such as fat, CSF, or bone, identified with CT or MRI, may suggest a specific diagnosis. The absence of contrast enhancement may indicate the presence of a less aggressive lesion such as a developmental lesion.
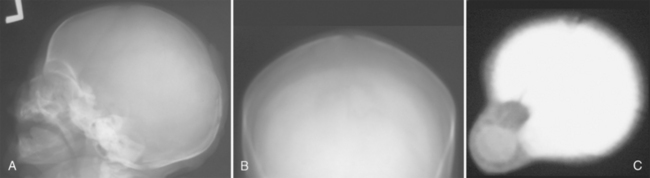
The majority of congenital skull lesions are detected in the pediatric age group (see Table 7-1). Cephaloceles often present at birth as a skin-covered protrusion of variable amounts and types of intracranial contents through a defect in the skull.5,6 They have an incidence of 1 to 3 per 10,000 births. Two thirds of cephaloceles occur within the skull vault and involve the occipital region (Fig. 7-1). In addition to presenting as a palpable mass, the clinical presentation will depend on the extent and severity of associated central nervous system (CNS) anomalies (Fig. 7-2). The latter can be fairly well characterized by MRI, as can the cephalocele contents and the relationship to the superior sagittal sinus (Fig. 7-3). The skull defect can be defined with CT especially using 2D and 3D reformatted images. Dermoids and epidermoids are ectodermal inclusions that can involve the skull.7 Dermoids are often found in a midline location adjacent to the anterior fontanelle. Because of the presence of different ectodermal derivatives such as sweat and sebaceous glands, hair follicles, and teeth, these lesions can demonstrate a variable imaging appearance with certain features predominating within a given modality (Figs. 7-4 to 7-6). Plain radiography and CT are helpful in identifying any tooth-like structures or calcifications. The relationship to the midline as well as the outward beveled margins of the skull can best be seen with CT using multiplanar reformatted images. A focal skull defect may indicate the presence of a dermal sinus tract; the latter may serve as route for the spread of infection (Fig. 7-7). The MR signal characteristics will depend on the cyst contents; if a dermoid contains lipid, hyperintense signal may be seen on T1W images. Epidermoids are usually located within the lateral aspect of the skull vault and present as expansile intradiploic cystic masses. The wall of the epidermoid is lined by stratified squamous epithelium but is rarely seen on imaging because it rarely calcifies and usually does not enhance after intravenous contrast agent administration. On CT, an epidermoid presents as a well-circumscribed hypodense intradiploic expansile mass with smooth, sclerotic margins. The cyst contents show low to intermediate signal intensity on T1W MR images and increased signal intensity on T2W MR images (Fig. 7-8). A few of the congenital lesions that are usually seen in children may occasionally be encountered in adults (Fig. 7-9). These include dermoids and epidermoids where the imaging presentation is similar to that seen with children. Arachnoid cysts are not uncommon in adults, and whereas the majority of these cysts are intracranial extra-axial lesions they sometimes can present as an intradiploic lesion. Arachnoid cysts are intracranial cysts that can rarely present as skull involvement. In the latter situation, the cyst enlarges and gradually erodes, thins, and deforms the adjacent portion of the inner table through CSF pulsations (Fig. 7-10). These are large simple CSF-containing cysts that do not enhance and are readily diagnosed with CT and MRI.8 These arachnoid cysts can be differentiated from parietal thinning, a normal developmental variant of the parietal bones that can present as a palpable skull deformity and may involve either one side or both sides of the skull vault (Figs. 7-11 to 7-13).9
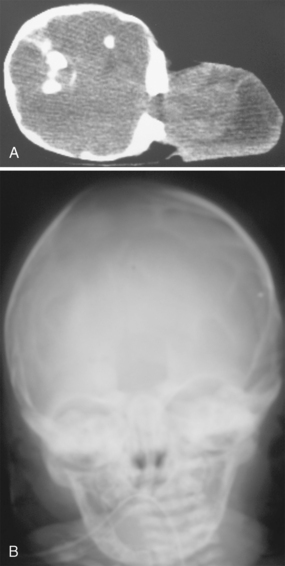
FIGURE 7-1 Cephalocele: female neonate with occipital mass. A, Unenhanced axial decubitus CT image shows a large occipital soft tissue mass that includes cerebral tissue that has herniated through a large defect in the occipital bone. B, Frontal skull radiograph shows the well-marginated osseous defect within the skull vault.
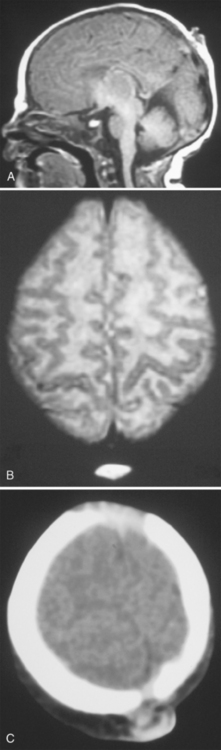
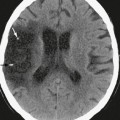
FIGURE 7-2 Meningocele: 3-day-old male neonate with palpable mass. A, T1W midline sagittal MR image shows a small hypointense mass within the parietal scalp. B, T2W axial MR image shows hyperintense contents. C, Unenhanced axial CT image shows a small somewhat hypodense soft tissue mass adjacent to the sagittal suture.
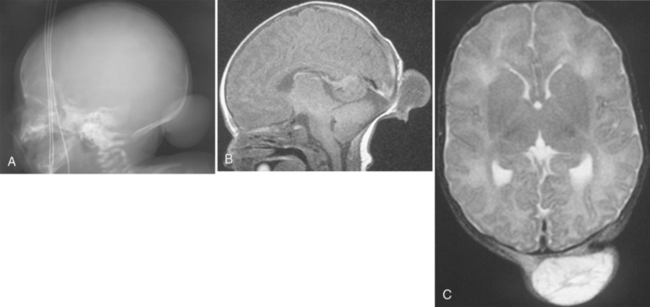
FIGURE 7-3 Cephalocele: 1-day-old female neonate with occipital mass. A, Lateral skull radiograph shows the mass. B, T1W axial MR image shows a predominantly hypointense mass; the sagittal sinus is in normal location. C, T2W axial MR image shows increased signal intensity within the herniated sac. Normal flow void is seen within the sagittal sinus, which, in this case, is separate from the lesion.
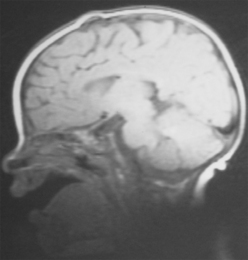
FIGURE 7-4 Dermoid: 4-month-old female infant with palpable anterior fontanelle mass. T1W midline sagittal MR image shows a small hyperintense mass just underneath the scalp.
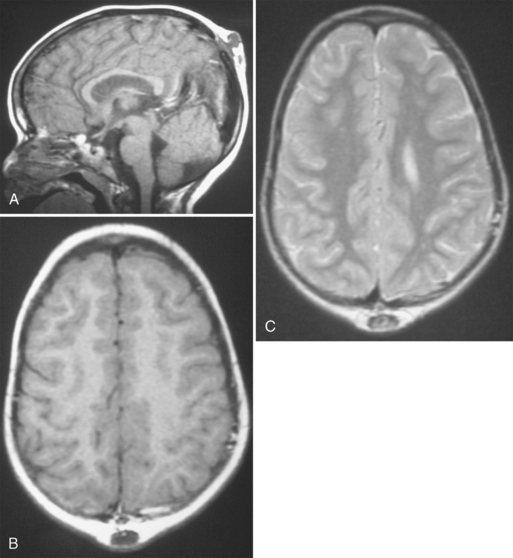
FIGURE 7-5 Dermoid: 5-year-old girl with incidental parietal mass detected during MRI evaluation for possible seizures. A, T1W midline sagittal MR image shows a hypointense mass that communicates via a stalk or tract with a small midline defect in the calvaria. B, The T1W axial MR image confirms the extracranial location of this hypointense mass. C, The lesion is heterogeneously hyperintense on the T2W MR image and does not communicate with the superior sagittal sinus.