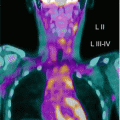
Fig. 10.1
Female, 3 months. MAG3 diuretic renography (protocol F+2). No washout even after micturition. Typical ascending pattern of the right renographic curve. Obstruction of the right pyelo-ureteral junction
Furosemide (1 mg/kg iv) is administered intravenously, and time of injection should be standardized in the single laboratory, to ensure repeatability. The “classic” protocol (F+20: furosemide injection 20 min after radiopharmaceutical administration) and the “fast” protocol (F0: furosemide injection immediately after radiopharmaceutical administration) are the most widespread options. In both cases the acquisition has to be continued for at least 20 min after the diuretic stimulus, and it is mandatory to acquire a post-micturition image of 1–2 min, after keeping the patient upright for a couple of minutes (Fig. 10.2).
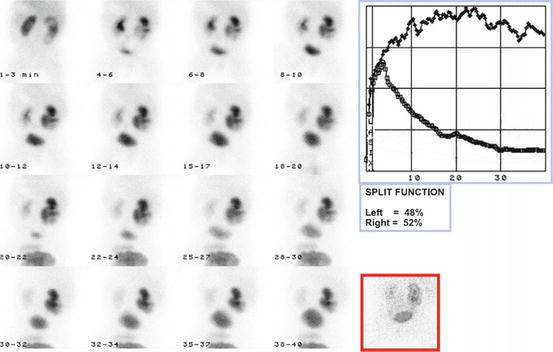

Fig. 10.2
Male, 7 months. MAG3 diuretic renography (protocol F+2). Slow washout during the dynamic phase. Good washout after voiding. Indeterminate pattern of the right renographic curve. Preserved differential renal function in the right kidney. No indication for surgery
10.3.1.4 Processing
Images are summed for a better evaluation of morphology and parenchymal uptake. Regions of interest (ROI) are drawn on both kidneys and on the surrounding tissue (background) to produce background-subtracted time/activity curves and for calculation of differential renal function, expressed as percent of the renal uptake between 60 and 120 s after injection for each kidney (normal range 45–55 %). The estimation of absolute clearance should be based on blood sampling, because gamma-camera methods are plagued by many sources of error.
Excretion must be evaluated taking into account post-micturition images because the pattern of the renogram is not reliable for a diagnosis of obstruction, even if typical obstructed and nonobstructed patterns are usually diagnostic
Many parameters have been described for the quantitative analysis of the excretion phase; output efficiency (OE) and normalized residual activity (NORA) probably offer the best balance between robustness, reproducibility, and practicability.
10.3.2 Static Renal Scintigraphy
10.3.2.1 Radiopharmaceuticals
99mTechnetium-dimercaptosuccinic acid (DMSA) is the best cortical imaging agent available at present, presenting a very high renal uptake (45 % of the injected dose) and low urinary excretion. Therefore, the visualization of the renal cortex, the binding site of the tracer, is excellent, since the interfering activity in the pelvi-calyceal system is negligible after 2–3 h from the administration.
10.3.2.2 Patient Preparation
No special preparation is required for DMSA intravenous injection, but it is important to wait at least 2 h after injection and it is recommended to increase the waiting time to 3 h whenever possible, to increase the quality of the acquired images.
10.3.2.3 Acquisition
The child should lie on the collimator surface, using Velcro strap and/or sandbags to maintain the required position. The mandatory views are posterior, left, and right posterior oblique. The image should be acquired with a 256 × 256 matrix (recommended minimum 128 × 128) using a zoom factor adequate to the body size (usually between 1.5 and 2, depending on the size of the detector). Counts should be at least 300,000/image when using a parallel-hole collimator or between 100,000 and 150,000 for pin-hole collimators.
There is no consensus on the use of single photon emission tomography (SPET). The higher image contrast could improve sensitivity for smaller cortical defects, but it is prone to false-positive results and in most cases requires a sedation, which is not necessary for planar DMSA scanning.
10.3.2.4 Processing
Images are evaluated preferably on a monochrome gray scale and the ROI-based differential renal function, expressed as percentage between the two kidneys is calculated, using the geometric mean of the counts in posterior and anterior projection. It is possible to omit the counts form the anterior projection when the kidneys have normal morphology and dimensions and they are in normal position.
10.3.3 Cystoscintigraphy
Radionuclide cystography is the most sensitive method to evaluate for vesicoureteral reflux and results in significantly less radiation exposure when compared with conventional radiographic technique, but it cannot provide anatomical information (e.g., on the bladder or urethra).
Direct radionuclide cystography (DRC) requires instillation of tracer (usually 99mTc-pertechnetate, 18–40 MBq) through a bladder catheter allowing imaging during filling, voiding, and after voiding.
Indirect radionuclide cystography does not require catheterization, since it is performed at the end of a dynamic renal scintigraphy, which requires an intravenous injection of radio tracer. Moreover, the patient has to be able to control a voluntary micturition.
Sterile urethral catheterization should be performed with a Foley catheter or a feeding tube. The latter is often the preferred option, particularly in infants.
Similarly to the other nuclear medicine procedures, neither preparation nor sedation is needed, but in many centers, the antimicrobial prophylaxis is considered mandatory, to reduce the risk for catheter-induced infections.
The patient is positioned whenever possible on the collimator, taking every precaution (plastic-lined absorbent paper) to prevent any contamination of the detector.
The bladder volume can be estimated using a formula (e.g., (age in years + 2) × 30 ml), and the corresponding volume of saline (gently warmed to 37 C) is instilled at slow rate (10 ml/min) to avoid a too fast stretching of the bladder wall. The height of bottle should be less than 80 cm from the table. The radiotracer is injected in the catheter as a small bolus at the beginning of the infusion
10.3.3.1 Acquisition
Dynamic images are acquired in the posterior projection (matrix 128 × 128) including kidneys, ureters and bladder in the field of view. During the filling phase the frame rate is 5–10 s/frame and the acquisition usually ends when the calculated volume has been instilled or the infusion stops due to back pressure. In cooperative patients the acquisition is usually interrupted by the necessity to voiding.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree