Fig. 1
Biocontinuum of adverse early and late effects of the ear (with permission from Rubin and Casarett 1968)
Otologic structures are seldom primarily affected by malignancies. Malignancies may extend into the ear from the overlying skin or anteriorly and caudally from the parotid. Nasopharyngeal tumors may extend into the parapharyngeal space with extrinsic eustachian tube obstruction or tubal invasion. The resulting functional impairment to the eustachian tube commonly leads to the development of otitis media with effusion (OME) (Sato et al. 1988). RT-induced complications in head-and-neck cancers and VS commonly involve a range of otologic dysfunction.
RT-associated ear morbidity may be both acute and delayed and vary in degree from mild to severe. In the external ear, these morbidities range from acute otitis externa to external auditory canal stenosis (van Hasselt and Gibb 1999). Eustachian tube dysfunction, OME, and conductive hearing loss (CHL) are the most common middle ear complications (van Hasselt and Gibb 1999; Elwany 1985). Thickening of the tympanic membrane, tympanosclerosis, and perforation has also been reported (Elwany 1985). Higher doses of radiation may cause middle ear fibrosis or ossicular atrophy (Gyorkey and Pollock 1960). Morbidities associated with the inner ear include tinnitus, vertigo or imbalance, and sensorineural hearing loss (SNHL). Hearing loss and neurological deficits are the most significant of the RT-induced morbidities (Borsanyi and Blanchard 1962). Biocontinuum of adverse early and late effects is shown in Fig. 1.
2 Anatomy and Histology
2.1 Anatomy
The ear’s sensory system is primarily comprised of the auditory and vestibular systems. Anatomically and functionally, the ear is divided into three distinct regions: the external, middle, and inner ear. All three regions are involved in hearing function. Only the inner ear functions in the vestibular system (Fig. 2).
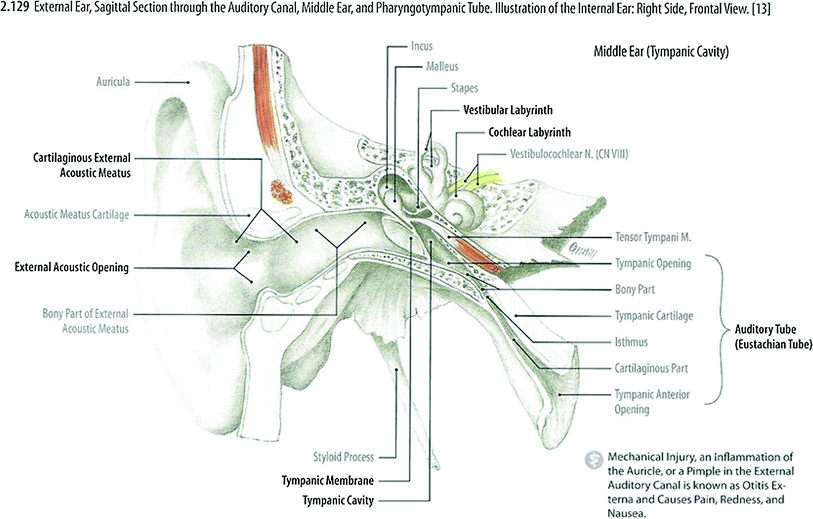

Fig. 2
External ear, sagittal section through the auditory canal, middle ear, and pharyngotympanic tube. Illustration of the internal ear: Right Side, Frontal View (with permissions from Tillman 2007)
2.1.1 External (Outer) Ear
The outer ear includes the visible part commonly known as the auricle or pinna and a slightly curved short external auditory canal that extends inward from the floor of the deepest depression of the auricle, called the concha, and ends at the tympanic membrane. The auricle consists of a thin plate of cartilage and overlying skin that forms an irregular, shallow funnel that is continuous medially with the external auditory meatus. The external auditory meatus is an S-shaped canal that extends about 1.25 inches. The outer third wall of the external auditory canal is composed of cartilage, while the inner two-thirds are formed mostly by the tympanic portion of the temporal bone. The isthmus is the narrowest point of the external auditory canal and is situated just medial to the bony-cartilaginous junction. The inferior wall of the external auditory meatus is about 5 mm longer than the superior wall, conferring obliquity to the tympanic membrane. The entire length of the passage is lined with skin, which also covers the outer surface of the tympanic membrane. The outer cartilaginous portion is covered by thicker skin with fine hairs directed outward and lined with secretory glands that produce cerumen. The ear canal is bound superiorly by the middle cranial fossa, anteriorly by the temporomandibular joint and parotid, medially by the tympanic membrane, posteriorly by the mastoid (a confluence of the squamous and petrous portions of the temporal bone), and inferiorly by the skull base and soft tissues of the neck. The tympanic membrane is a thin, semitransparent oval, and obliquely placed membrane that separates the external and middle ear. The long process of the malleus is attached to the tympanic membrane, resulting in concavity toward the external auditory meatus (Fig. 2).
2.1.2 Middle Ear
The middle ear is an air-filled cavity in the petrous portion of the temporal bone located directly internal to the tympanic membrane. It is connected anteriorly to the nasopharynx via the eustachian tube and posteriorly with mastoid air cells via the mastoid antrum. The labyrinth of the inner ear forms much of the internal surface of the middle ear, with the basal turn of the cochlea representing the bulk of the surface directly medial to the tympanic membrane (a.k.a., the mesotympanum). The carotid artery passes through the medial face of the junction of the middle ear and eustachian tube (a.k.a., the protympanum). A complex air-cell system surrounds the labyrinth, connecting the middle ear to the petrous apex. The middle ear is lined by respiratory mucosa and contains three bones (malleus, incus, and stapes) and two muscles (tensor tympani and stapedius). The facial nerve passes from the internal auditory canal, over the cochlea, around the stapes, and caudal to the lateral semicircular canal en route to the stylomastoid foramen at the skull base. In the process, it emanates branches, most importantly, the greater superficial petrosal and chorda tympani nerves. Primary sensory innervations to the middle ear travel by way of Jacobsen’s nerve, or the tympanic plexus, a branch of the glossopharyngeal nerve, as it exits the jugular foramen just caudal to the cochlea (a.k.a., the hypotympanum). A branch of the vagus nerve (Arnold’s) similarly passes across the middle ear to contribute sensory fibers to the external auditory canal and pinna.
2.1.3 Inner Ear
The inner ear is located in the petrous portion of the temporal bone. This is grossly divided into the cochlea, the vestibule, and the semicircular canals. Within the vestibule are the otolithic organs, the utricle, and the saccule. The inner ear consists of a bony capsule that is filled with perilymph. Within the bony labyrinth is the membranous labyrinth that is filled with endolymph. The membranous labyrinths contain the hair cells that are responsible for the detection of sound, movement, and gravity. A detailed description of the microanatomy of the inner ear is beyond the scope of this text. The internal auditory canal passes from the petrous apex to the labyrinth, just medial to the vestibule. This contains the cochlear, facial, superior vestibular, and inferior vestibular nerves. The petrous apex may contain air cells contiguous with the middle ear or bone marrow.
2.2 Histology
The cochlea is the portion of the boney labyrinth that contains the organ. It is a spiral boney canal about 35 mm in length, which spirals for two 3-quarter turns around a conical pillar of spongy bone, the modiolus, and forming a conical spiral shape contour (Fig. 3a). The cochlear canal is an enlargement of one cross-section of the cochlea. It consists of three parts, scala vestibuli, and scala tympani, are lined by a simple squamous epithelium, and supported by the connective tissue, blending with the periosteum. These are perilymphatic spaces filled with perilymph. The scalae are in communication with the helicotrema at the apex of the cochlea. The perilymph of the scala vestibule is continuous with that in the perilymphatic space of the vestibule, and thus reaches the inner ear of the oval window, which is occluded by the base plate of the stapes. The organ of the Corti, the auditory sensory organ, is supported on a basilar membrane, while the tectorial membrane is formed on the surface of the spiral limbus. Bundles of afferent nerve fibers arise from the base of the organ of Corti, and converge toward the spiral ganglion, which is located in the bone of modiolus (Fig. 3a, b).
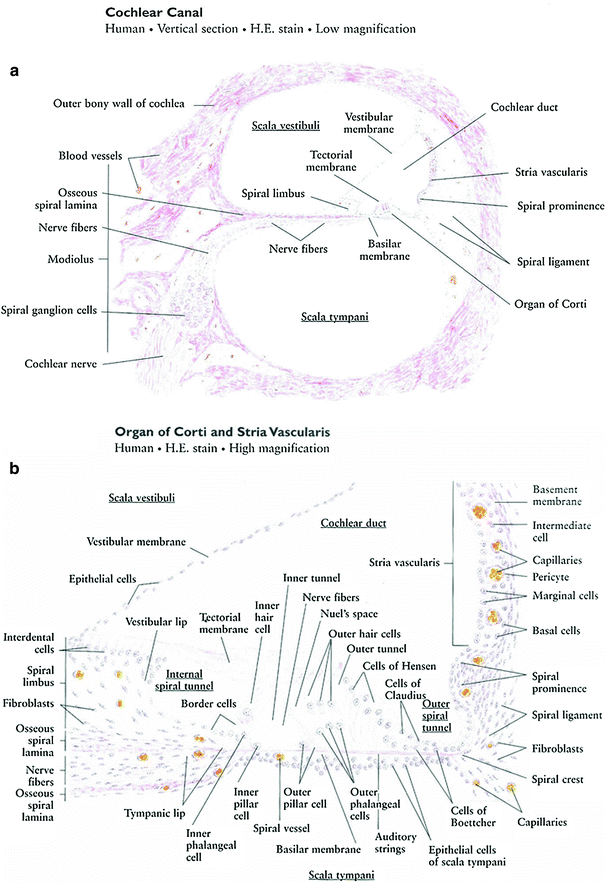

Fig. 3
a Cochlear canal: vertical section low magnification. b Organ of corti and stria Vascularis: high magnification (with permissions from Zhang 1999)
The organ of Corti is an epithelial auditory receptor composed of supporting and hair cells, resting on the basilar membrane of the cochlear duct. Two rows of pillar cells, the inner and outer pillar cells, form a triangular canal, the internal tunnel, which is an important landmark of the organ of Corti. At the inner side of the organ of Corti, neighboring inner hair cells and inner phalangeal cells surround the internal spiral tunnel. The basilar membrane, supporting the organ of Corti, contains radial range auditory strings. Vibrations of the auditory strings are transmitted to the hair cells by displacement of the tectorial membrane over the hair cells, and then converted into adequate bioelectrical impulses, which result in auditory sensations (Fig. 3b).
3 Physiology
Sound waves are collected and transmitted from the external ear to the middle ear, where they are converted into mechanical vibrations. The mechanical vibrations are then converted at the oval window into fluid vibrations within the internal ear. Fluid vibrations cause displacement of the basilar membrane on which rest the auditory sensory hair cells. Such displacement leads to stimulation of the hair cells and a discharge of neural impulses from them. The cochlear duct is shown here as if straightened (Fig. 4).
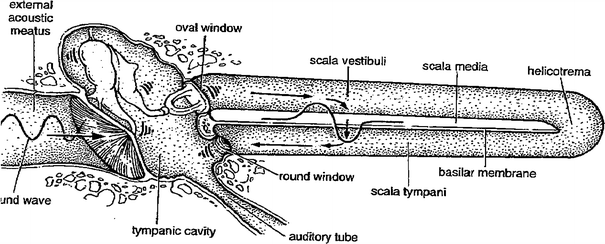

Fig. 4
Physiology Schematic diagram illustrating the dynamics of the three divisions of the ear (with permissions from Karmody 1983)
4 Pathophysiology
4.1 External Ear
Many inherent defense mechanisms are associated with the external ear, including hair follicles that help prevent gross contamination. The acidic ear canal environment inhibits bacterial and fungal growth. Cerumen is relatively hydrophobic and contains antimicrobial products. The first step in the pathogenesis of otitis externa is the breakdown of the skin-cerumen barrier. Radiation leads to progressive obliteration of vascular channels, inflammation and scaling of ear canal skin, and loss of adnexal structures, including the cerumen-producing glands. Resulting pruritus leads patients to clean their ears (e.g., with cotton swabs), which may further breach the natural barriers. The dark, warm, and moist ear canal offers favorable conditions for microbial growth (Fig. 5 and Tables 1, 2, 3).
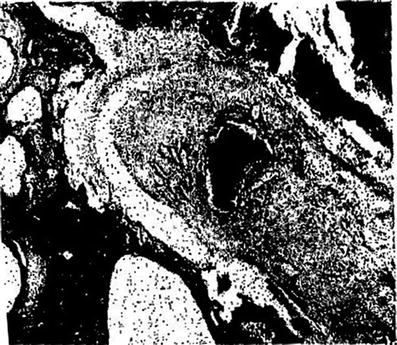

Fig. 5
Fibrosis of the middle ear with old hemorrhage at the center, hollow spaces where cholesterol crystals have dissolved and aseptic osteitis in bone walls with lacunar absorption (with permission from Rubin and Casarett 1968)
Table 1
Post-radiation noninfectious otologic histopathologic findings in human and animal studies: External ear
Edema, hemorrhage, epithelial hyperemia, hyperplasia, and atrophy |
Atrophy of ceruminous glands and hair follicles |
Vasculitis of soft tissue surrounding the external auditory canal |
Subepithelial fibrosis |
Leucocytic infiltration |
Chondromalacia |
Canal cartilage necrosis |
Canal stenosis |
Canal osteonecrosis |
Table 2
Post-radiation noninfectious otologic histopathologic findings in human and animal studies: Middle ear
Mucosal hyperemia, stasis, hemorrhages, edema, and fibrosis |
Diminution of epithelial cytoplasmic mass, ciliary loss, widening intercellular spaces, changes in the number of goblet cells |
Tympanic membrane sclerosis and perforation |
Osteonecrosis of temporal bone |
Necrosis of ossicular chain |
Petrous bone necrosis |
Eustachian tube fibrosis, stenosis/occlusion |
Atrophy of pharyngeal orifice of eustachian tube |
Serous otitis media |
Secondary cholesteatoma |
Table 3
Post-radiation noninfectious otologic histopathologic findings in human and animal studies: Inner ear
Cochlea and spiral ganglion |
Hemorrhage |
Leucocytic infiltration |
Organ of Corti destruction |
Damage and loss of sensory hair cells |
Stria vascularis atrophy, disruption/disintegration |
Vascular fibrosis |
Papilla disruption/disintegration |
Edema of Dieter cells and Henson cells |
Basilar membrane disruption |
Elevation of tectorial membrane |
Spiral membrane disruption |
Destruction of cochlear duct |
Spiral ganglion atrophy and nerve fibre loss |
Degeneration of cochlear nerve |
Vestibular labyrinth and ganglion |
Hemorrhage |
Leucocytic infiltration |
Degeneration and atrophy of sensory epithelium of crista ampullaris |
Endolymph/perilymph system |
Hemorrhage into perilymph |
Distension of pelymphatic spaces |
Distension of endolymphatic spaces (Endolymphatic hydrops) |
Facial Nerve |
Facial nerve edema, hyperemia, and demyelination |
Radiation-induced acute external ear reactions involve skin reactions including erythema and dry or moist desquamation (Table 1). Occasionally, it leads to otorrhea and pain, occasionally combined with skin ulceration (Carls et al. 2002). Microbial overgrowth may lead to acute otitis externa. Late reactions include atrophy, deep ulceration, thickening of the epithelium of the canal, and subepithelial fibrosis. Chronic otitis externa is often followed by pain with otorrhea and otalgia. Radiation may induce osteitis, vasculitis of surrounding soft tissue of the external auditory canal, deep ulceration of the external auditory canal, cartilage necrosis of the external canal, and osteonecrosis. Underlying osteonecrosis of the temporal bone may manifest as otitis externa refractory to treatment, including necrotizing otitis externa (a.k.a., skull base osteomyelitis). Occasionally, RT injury may lead to canal stenosis (Carls et al. 2002). Chronic otitis externa and canal stenosis have been linked in patients receiving high doses of radiation to the external auditory canal (Carls et al. 2002; Bhandare et al. 2007).
4.2 Middle Ear
Middle ear mucosa consists of ciliated and nonciliated cells (squamous, cuboidal, and columnar), secretory cells, and support cells. The cilia usually covers the anteroinferior one-third to two-thirds of the middle ear mucosa and are important components of the mucociliary transport system of the middle ear. The eustachian tube lumen contains ciliated epithelium and secretory cells over the hyaline cartilage framework (Table 2).
4.2.1 Animal Studies
The early effects of radiation on the ear are typical of any inflammatory reaction. This includes vascular dilation with pyknotic changes in the endothelium and necrosis of damaged cells (Klemm 1967), swelling of endothelial cells, extravasation of blood cells, stasis, perivascular edema, sticking of platelets, and leukocyte migration leading to subendothelial proliferation (Berg and Lindergren 1961). Fibrosis and other related changes in the middle ear mucosa in rabbits exposed to 3,000–10,500 Roentgen have been reported (Berg and Lindergren 1961). In mild cases, the mucosa was thickened and fibrotic. The epithelium subsequently showed atrophy with some inflammatory cells and hemorrhagic exudates in a few cases. More severe cases exhibited destruction of the epithelium, involvement of the ossicles, evidence of lacunar absorption in the bony walls, and often purulent exudates with both plasma cells and lymphocytes. An example of middle ear fibrosis is shown in Fig. 5.
4.2.2 Human Studies
The radiation-induced changes (i.e., ciliary, vascular, and stromal) in the middle ear mucosa of patients treated with RT for head-and-neck tumors have been observed under transmission and scanning electron microscopes (Elwany 1985) (Table 2). The epithelial changes have included diminution of the cytoplasmic mass, variable degrees of ciliary loss, widening of the intercellular spaces, and reduction in the number of goblet cells in some cases while increases in others. Middle ear mucosa usually only has a few goblet cells (Tos 1979). The density of goblet cells typically increases significantly with chronic tubal occlusion, OME, and active chronic suppurative otitis media. Few tubular seromucous glands may be present in the anterior part of the normal mesotympanic mucosa (Gristwood and Beaumont 1979). The new-gland formation may occur through the invagination of surface epithelium or the division of basal cells. The number and activity of the newly formed glands depends on the nature of the pathologic process in the middle ear, as the glands are highly active in secretory otitis. Normal cilia should beat in the same direction and in a synchronized fashion (metachronal beating). The abnormal ciliary motility is indicative of early ciliary damage. Ciliary loss represents the end stage (Dudley et al. 1982). The reduced cytoplasmic mass could be directly due to cell damage from RT, or it could be secondary to the obliterative vascular changes. The changes in lamina propria include decreased activity of the seromucous glands, depleted secretory granules in acinar cells, and moderate swelling of mitochondria.
Similar to radiation-induced vascular changes in the other parts of the body (Rubin and Cassarett 1968), the capillaries of the middle ear mucosa show endothelial swelling, duplicated basement membrane, and fibrosis and thickening of the vessel walls with replacement by a cord of fibrous tissue, thereby altogether occluding the lumen. The vascular changes are representative of successive stages in the obliterative disease process and are also related to the amount of radiation received by the vessel. Another important factor contributing to vessel damage is injury to surrounding soft tissue with the release of different breakdown products, such as free radicals and peroxides (Baker et al. 1978).
The post-RT connective tissue stroma shows increased production of collagenous fibrous tissue and an increase in synthetically active fibroblasts in the tunica propria. These fibroblasts are characterized by abundant, irregular cytoplasmic processes, numerous mitochondria, and a well-developed granular endoplasmic reticulum.
Morbidity associated with the tympanic membrane involves thickening, sclerosis and perforation without, or after removal or extrusion of tympanostomy tubes (Bhandare et al. 2007). Fibrovascular granulation tissue formation may lead to the growth of inflammatory polyps through a tympanic membrane perforation and persistent otorrhea (Jereczek-Fossa et al. 2003). However, thickening of the tympanic membrane may persist several months after RT and permanent changes are uncommon (van Hasselt and Gibb 1999). Tympanic membrane perforations have been reported to develop long after RT (Carls et al. 2002).
Chronic otitis media (COM) and OME are frequently encountered in patients with head-and-neck cancers (particularly nasopharyngeal cancer) after RT. The development of COM and OME in post-RT patients is attributed to both impaired tubal function and middle ear inflammation (Sato et al. 1988; Young and Hsieh 1992; Young et al. 1995; Ho et al. 2002). Radiation-induced OME occurs in patients with no pre-RT tubal pathology and may have no bearing to the patency of the eustachian tube (Anteunis et al. 1994). The likelihood of an ear with pre-RT OME to have long-term post-RT OME was observed to be much higher compared to an ear without pre-RT OME (Low and Fong 1998).
The effects of radiation on middle ear mucosa in the acute phase include mucositis, sloughing, and edema (Borsanyi and Blanchard 1962), Moss (1959) coined the term “radiation otitis media” for this process. In the chronic phase, it includes epithelial atrophy and fibrosis (Berg and Lindergren 1961).
Acute otitis media most commonly occurs during or within a few weeks after completing RT. Swelling, puffiness, and transient edema of middle ear mucosa caused by the acute inflammatory reaction lead to functional impairment of the tube. Mucosal sloughing exposes binding sites for pathogens and impairs the normal mucociliary clearance mechanism.
OME may develop as a result of dilated capillaries and fluid transduction resulting in edema from further reduction in middle ear pressure. If the eustachian tube is impaired, the subsequent resorption of middle ear gas may create a negative pressure, which promotes further transudation (i.e., “hydrops ex vacuuo”). Inflammatory components of the middle ear effusion may then perpetuate the inflammation, creating a “vicious cycle”. Subsequent to high doses of radiation to the middle ear, two types of chronic otitis media may be observed. The presence of post-RT middle ear effusion without signs of acute inflammation indicates chronic OME (COME), whereas persistence of the suppurative process through a perforation or a tympanostomy tube indicates chronic suppurative otitis media (CSOM).
4.2.3 Eustachian Tube
The eustachian tube connects the middle ear cavity and the nasopharynx. The eustachian tube framework is cartilage. It is lined with respiratory mucosa. Between the cartilage and mucosa is Ostmann’s fat pad, which generally occludes the tube lumen at rest (Aoki et al. 1994). The tensor veli palatini and levator veli palatini muscles arise from the tubal cleft before inserting on the palate. In its resting state, the eustachian tube is closed. Movements of the palate lead to a transient tubal opening.
Differences between pre- and post-RT eustachian tube dysfunction have been reported and are usually observed in nasopharyngeal carcinoma. Pre-RT eustachian tube dysfunction in nasopharyngeal carcinoma patients is caused by tumor invasion of the tensor veli palatini muscle, extrinsic compression, or inflammation (Young and Hsieh 1992; Young and Sheen 1998. Post-RT eustachian tube dysfunction in nasopharyngeal carcinoma patients is caused by both tumor obstruction and functional impairment of the tube (Young et al. 1994; Bhide et al. 2007. The tubal patency and clearance function are affected by radiation, but dynamic function is preserved after radiation (Young and Hsieh 1992).
Post-RT COME has been attributed to obstruction of the eustachian tube opening and fibrosis of the fascial space around the levator veli and tensor palatini muscle (Lederman 1962). Edema and fibrosis resulting from RT can interfere with muscle function and lead to eustachian tube dysfunction (Dias 1966). Abnormalities of the mucosa of middle ear affect the production of mucous and its transportation down the eustachian tube (Fischer et al. 1978). RT has been shown to affect the mucosal cells in the middle ear (Gyorkey and Pollock 1960). The altered nature of mucous production both in quality and quantity after RT can contribute to serous otitis media in these patients.
The blockage of the pharyngeal orifice of the eustachian tube due to radiation-induced inflammatory reactions and the resorption of gas from the middle ear results in negative pressure, retraction of the tympanic membrane, and decreased mobility of the ossicular chain. The end result is pressure, pain, and hearing loss.
The supporting structures surrounding the eustachian tube were seen to be replaced by fibrous tissue in one study on the histopathology of the human eustachian tube (Takasaki et al. 2000). In some cases, the recovery of its functional impairment during the 5-year period post-RT has been observed and attributed to a subsiding inflammatory reaction (Young et al. 1994). Further delayed fibrosis and atrophy of the pharyngeal orifice of the eustachian tube, its mucosal lining, and Ostmann’s fat pad may contribute to the development of a pathologically patent, “patulous,” tube 5–10 years after RT (Takasaki et al. 2000). This may lead to a constant sensation of aural fullness and the patient hearing his or her own breathing or voice (known as autophony).
4.2.4 Osteonecrosis of the Temporal Bone and Ossicular Chain
Osteoradionecrosis of the temporal bone is a well recognized, pathological entity that manifests with symptoms of refractory otitis externa, otalgia, otorrhea, and hearing loss (Leach 1965; Schuknecht and Karmody 1966; Thornley et al. 1979; Wurster et al. 1982). The basic reactions in the temporal bone to radiation involve vasculitis leading to obliterative endarteritis and aseptic necrosis of bone with compensatory reparative fibrosis enhancing the susceptibility of bone to injury, infection, and fracture. The effects of radiation have been divided into effects on soft-tissue damage and effects on bone (Leach 1965; Schuknecht and Karmody 1966; Fajardo and Berthrong 1988). The soft-tissue damage includes dermatitis of the external auditory canal, middle ear mucosal inflammation, otitis media, and aseptic labyrinthitis. The histologic observations on bone include destruction of osteoblasts from trabecular margins, loss of osteocytes (empty lacunae), progressive replacement of hematopoietic elements by loose connective tissue, thickening of vascular walls, narrowing of lumen with a concomitant decrease in the number of capillaries and sinusoids, and the absence of osteocytes followed by a lack of new osteoid deposits. The ischemic changes make the bone susceptible to infection.
Osteonecrosis of the temporal bone has been separated into two distinct patterns, localized and diffuse (Ramsden et al. 1975). The most common finding in the localized pattern was an area of exposed dead bone in the external acoustic meatus (EAM), usually in the floor of the tympanic ring, and occasionally in the anterior wall. These cases presented with mild otalgia and severe otorrhea. Although the bony dehiscence may heal when the bone sequestrum forms and gradually separates, the process may take years. In diffuse osteonecrosis of the temporal bone, there can be extensive ischemic necrosis of a large part or all of the temporal bone (van Hasselt and Gibb 1999). With diffuse osteoradionecrosis of the temporal bone, multiple bony sequestra form, and the patient may experience more serious problems such as cerebrospinal fluid leak, labyrinthine fistula, facial palsy, chronic mastoiditis, meningitis, sinus thrombosis, brain abscess, and death (van Hasselt and Gibb 1999; Leonetti et al. 1997; Sikand and Longridge 1991; Ito et al. 2000; Horan et al. 2007). Radiographs of the temporal bone may reveal a widespread moth-eaten radiolucency. The clinical manifestations in these cases often include pain, refractory otorrhea, CHL, SNHL, or mixed hearing loss. Associated diffuse white matter injury may lead to progressive cognitive neurological impairment with simultaneous vestibular or gait disorders (Jereczek-Fossa et al. 2003). A study on the incidence of radiation otomastoiditis using T2-weighted MRI reported significant increases above RT doses of 50 Gy, but decreases over time (Nishimura et al. 1997).
Delayed persistent CHL without OME is considered to be secondary to osteoradionecrosis of the ossicular chain with or without damage to the auditory end organ. Destruction of the incudostapedial joint after high-dose RT has been described in a case report (Thornley et al. 1979). Cases of osteoradionecrosis of the temporal bone along with CHL have been presented with speculation of radionecrosis of the ossicular chain (Kristensen and Jorgensen 1967). Surgical exploration in a case of CHL consistent with interruption of the ossicular chain or stapes fixation after high-dose RT revealed that the long process of the incus was replaced by a fibrous band (Kristensen and Jorgensen 1967). Another report on five patients with post-RT temporal-bone necrosis with mixed hearing loss (MHL) years after RT without symptoms of OME speculated the cause to be ossicular chain necrosis (Bhandare et al. 2007). Extensive osteonecrosis of the ossicular chain after resection of extensive osteonecrosis of the temporal bone has been documented (Gyorkey and Pollock 1960).
4.3 Inner Ear
Radiation-induced inner ear morbidity may include damage to the labyrinth (cochlea, otolithic organs, and semicircular canals), cochlear or vestibular nerves, and central auditory and vestibular pathways. Most of the information on the pathogenesis of radiation-induced inner ear damage and dysfunction comes from animal experiments (Novotny 1951; Berg and Lindergren 1961; Kelemen 1963; Winther 1969; Bohne et al. 1985) with a few human autopsy studies (Gyorkey and Pollock 1960; Hoistad et al. 1998; Leach 1965). Despite the animal studies and other patient data, a relationship among pathogenesis, pathophysiology of the radiation lesion, and subsequent clinical manifestations has not been established (Table 3).
4.3.1 Vestibular Damage
4.3.1.1 Animal Studies
There is limited experimental data on radiation damage to the vestibular labyrinth. Gross clinical vestibular findings without detailed pathologic studies of the vestibular structure in guinea pigs have been reported 2 months after single-dose X-ray RT (Gamble et al. 1968). These observations included constant turning in one direction and a tendency to fall to the affected side. In a study of guinea pigs radiated to single-fraction high doses of 7,000 Roentgen, impaired balance function with positional nystagmus was correlated with radiation-induced degenerative changes in vestibular sensory epithelia on electron-microscopic examination. The vestibular apparatus was felt to be more resistant than the cochlea to the effects of radiation (Winther 1969). Another study (Nadol 1988) reported disturbed equilibrium in animals after a single-fraction RT to doses greater than 50 Gy.
4.3.1.2 Human Studies
In early human studies, a number of patients treated to doses between 30 and 120 Gy were reported by Leach (1965) to exhibit episodic, occasional, intermittent vertigo and moderate to severe balance problems, and no caloric response. An autopsy of a patient treated to high-dose RT demonstrated the absence of the organ of Corti, macula of utricle, and cristae of semicircular canals, as well as atrophic spiral ganglions and nerves (Leach 1965).
Imbalance and vertigo are the predominant symptoms of vestibular dysfunction. Gradual development of vestibular dysfunction will be offset by central nervous system compensation and varying degrees of imbalance. Acute onset of vestibular dysfunction will be manifest by vertigo. If mild, this may be noted only with movement. If more severe, it may be spontaneous, violent, and accompanied by nausea and vomiting. Acute onset of vertigo reported as lasting more than a day and accompanied by SNHL is often classified as labyrinthitis. In diagnostic evaluation of post-RT vertigo, the differential diagnosis of the peripheral etiology due to vestibular dysfunction from the central etiology caused by dysfunction of central vestibular structures in the brainstem or cerebellum is essential. Though a number of studies have reported vestibular dysfunction (Bhandare et al. 2007; Young et al. 2004; Zabel et al. 2004), data on the mechanism and pathophysiology of radiation-induced vestibular damage and subsequent clinical manifestations remain sparse.
4.3.2 Cochlear Damage
Table 3 lists the post-radiation noninfectious otologic histopathologic findings in human and animal studies.
4.3.2.1 Animal Studies
Subsequent to experimental fractionated RT (2 Gy per fraction, 5 days per week, 40–90 Gy) to chinchillas, Bohne et al. (1985) reported damage to the organ of Corti in 31 % of animals treated with 30–40 Gy and 62 % of animals treated with 60–90 Gy. The observations included loss of myelinated nerve fibers in the osseous spiral lamina, loss of peripheral processes of the spiral ganglion cells indicating degeneration of the ganglion cell bodies, scattered cell loss throughout the organ of Corti, significant losses of the inner and outer hair cells, and supporting cell losses in the inner and outer pillars.
Winther (1969) subjected guinea pigs to single-fraction RT ranging from 1,000 to 7,000 Roentgens. During their short follow-up, they did not notice any changes in hair cells with doses less than or equal to 2,000 Roentgens. The earliest post-RT changes with lower doses were observed in the stria vascularis, and increases in dose caused apoptosis of hair cells and distention of Reissner’s membrane, consistent with endolymphatic hydrops, which may impair function of remaining hair cells (Gamble et al. 1968). The stria vascularis has been suggested to be more sensitive to radiation than the organ of Corti or spiral ganglion in both animal and human temporal bone studies (Gamble et al. 1968; Schuknecht and Karmody 1966). The vestibular apparatus is suggested to be more resistant to the effects of radiation than the cochlea (Gamble et al. 1968). A study in guinea pigs suggested that hair cell damage and compound action potential changes (i.e., hearing loss) occur secondary to stria vascularis degeneration (Ocho et al. 2000).
Auditory neurons in humans and animals differ (Nadol 1988; Felix 2002). The general pattern of neural degeneration is evident in the human cochlea, but at a significantly slower rate than in animals (Felix et al. 1990; Nadol 1990; Felder et al. 1997). The effects of radiation observed on stria vascularis and the organ of Corti in animal experiments have been reported to be similar to those found in the temporal bone of a patient who received radiation to his or her ear (Gamble et al. 1968; Schuknecht and Karmody 1966).
4.3.2.2 Human Studies
Histologic changes in connective tissue as well as vascular insufficiency have been suggested to result in cochlear anoxia and manifest as SNHL (Borsanyi and Blanchard 1962). Autopsy studies of nine temporal bones in post-RT patients with head-and-neck tumors showed a loss of inner and outer hair cells and spiral ganglion cells in the basal turn, atrophy of the stria vascularis, and changes in the vessels of the facial nerves (Hoistad et al. 1998). The loss of hair cells in mammalian cochlea leads to permanent SNHL and initiates a number of pathological changes to the primary auditory neurons that normally project to the organ of Corti (Schuknecht and Karmody 1966). The pathological changes start with a rapid and extensive loss of the unmyelinated peripheral processes within the organ of Corti that normally innervate the inner hair cells (Terayama et al. 1977), followed by a more gradual degeneration of the myelinated portion of the peripheral processes and ultimately cell death (Spoendlin 1984; Leake and Hradek 1988; Shepherd and Javel 1999; Hardie and Shepherd 1999). One histological study supports the theory that the greatest damage to the labyrinth is the result of injury to the vessels of the stria vascularis and perhaps the cells of stria, which appear to be more easily affected than other nonvascular structures in the cochlea (Leach 1965).
Acute hearing loss after RT has been suggested to be partly due to transient alterations in the endolymph and perilymph physiology caused by disturbances in the stria vascularis (Linskey and Johnstone 2003). The mechanisms responsible for transient SNHL remain unknown. Radiation damage to the organ of Corti with loss of inner and outer hair cells and pillar cells (Winther 1969; Bohne et al. 1985; Hoistad et al. 1998; Schuknecht and Karmody 1966, degeneration of endothelial cells in vessels with a reduction in the number of capillaries (Borsanyi and Blanchard 1962; Berg and Lindergren 1961; Schuknecht and Karmody 1966; Sikand and Longridge 1991; Nadol 1990), and atrophy and degeneration of the basilar membrane, spiral ligament, stria vascularis (Hoistad et al. 1998; Schuknecht and Karmody 1966), spiral ganglion cells, and cochlear nerve (Bohne et al. 1985; Hoistad et al. 1998; Schuknecht and Karmody 1966) have been reported in histopathological studies in both animals and humans (Winther 1969; Bohne et al. 1985; Gamble et al. 1968; Leach 1965; Hoistad et al. 1998). Linskey and Johnston (2003) have reported that cochlear hair cells and the stria vascularis are suggested to be the two major sites of damage after high-dose RT. The basal turn of the cochlea, responsible for detecting higher frequencies, shows greater susceptibility to radiation damage. Delayed SNHL most commonly shows a chronic, progressive, and irreversible evolution (Pollock et al. 1995).
Radiation-induced vascular insult, subsequent inflammatory process resulting in progressive microvascular endothelial reactions that lead to slow degeneration and atrophy of the inner ear sensory structures, has been suggested as a process involved in delayed inner-ear damage (Honore et al. 2002). Contrary to this, Gibb and Loh (2000) presented a post-mortem histological examination of the temporal bone in a case of post-RT SNHL with a well-preserved organ of Corti and the presence of a normal number of inner hair cells, scattered losses of outer hair cells, and severe patchy atrophy of the stria vascularis after high-dose radiation. They concluded that degeneration of cochlear nerve and central auditory pathway was possible, instead of damage to the sensory end organ, as an etiology. Other causes of damage, such as free oxygen radicals and resulting apoptosis, needed to be excluded.
5 Clinical Syndromes
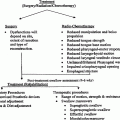
RT-induced morbidities show significant variation throughout the auditory system. To objectively compare studies, the criteria for diagnosis of any radiation-induced morbidity must be specified in the study. While identifying and diagnosing some post-RT morbidities and dysfunctions is based solely on clinical manifestations, others cannot be diagnosed without subjecting the patients to specific tests, and outcomes cannot be evaluated without an objective criteria. Table 4 summarizes the RTOG scoring criteria. The SOMA LENT system provides a means to categorize and grade late effects (Tables 4, 5).
Table 4
Acute radiation ear morbidity according to the RTOG scoring criteria
Score | Criteria |
|---|---|
0 | No change over baseline |
1 | Mild external otitis with erythema, pruritus, secondary to dry desquamation not requiring medication. Audiogram unchanged from baseline |
2 | Moderate external otitis requiring topical medication, serous otitis media, and hypoacusis on testing only |
3 | Severe external otitis with discharge or moist desquamation, symptomatic hypoacusis, tinnitus, and not drug related |
4 | Deafness |
Table 5
LENT SOMA Scoring System of the Ear
EAR | ||||
|---|---|---|---|---|
Grade 1 | Grade 2 | Grade 3 | Grade 4 | |
Subjective | ||||
Pain | Occasional and minimal | Intermittent and tolerable | Persistent and intense | Refractory and excruciating |
Tinnitus | Occasional | Intermittent | Persistent | Refractory |
Hearing | Minor loss, no impairment in daily activities | Frequent difficulties with faint speech | Frequent difficulties with loud speech | Complete deafness |
Objective | ||||
Skin | Dry desquamation | Otitis externa | Superficial ulceration | Deep ulceration, necrosis, and osteochondritis |
Hearing | <10 decibel loss in one or more frequencies | 10–15 decibel loss in one or more frequencies | >15–20 decibel loss in one or more frequencies | >20 decibel loss in one or more frequencies |
Management | ||||
Pain | Occasional non-narcotic | Regular nonnarcotic | Regular narcotic | Parenteral narcotics |
Skin | Occasional lubrication/ointments | Regular eardrops or antibiotics | Eardrums | Surgical intervention |
Hearing loss | Hearing aid | |||
Analytic | ||||
Pure tone audiometry | Assessment of characteristics of sensorineural perception | |||
Speech audiometry | Assessment of characteristics of speech perception | |||
5.1 Detection and Diagnosis
5.1.1 External Ear
Several conditions and their associated presentation include the following (some with overlap with the middle ear):
(a)
Necrosis of the pinna. Necrosis of the pinna exhibits both temporary and persistent ulceration of soft tissue and/or chondritis.
(b)
Acute otitis externa. Acute otitis externa is indicated by acute onset of pain, drainage, and swelling of the ear canal with extreme tenderness to traction on the pinna.
(c)
Chronic otitis externa. Chronic otitis externa includes pruritus, otorrhea, scaling, edema, and erythema of the external auditory canal or auricle.
(d)
Canal stenosis. Although more stringent criteria, such as 50 % constriction of bony lumen on radiographic examinations, can be used to define canal stenosis, post-RT stenosis of the external acoustic canal can be identified on clinical examination by otoscopy of the external acoustic canal and/or supported by radiography.
(e)
Osteonecrosis of the external auditory canal. This condition is diagnosed by the exposure of the bone in the ear canal or radiographic evidence of bony sequestra (Tables 6, 7).
Table 6
Criteria used to report hearing status after SRS or FSRT (stereotactic radiosurgery or fractionated stereotactic radiotherapy)
Score | Criteria |
|---|---|
1 | Preservation of pretreatment hearing level |
2 | Useful/serviceable hearing (corresponds to Gardener-Robertson Hearing Grade[GRHG] I-II), with commonly used criteria to define serviceable hearing as PTA ≤ 50 % and SDS ≥ 50 % |
3 | Measurable hearing; any hearing with detectable PTA |
4 | Preservation of the pretreatment hearing level corresponds to (a) GRHG I–IV hearing as preservation of preradiation GRHG, (b) for GRHG V patients with no speech discrimination but testable PTA as preservation of PTA scores |
5 | Improvement as well as loss in hearing expressed as change in GRHG |
Table 7
Gardner-Robertson scale
Grade | PTA or SRT | SDS (%) |
|---|---|---|
1 | 0–30 | 70–100 |
2 | 31–50 | 69–50 |
3 | 51–90 | 49–5 |
4 | 91(ML) | 4–1 |
5 | NR | NR |
5.1.2 Middle Ear
Several conditions and their associated presentation include the following:
(a)
Acute otitis media. Acute otitis media during or after RT is diagnosed by the abrupt onset of symptoms due to middle ear inflammation (i.e., otalgia and CHL) accompanied by effusion, bulging, opacification, and erythema of the tympanic membrane. Spontaneous tympanic membrane perforation may occur.
(b)
Chronic otitis media. Chronic otitis media after radiation is divided into two categories: chronic otitis media with effusion (COME) and chronic suppurative otitis media (CSOM). The criteria for COME include the presence of middle ear effusion without overt signs or symptoms of infection. Patients typically note aural fullness and hearing loss. CSOM is diagnosed when the suppurative process persists despite treatment and is nearly universally accompanied by otorrhea through a perforation or a tympanostomy tube.
(c)
Mastoiditis. The diagnosis is derived from a clinical judgment based on the spread of otitis media across the mastoid with swelling, erythema, and occasionally fluctuance behind the pinna. If severe, the underlying bone may become demineralized, which is known as coalescent mastoiditis. Post-RT patients with acute mastoiditis receive intravenous antibiotics, a mastoidectomy, or both. These patients should be coded as having true mastoiditis.
(d)
Eustatchian tube. A functional, patent eustachian tube is necessary for ideal middle ear mechanics. Unfortunately, testing of eustachian tube patency and function after RT can be difficult to perform, so it is not usually directly measured. Eustachian tube function is most commonly assessed using tympanometry. Evaluation of eustachian tube function may be performed using four parameters: opening pressure, which tests for the passive opening function of the eustachian tube; positive and negative pressure tests, which indicate the dynamic functions of the tube; and clearance time, which indicates the clearance function of the tube. Pneumatic otoscopy, for retraction and stiffness secondary to effusion, indirectly assesses the tubal patency and function. Valsalva, Politzer, and Toynbee tests may yield gross information on tubal patency, but they are not routinely done by otologists.
5.1.3 Inner Ear
Tinnitus, the perception of a sound without acoustic stimulation, has been associated with cochlear damage, hearing loss due to pathologically increased spontaneous firing rates, or hyperactivity of neurons in the auditory pathway. Hypoxia/ischemia may play an important role in the pathogenesis of tinnitus secondary to SNHL, although the exact pathophysiologic processes involved in post-RT tinnitus is unknown (Mazurek and Haupt 2005).
The availability of a baseline evaluation is necessary for accurate diagnosis. Before beginning RT with high doses to the hearing apparatus, patients should be considered for a basic otologic evaluation including morphological evaluation by otoscopy, tympanometry, and other basic functional audiometric tests including pure-tone audiometry, speech audiometry (for speech-reception threshold and speech discrimination), and stapedial reflexes. Tympanometry objectively measures the middle ear acoustic impedance as well offers information on middle ear aeration, ossicular chain mobility, and eustachian tube function. Tympanometry may be followed by pure-tone audiometry to exclude otitis media and ossicular fixation as the cause of hearing loss (Raaijmakers and Engelen 2002). The audiologic assessment should be followed by repeated biannual evaluations for a period of at least 5 years after RT. Tuning-fork tests (e.g., Rinne and Weber) may be done with a 512 Hz tuning fork and yield relatively crude insights into the nature of the hearing loss (i.e., CHL vs. SNHL).
Pure-tone audiometry, including air and bone conduction, can subjectively assess patients’ hearing thresholds and detect sensorineural, conductive, or mixed hearing loss. Transient CHL secondary to middle ear effusion is evaluated by pure-tone audiometry (air-bone-gap measurement), tympanometry, and otoscopy.
SNHL can be detected with audiometric assessment by measuring speech understanding and pure-tone thresholds with bone conduction at 0.5, 1, 2, 4, and 6 kHz. Pure-tone stimuli are presented via a bone oscillator placed on the mastoid of the ear to be tested, thus by passing the conductive mechanism. “Masking” of the contralateral ear is often necessary to avoid inadvertent detection of sound by the contralateral ear. There remains significant variation in the criteria used for coding of post-RT SNHL in the literature in terms of frequencies and the thresholds (Bhandare et al. 2007; Pan et al. 2005; Chen et al. 1999; Kwong et al. 1996) making the comparison between the studies difficult. To standardize reporting following criteria has been suggested (Bhandare et al. 2010).
The effect should be determined through pre- and post-RT audiometry evaluations of same ear (i.e., the contralateral ear should not be used).
To avoid transient post-RT hearing fluctuations, hearing should be tested starting 6 months post-RT and at least biannually thereafter.
Speech discrimination (SD) and 4-frequency (0.5, 1, 2, and 3 kHz) bone-conduction pure-tone average should be used, as endorsed by the American Academy of Otolaryngology-Head and Neck Surgery Committee on Hearing and Equilibrium (Committee on Hearing and Equilibrium 1995).
Additionally, 6 kHz bone conduction thresholds should be measured, because (1) the basal turn of the cochlea (i.e., highest frequencies) is the first to be affected, (2) 6 kHz is highest frequency bone conduction threshold measured with standard bone conducting transducers, (3) bone conduction thresholds minimize the influence of concomitant middle and external ear pathology. Above 6 kHz, measurements are performed using air-conduction thresholds alone, because bone conducting transducers generally cannot produce stimuli above 6 kHz.
An air-bone gap, assessed by the difference in the air-bone-conduction thresholds, is consistent with CHL. Persistent CHL, without symptoms of OME, is an attenuation of signals stimulating the cochlea without damage to the auditory end organ. It is attributed to fixation and/or osteoradionecrosis of the ossicular chain. Variation in the hearing thresholds over consecutive evaluations should be used to determine if SNHL is transient or persistent. Note that bone-conduction thresholds may also, to a much lesser extent, be altered by the presence of middle ear pathology in addition to the usual test-retest variabilities (Anteunis 1996). In addition to these, in patients with age-adjusted normal values, additional tests may provide useful information. Auditory brainstem response (ABR) testing may be used to assess retrocochlear pathology, such as radiation damage to the auditory nerve. An intra-neural latency response difference of at least 0.30 ms for wave V of the auditory brainstem indicates neural involvement.
In post-RT audiometric evaluations, a recovery from a higher bone-conduction threshold to a threshold of less than 10 dB in consecutive tests may indicate transient SNHL (Kwong et al. 1996). An increase in the bone-conduction threshold exceeding 10 dB in two consecutive tests performed at least 6 months apart may be considered persistent SNHL.
5.1.3.1 Additional Function Tests
Speech audiometry tests include a speech reception threshold and word discrimination scores (WDS). The speech reception threshold serves as a cross-reference for pure-tone air-conduction thresholds, whereas poor WDS may indicate radiation-induced neural damage and may help determine candidacy or ineligibility for hearing aids.
The stapedial-reflex examination evaluates the contraction of the stapedial muscle in reaction to loud sounds, is mediated by neural network with afferent input from auditory nerves and efferent input from the auditory nerve. It provides additional information on the status of neural loop and complements tympanometric measurements (Jereczek-Fossa et al. 2003; Jereczek-Fossa and Orecchia 2002).
Vestibular dysfunction. Spontaneous and induced nystagmus can be recorded and quantified with electronystagmography (ENG) using electrodes placed around the eyes and videonystagmography (VSG) using infrared video cameras over the eyes. These tools record eye movement and can be used to evaluate the vestibule-ocular reflexes for assessment and follow up of vestibular dysfunctions.
Caloric response. Loss of horizontal semicircular canal function is called canal paresis. The caloric test can evaluate vestibular function by irrigating the ear canal with 30 °C (cold) and 44 °C (warm) water or air for 1 min and measuring the resulting eye movement slow-phase velocities (Jongkees and Philipszoon 1964). An interaural difference of more than 20 % is considered abnormal. In patients with unilateral RT, a significantly reduced response to caloric stimulation of the irradiated side compared to the nonirradiated side indicates canal paresis of the irradiated side.
Otolith dysfunction. RT-induced otolith dysfunction can be evaluated using vestibular evoked myogenic potentials, or VEMPs, by recording the muscular activity in the ipsilateral sternocleidomastoid muscle after delivering a loud sound or electrical stimulus to the ear. This test is subject to numerous confounding factors. Absent responses do not necessarily imply absent vestibular function.
Cochlear and auditory pathway. Brainstem auditory evoked response (BAER), brainstem auditory evoked potentials (BAEP), or ABR detect electrical activity in the cochlea and auditory pathway in the brain by measuring the electrical waves from the brainstem in response to clicks or tone bursts in the ear. Neural waves result from synchronized neural discharge along the auditory neural pathway. Outcomes can be affected by pathology anywhere along the auditory pathway, thus abnormal results are nonspecific.
Transient-evoked otoacoustic emissions (TEOAE), which record an acoustic signal generated by the contracting outer hair cells of the inner ear, may reveal radiation damage to the outer hair cells in the organ of Corti. It is only detectable when the external, middle, and inner ear functions normal or close to normal. The emission strength (dB SPL) in the TEOAE reflects the number of active outer hair cells. This method has been used to study the ototoxic side effects of cisplatin, but has not been widely used in evaluating patients after RT (Allen et al. 1998; Biro et al. 1997). However, one study reported accurate identification of patients with a hearing level better than 25 dB for frequencies between 1 and 4 kHz. Also, a TEOAE correctly indentified all cases with a hearing threshold worse than 25 dB HL for all frequencies between 1 and 4 kHz (Johannesen et al. 2002).
5.1.3.2 End-Point Criteria After RT for Vestibular Schwannoma
5.1.4 Radiologic Imaging
Radiology studies such as CT, positron emission tomography (PET), MRI, and nuclear medicine (bone scans) studies can help reveal the development and extent of delayed radionecrosis of the brain or osteoradionecrosis of the temporal bone (Nishimura et al. 1997).
Post-RT morbidities, including hemorrhage into the inner ear, labyrinthitis, and neuronitis, can be revealed by MRI. MRI has been used to visualize the eustachian tube and to assess its anatomy and pathology in patients with nasopharyngeal carcinoma. MRI has been a powerful tool in evaluating endocochlear diseases that cause sudden SNHL (Hegarty et al. 2002). Findings of high labyrinthine signal on unenhanced T1-weighted MRI of the internal acoustic canal in patients who presented with a sudden onset of SNHL 5–20 years following head and neck RT have been reported by Poh and Tan (2007). They posited that these findings are due to labyrinthine hemorrhage as a rare, delayed complication of head-and-neck RT seen in T1-weighted signal changes in the labyrinths of patients who experienced sudden SNHL. Further patency of inner ear fluid spaces as well as occlusion of inner ear fluid spaces by a fibrotic process can be revealed by MRI along with CT (Jereczek-Fossa et al. 2003).
5.1.4.1 Anatomical Definition of the Middle and Inner Ear for Treatment Planning
The maximum dimensions of the middle ear, vestibular apparatus, and cochlea are known to vary from 1 to 2.2, 1.4 to 1.8, and 0.5 to 1 cm, respectively (Pacholke et al. 2005). A 5-mm slice thickness for the computed tomography (CT) scans is insufficient to accurately delineate and evaluate the volume of the middle and inner ear structures. Slice thickness less than or equal to 1 mm through the temporal bone is optimal. Proper treatment planning would include magnetic resonance imaging (MRI) fused with CT and selecting the appropriate window width and level settings (e.g., bone window setting on CT).
The tympanic membrane is often difficult to visualize on CT. Superiorly inferiorly, it extends between two bony projections. The middle ear is defined laterally by the tympanic membrane, annulus, and handle of malleus, medially by the promontory of the cochlea, superiorly by the tegmen tympani, and inferiorly by the bony wall covering the jugular bulb. These landmarks assist with defining and delineating the middle ear cavity on CT during treatment planning.
The eustachian tube is over 3 cm long and has bony and cartilaginous parts. The bony part is over 1 cm long and tapers down from the anterior wall of the middle ear to its orifice. This narrowest part of the tube is the isthmus, which has been related to the pathogenesis of OME (Wang et al. 2007). The cartilaginous part is over 2 cm long and joins the bony orifice at the isthmus. It is located in the groove between the greater wing of the sphenoid and the apex of the petrous apex of the temporal bone. To reduce the dose to these parts, the middle ear cavity and isthmus must be localized by identifying these landmarks for accurate delineation on the CT scans before optimizing a treatment plan.
The inner ear can be well demonstrated on CT scans. The base of the cochlea abuts the anterior aspect of the internal auditory canal fundus. The cochlea is a snail shell-shaped structure that lies anteriorly with its apex pointed anteriorly, inferiorly, and laterally. The vestibule is located posterior to the cochlea and medially adjacent to the internal auditory canal with three semicircular canals emanating at right angles. The internal auditory canal is a landmark that is significant for identifying, localizing, and delineating the cochlea and vestibule.
6 Radiation Tolerance of Otologic Organs
6.1 External Ear
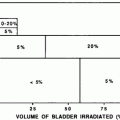
Earlier studies suggested tolerance dose for 50 % complication for acute radiation otitis to be 40 Gy and for chronic otitis, it was 65–70 Gy (Burman et al. 1991; Emami et al. 1991). In a single-institution study of radiation for head-and-neck cancers, external-ear toxicity occurred in 33 % of the patients receiving doses above 50 Gy for megavoltage X-ray radiation (Bhandare et al. 2007). Both acute and chronic otitis externa increased significantly (above 5 % in each dose bin of 5 Gy) as the dose received by the external ear increased above 50 Gy. The incidence of atrophy and canal stenosis was reported to increase significantly above 55 Gy to the external auditory canal. Incidence of both acute and chronic otitis externa as well as atrophy and canal stenosis increased with increases in the radiation dose received by the external auditory canal. A correlation between the incidence of chronic otitis externa, atrophy, and canal stenosis was reported in patients receiving high doses of radiation (>55 Gy) to the external auditory canal (Bhandare et al. 2007). In patients treated with hypofractionated orthovoltage X-ray and electrons for epithelial tumors of the pinna, the incidence of skin necrosis was reported to be 13 % (Ashamalla et al. 1996; Lim 1992; Silva et al. 2000) (Fig. 6a–c, Table 8).
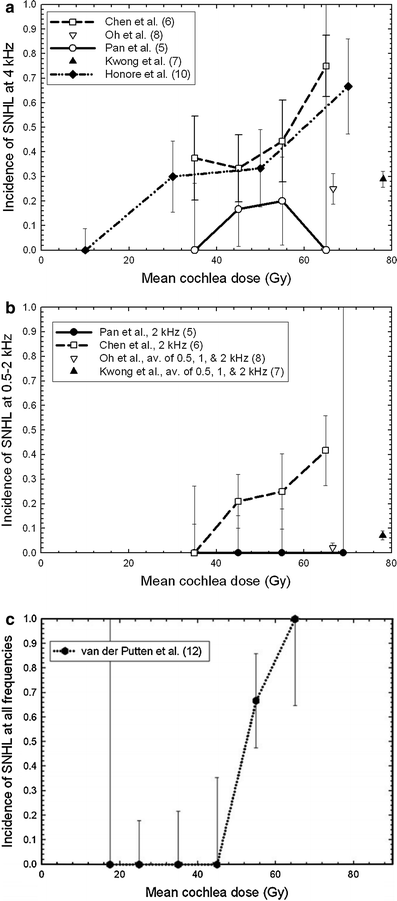

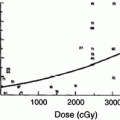
Fig. 6
The rate of SNHL versus mean dose to the cochlea at a 4 kHz, b 0.5–2 kHz, and c all frequencies (with permissions from Bhandare et al. 2010)
Table 8
Selected studies on the treatment of vestibular schwannomas
Author and year | Number of patients in study |
|---|