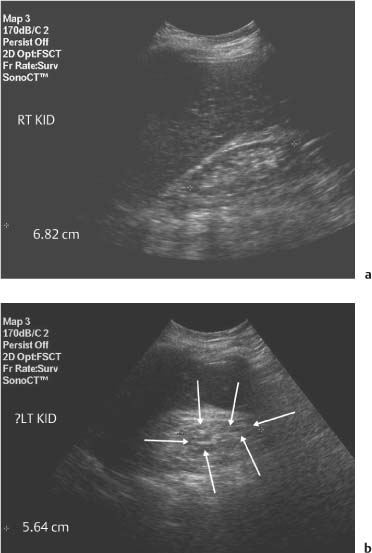
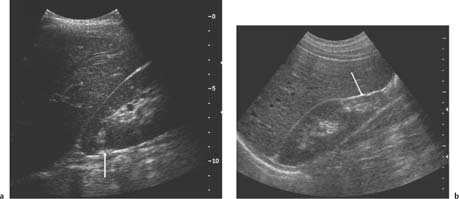
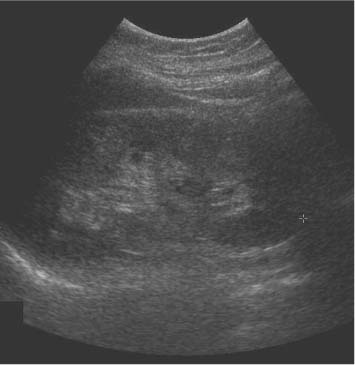
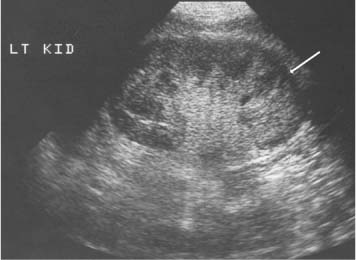
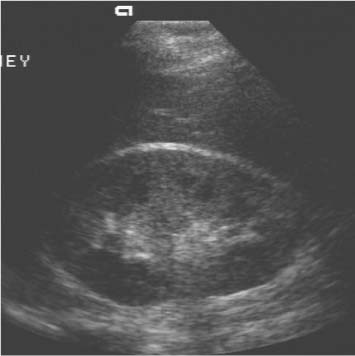
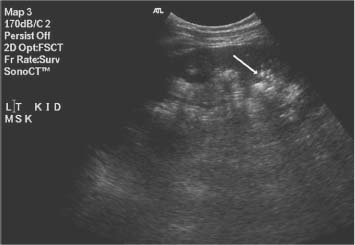
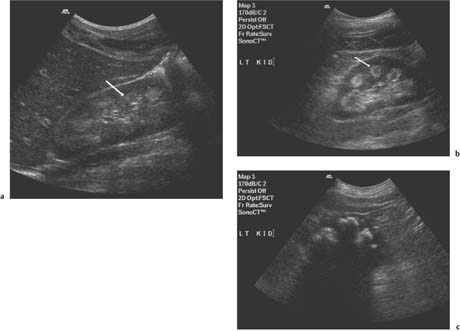
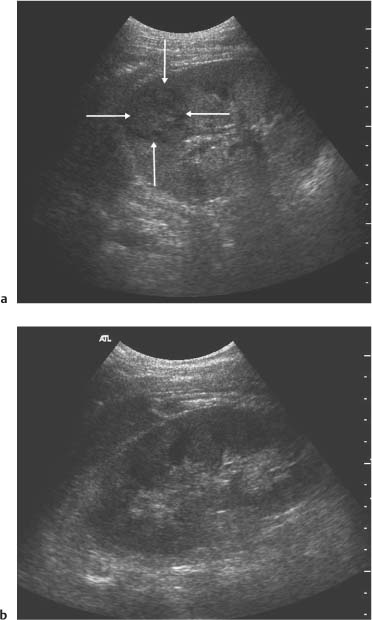
3 Parenchymal Diseases of the Kidney There are a wide variety of conditions which can diffusely affect the renal parenchyma and produce varying degrees of renal impairment. In patients with acute renal failure (ARF), the primary role of ultrasound is to exclude obstruction as a cause of the failure, which will be the case in about 5–10% of cases.1 For the remainder, the diagnosis will depend upon a combination of the clinical situation, biochemical findings, and, in a proportion of patients, renal biopsy. Many of these disorders do not produce specific changes in the appearances of the kidneys on ultrasound, but analyzing any changes that are seen, in the light of the clinical situation, may allow a differential diagnosis to be made. Some disorders affect mainly the cortex, others affect primarily the medulla, and some may affect the parenchyma as a whole. During the ultrasound examination, attention should be given to each of the components of the kidney. Renal size and overall appearance: In some acute disorders, swelling and edema of the kidney may be seen, although in most cases the size of the kidneys is within normal limits. In chronic renal disease the kidneys tend to contract in size, but this is variable and there is little or no correlation with either renal function, or the cause of renal disease.2 These small, relatively echogenic kidneys may be difficult to distinguish within the retroperitoneal fat and may only be identified through their movement on respiration (Fig. 3.1). In some patients with end-stage renal disease (ESRD), changes of acquired cystic disease of the kidney may be seen and parenchymal calcification can be seen with some disorders. Cortex: The appearances of the cortex may be within normal limits, or show an abnormal increase or decrease in echogenicity: Normal renal echogenicity is less than that of the adjacent liver or spleen (assuming that these are normal). In some acute conditions edema and swelling of the cortex are seen. Cortical scarring may be seen (Fig. 3.2); this is distinguished from fetal lobulation as it is usually opposite a calyx or pyramid, whereas the cortical “defects” in fetal lobulation lie between pyramids. Cortical calcification can occur but it is less common than medullary, or collecting system calcification. Fig. 3.1 a A kidney measuring 6.8 cm in length in a patient with chronic renal disease. b A smaller kidney that is more difficult to identify in another patient (arrows); it is easier to appreciate in real-time as it moves with respiration Medulla/pyramids: The appearances of the pyramids can vary, depending upon a number of factors relating to the medullary tissues, or to the adjacent cortex. Normal pyramids are of lower echogenicity than the adjacent cortical tissue and are evenly spaced around the margins of the renal sinus. In acute cortical disorders the pyramids may be more visible due to greater contrast between them and the increased cortical echo-genicity. In other cases there may be a decrease in corticomedullary differentiation. In patients with a primary abnormality of the medulla, the pyramids may be enlarged and hypoechoic, as sometimes seen in acute tubular necrosis (ATN), or show increased echogenicity, as in cases of nephrocalcinosis. Fig. 3.2 a A small scar at the upper pole of the right kidney (arrow). b More marked scarring at the lower pole of another right kidney (arrow) Fig. 3.3 A diffusely echogenic kidney with loss of corticomedullary differentiation in a patient with acute glomerulonephritis Renal sinus: The region of the renal sinus appears as an area of increased echogenicity in the central part of the kidney because of the fat that it contains surrounding the major vessels. It is not usually involved in diffuse parenchymal disease but may be reduced in size with severe parenchymal edema, or show a relative increase in size with parenchymal atrophy. The components of the collecting system are more prominent in cases of ureteric obstruction and calyceal prominence can be noted in cases of papillary necrosis. Renal vessels: Color and spectral Doppler provide information on local conditions in the vessels at the point of interrogation, as well as indirect evidence on the peripheral circulation in the kidney from changes to the spectral waveform. There are several inflammatory and noninflammatory pathologies which can affect the glomerulus and interfere with its function; many have an autoimmune component. Clinically, they result in hematuria and proteinuria and a variable degree of renal impairment. There are no specific features on ultrasound and the kidneys will appear normal in many cases. In some cases there is a variable increase in the cortical echo-genicity (Fig. 3.3). This is nonspecific and there is no relationship with the pathological type, degree of renal impairment, or prognosis; there is some correlation between cortical echogenicity and the degree of inflammatory infiltrate or fibrosis found on histology but this is of little clinical value.3 In acute cases the kidneys may show some enlargement and decreased corticomedullary differentiation with a tendency to shrink if chronic disease develops. This is a rare cause of ARF in the West but is more common in India and Eastern Asia.4 Classically it is associated with abruptio placentae, or postpartum hemorrhage but it can also occur with shock, sepsis, snake bites, and exposure to toxins. There is microvascular thrombosis, and a diffuse intravascular coagulopathy develops resulting in cortical ischemia, except for a thin subcapsular rim of tissue supplied by capsular vessels. There is loss of the normal corticomedullary differentiation and ill-defined patchy areas of increased echogenicity within the parenchyma; the peripheral, perfused rim of parenchyma may be distinguished as an hypoechoic, subcapsular zone (Fig. 3.4).5 Calcium may be deposited at the junction of the ischemic and peripheral zones; this may develop within a few days from the onset of renal failure. Acute interstitial nephritis usually results from an immunologically-induced hypersensitivity reaction to a drug or infective antigen.6 Rarely, it is associated with sarcoid, systemic lupus erythematosus, or malignancy. Histologically, there are diffuse inflammatory infiltrates in the interstitial tissues of the kidney. Clinically, it can imitate ATN, as there is usually a degree of tubular dysfunction. On ultrasound, the kidneys may be normal or enlarged, depending on the severity of the condition; the echogenicity of the cortex may be normal or increased and the finding of a markedly echogenic parenchyma in a patient with ARF (Fig. 3.5) should raise the possibility of acute interstitial nephritis.7 Cortical nephrocalcinosis is rare compared with medullary nephrocalcinosis. Focal calcifications can occur following trauma, infarction, or infection; they may also be associated with tumors. Diffuse cortical calcification may be seen following acute cortical necrosis, renal transplant rejection, Alport syndrome, and rarely with chronic glomerulonephritis. Following acute cortical necrosis the calcification is characteristically seen at the junction between the viable and necrotic regions; this can be seen as early as six to seven days after the onset of the disorder.5 Fig. 3.4 A kidney with acute cortical necrosis showing generally increased echogenicity but with a thin rim of perfused parenchyma (arrow) Fig. 3.5 A diffusely enlarged, echogenic kidney in a patient with acute interstitial nephritis. However, the appearances are nonspecific and are similar to those seen in several other acute disorders of the renal parenchyma Acute tubular necrosis (ATN) is one of the most common causes for ARF in hospital patients.8,9 The injury is usually due to a combination of cellular ischemia and direct tubular epithelial cell injury induced by nephrotoxins. The tubules are more vulnerable to ischemia because of their relatively high metabolic rate and the relatively poor blood supply with low oxygenation when compared with the more luxuriant flow to the cortex and glomeruli. In addition, sloughing of cells and debris into the tubular lumen leads to obstruction by proteinaceous casts. There are several factors which may contribute to the development of this disorder, including ischemia and hypotension, drugs and toxins, sepsis and myoglobinuria/hemoglobinuria. In any individual case, more than one factor is often involved. Fig. 3.6 Echogenic foci in the region of the medullary pyramids (arrow) in a patient with medullary sponge kidneys. A little patchy shadowing is visible but this is not a strong feature The kidneys often appear normal in these patients, and the main role of ultrasound is to exclude obstruction as a cause of the renal failure. Changes which may be identified on ultrasound include diffuse swelling of the kidneys, increased prominence of the medullary pyramids due to edema, or some increase in cortical echogenicity with preservation of corticomedullary differentiation. Doppler ultrasound shows an increase in the resistive index (RI) in the intrarenal vessels,10 with those patients who suffer persistent renal dysfunction tending to have higher values than those who recover adequate function.11 Renal papillary necrosis is part of a spectrum of chronic tubulointerstitial nephritic disorders in which there is progressive scarring and fibrosis of tubulointerstitial structures. This is the final common pathway to ESRD for many disorders, including analgesic nephropathy, diabetes, vesicoureteric reflux (VUR), sickle cell disease, sarcoid, chronic transplant rejection, and heavy-metal poisoning. Over 50% of cases in the United Kingdom are associated with diabetes. In renal papillary necrosis, ultrasound will not show any changes until the disease is advanced and the papillae have sloughed away producing small round, or triangular cystic areas at the site of the missing papilla. Calcification of affected papillae can occur, particularly in cases of analgesic nephropathy, resulting in echogenic foci in the region of the papillae, described as a “garland pattern” around the renal sinus.12 Sloughed papillae may result in ureteric obstruction and dilatation of the pelvicalyceal system.13,14 The cause of medullary sponge kidney is uncertain, but it is thought that it may be a minor developmental anomaly, and links with other disorders such as Caroli disease, Marfan syndrome, and Ehlers–Danlos syndrome have been reported.15 In medullary sponge kidney there are ectatic and cystic changes affecting the medullary collecting ducts; small calculi can form within these dilated segments. Ultrasound may show small, calcific foci in the region of the pyramids but it may just show increased echogenicity of the pyramids if the calculi are too small to show up as distinct entities (Fig. 3.6). There is a wide range of disorders which result in medullary nephrocalcinosis. Raised serum calcium, phosphate, or an alkaline urine can result in nephrocalcinosis. The commonest causes are primary hyper-parathyroidism and renal tubular acidosis, although any disorder leading to hypercalcemia may be implicated and a list of these is given in Table 3.1.16 Frusemide therapy is recognized as a cause of nephrocalcinosis in infants and recent reports suggest that nephrocalcinosis may also be seen in adults who are abusing frusemide to lose weight or reduce edema.17 The calcification is predominantly medullary in location but may rarely be seen within the cortex. Ultrasound has been shown to be reliable in the detection and grading of nephrocalcinosis in children.18 The calcified areas appear on ultrasound as regions of increased echo-genicity in the medulla. Initially the calcium deposits may appear as rings around the periphery of the medullary pyramids;19 with more marked deposition of calcium, the whole pyramid is involved (Fig. 3.7). Acoustic shadows may be absent if the calcium depositions are individually small in diameter in relation to beam width; with more marked degrees of calcification, acoustic shadowing can be variable in prominence. Hyperechogenic medullae are not always the result of nephrocalcinosis and may be seen in patients with gout (where urate deposits and the associated interstitial nephritis are responsible) and in primary hyper-aldosteronism in which chronic hypokalemia results in changes within the tubules and adjacent interstitial tissues.
Introduction
Cortical Disorders
Glomerulonephritis
Acute Cortical Necrosis
Acute Interstitial Nephritis
Cortical Nephrocalcinosis
Disorders of the Medulla/Pyramids
Acute Tubular Necrosis
Renal Papillary Necrosis
Medullary Sponge Kidney
Nephrocalcinosis
Medullary |
|
|
Renal tubular acidosis |
|
|
Oxalosis* |
|
|
Papillary necrosis |
|
|
Medullary sponge |
|
|
kidney |
|
|
Frusemide abuse |
|
|
Bartter Syndrome |
|
|
Hypercalcemia | Primary hyperparathyroidism | Hypercalcemia of malignancy |
| Sarcoid | Cushing disease |
| Vitamin D excess | Milk-alkali syndrome |
Cortical |
|
|
Renal cortical necrosis |
|
|
Chronic glomerulonephritis |
|
|
Transplant rejection |
|
|
Tuberculosis |
|
|
Oxalosis* |
|
|
* Oxalosis typically causes both cortical and medullary nephrocalcin
Fig. 3.7 a Early nephrocalcinosis identified as echogenic pyramids (arrow). b More marked changes showing as rings and echogenic pyramids (arrow). c Marked nephrocalcinosis in a patient with end-stage renal disease (ESRD)
Summary points:
• In patients with ARF, obstruction will be responsible in 5–10%
• Ultrasonic features of glomerulonephritis are variable and are often normal
• RI maybe of prognostic significance in patients with ATN
• Renal papillary necrosis is associated with diabetes in approximately 50% of cases in the UK
Renal Infections
A spectrum of infective disorders can affect the kidneys. The commonest form is acute bacterial pyelonephritis, but infection can be associated with obstruction, calculi, reflux, diabetes, immunosuppression, catheters, or hematogenous spread. The infection can affect primarily the parenchyma, or the collecting system, or both.
Fig. 3.8 a Focal lobar nephronia at the upper pole of a kidney (arrows). b Six weeks later this area has returned to normal
Pyelonephritis
Acute bacterial pyelonephritis is the most common renal infective disorder, occurring more commonly in females. These infections usually respond to antibiotics, have no lasting sequelae, and patients do not usually attend for ultrasound examinations. Ultrasound is performed in patients who do not respond to therapy, have a severe infection, or who are considered to have a possible structural or functional abnormality which may predispose to infection. Even in severe infections, the kidneys may look normal on ultrasound.20 Diffuse or focal swelling of the kidney may be seen; evidence of a predisposing factor, including calculi and/or obstruction, may be discovered. The parenchymal echo pattern may be patchy in appearance due to focal areas of edema and inflammatory infiltration, or even focal hemorrhagic necrosis.21
In some patients focal areas of reduced echogenicity may be identified, corresponding to marked focal inflammatory change; these may resolve with appropriate antibiotic therapy, or may go on to frank abscess formation. These focal inflammatory areas are sometimes called focal lobar nephronia, or acute focal pyelonephritis. The appearances on ultrasound are variable; the affected kidney may be generally enlarged and focal areas of increased, reduced, or mixed echoes may be seen within it (Fig. 3.8). Focal areas of low echogenicity characteristically have no evidence of acoustic enhancement distally; some mass effect can also be apparent.22,23
Renal Abscess
Occasionally, renal parenchymal infections will progress to abscess formation; other abscesses may result from hematogenous spread of infection, particularly with Staphylococcus aureus. 24 Diabetes, renal calculi, and ureteric obstruction are also predisposing factors for the development of renal abscesses.25
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree