7 The Urogenital Tract: Surgical Overview The current chapter predominantly outlines the role of ultrasound, as well as other imaging modalities, in different urological clinical settings, emphasizing the uro-logical surgeon’s perception of the status of imaging in diagnosis and management. Urinary tract infections (UTIs) are a common urological problem, with the importance lying in the identification of those at risk of developing severe sequelae whilst avoiding investigation in patients with uncomplicated infections. Therefore it is useful to broadly divide UTIs into “uncomplicated” and “complicated,” which dictates the extent of evaluation and the duration of treatment and also acts as an indicator of potential treatment failure (Table 7.1). UTIs are classified according to the dominant clinical symptom: Urethritis, cystitis, prostatitis, or pyelonephritis.1 Clinical signs alone will not differentiate the site of the infection into an upper or lower urinary tract infection.2 A UTI in a male patient is regarded as complicated, as in the young and the elderly male it is often associated with structural or outflow-related abnormalities. A UTI in a male patient between age 15 and 50 is uncommon.1 In females an uncomplicated UTI may be cystitis, urethritis, or pyelonephritis, with cystitis the most common. The UTI may be entirely asymptomatic or associated with symptoms of urgency, hesitancy, and pain. Common bacterial pathogens include E. coli (70–95%), Staphylococcus saprophyticus, Proteus, and Klebsiella. The probability of a UTI in a female patient is high, namely 20–35% between age 20 and 40, 10–20% of which may acquire recurrent uncomplicated infections.1 Those patients with pyelonephritis in addition to cystitis have classical symptoms of fever, loin pain, and renal angle tenderness, but the absence of upper tract symptoms does not exclude pyelonephritis.
Introduction
Urinary Tract Infections
Uncomplicated Urinary Tract Infection
• Male gender |
• Elderly |
• Pregnancy |
• Recent urinary tract infection |
• Nosocomial infection |
• Indwelling urinary catheter |
• Recent antibiotic therapy |
• Symptoms for more than seven days |
• Functional or anatomical anomaly of the urinary tract |
• Diabetes mellitus |
• Immunosuppression |
Complicated Urinary Tract Infection
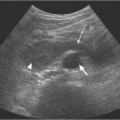
Investigation beyond a posttreatment urine analysis is unnecessary for uncomplicated UTI. With acute, un-complicated pyelonephritis, a white cell count, C-reactive protein, and blood cultures for bacteriology are necessary. An abdominal radiograph and ultrasound should be performed in both uncomplicated and complicated UTI. The plain radiograph may identify radio-opaque calculi or in rare instances demonstrate gas in the renal parenchyma, which is indicative of emphysematous pyelonephritis, or obliteration of the psoas muscle outline, a nonspecific sign of a perirenal abscess formation. An ultrasound examination is useful in excluding the presence of an abscess or pelvicalyceal obstruction. However, in some patients the upper urinary tract may be dilated in the absence of obstruction, attributed to “bacterial virulence factors.”
Routine use of an intravenous urography (IVU) in uncomplicated UTI is not recommended as over 75% of patients will have a normal study.3 However, if there is no clinical improvement within 72 hours of antibiotic therapy, further imaging is indicated. Computed tomography (CT) is the best option in such situations, as it offers greater anatomical detail of the renal parenchyma and the perirenal areas.
Emphysematous pyelonephritis is a rare, acute narcotizing condition caused by any “gas-forming” organism.4 Diabetes mellitus, immunosuppression, urolithiasis, and renal tract obstruction are common predis-posing conditions. These patients are often severely ill and may fail to respond to antibiotic therapy. Ultra-sound and CT imaging are the most useful methods of assessment, with CT imaging offering accurate anatomical detail, including areas of gas formation. Any perinephric infected fluid collection may be percutaneously drained under ultrasound guidance. There is a reported mortality rate of 50–60% in emphysematous pyelonephritis, with nephrectomy the only viable option for cure.4
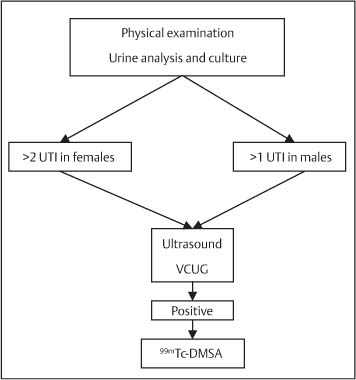
Fig. 7.1 Investigation of a child with a urinary tract infection (UTI) (VCUG = voiding cystourethrography; 99mTc-DMSA = 99mTcdimercaptosuccinic acid)
Urinary Tract Infection in Children
UTIs are the commonest bacterial infection in children. Clinical presentation in the pediatric age group may be variable and would include failure to thrive, episodes of fever, vomiting, and dehydration.1 Investigations are usually undertaken following the first episode of infection in male patients and following the second episode in female patients. The main objective of evaluating these patients is to exclude an underlying structural or functional anomaly that may be corrected. A diagnostic algorithm for the management of UTIs has been formulated by the European Association of Urology (Fig. 7.1).1
Ultrasound is the preferred imaging modality in the pediatric age group; there is the benefit of absence of ionizing radiation and adverse effects related to the use of iodinated contrast media. Ultrasound has been shown to be as accurate as IVU in detecting significant renal anomalies.5,6
In neonates and in children between age two and 18 years with a UTI, approximately 30% will have vesicoureteric reflux (VUR).5 Male patients with VUR are likely to be diagnosed in infancy, whereas in female patients the diagnosis is delayed until between age two and eight years. Although VUR will resolve spontaneously in the majority of patients, UTI associated with reflux facilitates ascending infection, resulting in possible renal parenchyma cortical damage and resultant scarring. The recommended investigation for VUR in children less than age one year is a voiding cystourethrography using low-dose fluoroscopy. Indirect cystography, using either99mTc-mercaptoacetyltriglycine (99mTc-MAG3) or99mTc-diethylenetriamine pentaacetic acid (99mTc-DTPA) renography with reflux studies is a suitable option for older children.1 Reflux may also be assessed using microbubble ultrasound contrast media, avoiding the use of ionizing radiation.7 A99mTc-dimercaptosuccinic acid (99mTc-DMSA) radioisotope scan is performed to identify renal scarring in those patients with VUR, as ultrasound is not as sensitive in the detection of renal parenchyma abnormalities.1 The presence of renal scarring due to VUR is a relative indication for corrective surgery. This may, however, not necessarily prevent recurrence of lower UTIs. The role of IVU is limited to those who require greater anatomical detail of the upper renal tract.
Summary points:
• Divided into “uncomplicated” and “complicated” which dictates extent of evaluation and duration of treatment
• Classified into urethritis, cystitis, prostatitis, or pyelonephritis
• UTI in a male patient is regarded as complicated
• Probability of a UTI in a female patient is high
Urinary Calculus Disease
The average life-time risk of renal calculus disease is 5–10%.8 Ureteric colic is a frequent cause of emergency urological referral, with the patient presenting with characteristic radiating loin pain, vomiting, a low-grade fever, and microscopic hematuria. The differential diagnosis is wide and includes biliary colic (on the right), peptic ulcer disease, pancreatitis, and abdominal aortic aneurysm. Urinary calculi are the commonest cause of renal colic, although sloughed renal papillae, a blood clot, and fungal debris are occasionally implicated. Investigations for renal colic that follow clinical diagnosis include analysis of urine (for red cells, leukocytes, nitrites, pH and cystene), urine culture, serum creatinine, calcium, albumin, urate, and a white cell count.9 Appropriate imaging will immediately confirm the diagnosis and help to decide on management. The choice of imaging includes a plain abdominal radiograph combined with an ultrasound examination, IVU, or an unenhanced CT examination.
CT is likely to be the best single imaging investigation for ureteric calculi, as both radiolucent and radio-opaque calculi are visualized. However, in a number of countries, including the United Kingdom, the investigation of choice in renal colic remains an IVU, a situation that is likely to change to CT imaging in the future. IVU offers accurate assessment of the calculus size, site, and the degree of renal obstruction. The absence of hydro-nephrosis on ultrasound does not reliably rule out renal obstruction. Over 80% of calculi < 4 mm in diameter will pass spontaneously, but this is less likely with larger calculi.10 Patients with persistent pain, UTI, risk of pyonephrosis, renal failure, single kidney, or bilateral obstruction need urgent decompression irrespective of calculi size.
Nonobstructing calculi in the kidney may be detected incidentally, or be imaged as a consequence of investigations for pain, UTI, or hematuria. Ultrasound and CT imaging may be useful in the evaluation of radio-lucent calculi.99mTc-DMSA scintigraphy is an optional investigation used to assess the differential renal function, especially in the presence of a large “stag-horn” calculus. A complete set of investigations for calculus disease, which comprises serum creatinine, uric acid, calcium, phosphate, and a 24-hour urine assessment, is recommended for patients at high risk of forming further calculi (indicated by a positive family history or previous episodes of calculus disease). Treatment options for renal calculi include extracorporal shock-wave lithotripsy (ESWL), percutaneous nephrolithotomy (PCNL), and endoscopic surgery; open surgery is now almost never necessary for calculus disease.9
Bladder calculi may be formed de novo in the bladder or migrate from the upper urinary tract. Bladder calculi are now rare, with primary bladder calculi occurring most often as a consequence of structural bladder abnormalities such as diverticula or voiding dys-function.9 Patients usually present with lower urinary tract symptoms (LUTS), suprapubic pain, or associated complications such as UTI, hematuria, or urinary retention. A plain abdominal radiograph often reveals the bladder calculi; an ultrasound is helpful in assessing any underlying abnormality such as residual urine and diverticula. Male patients should also have urine flow rate measured, as bladder outflow obstruction is a common cause of bladder calculi formation. Management includes removal of the calculi (usually by cystoscopy) and treating the underlying cause.
Summary points:
• Ureteric colic is a frequent cause of emergency urological referral
• CT is the best single imaging investigation for ureteric calculi
• Persistent pain, pyonephrosis, renal failure, single kidney or bilateral renal obstruction needs urgent decompression
• Bladder calculi are rare, occurring as a consequence of structural bladder abnormalities
Upper Urinary Tract Obstruction (Hydronephrosis)
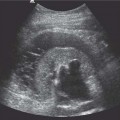
Upper urinary tract obstruction may result from a variety of causes (Table 7.2). The presentation and natural history may vary according to the degree of obstruction. Acute upper urinary tract obstruction is often due to calculi with sloughed papillae, blood clots, renal pelvis debris, and retroperitoneal abnormalities also implicated. Chronic upper urinary tract obstruction may be further divided into equivocal (doubtful) or unequivocal. Those with equivocal obstruction may have IVU and/or ultrasound appearance of hydronephrosis or clinical symptoms such as diuresis-induced loin pain, but fail to demonstrate evidence of obstruction on99-mTc-MAG3 renography.11 The main clinical concern with both acute and chronic upper urinary tract obstruction is the risk of any resulting renal injury causing loss of renal function.
In children pelviureteric junction (PUJ) obstruction is the commonest cause of upper tract obstruction.11 Congenital PUJ obstruction may be visualized on ultrasound as early as the second trimester of pregnancy with antenatal imaging. Not all of the PUJ obstructions present on the antenatal ultrasound examination will persist after birth.11 A follow-up ultrasound examination will identify those with a persisting PUJ obstruction and allow institution of the correct management. Older children with upper urinary tract obstruction may present incidentally with pain, infection, a palpable renal mass, renal impairment, hematuria, and hypertension.11 Dhillon et al. have demonstrated the risk of surgical correction according to the degree of hydronephrosis measured as the anteroposterior diameter of the renal pelvis (Table 7.3).11,12 The indications for surgical correction may be further refined when combined with differential renal function ascertained with99mTc-MAG3 renography.11 In neonates, 99mTc-MAG3 renography may be inaccurate due to renal immaturity, with99mTc-MAG3 renography best performed at age six weeks.11 IVU in this setting may not add any further information and is therefore not routinely recommended.
•Pelviureteric junction (PUJ) obstruction |
• Congenital megaureter |
• Ureterocele |
• Vesicoureteric junction (VUJ) obstruction |
• Ureteric stricture |
• Ureteric tumor |
• Pregnancy |
• Tuberculosis |
• Bilhaziasis |
• Ureteric duplication |
• Retroperitoneal pathology |
– Retrocaval ureter |
– Abdominal aortic aneurysm |
– Retroperitoneal fibrosis |
• Bowel/other malignancy extending into the retroperitoneum |
Anteroposterior diameter of therenal pelvis (mm) | Risk of requirement for pyeloplasty (%) |
<15 | 2 |
15–20 | 7 |
20–30 | 29 |
30–40 | 61 |
40–50 | 67 |
> 50 | 100 |
Surgical correction is recommended when the divided renal function is < 40% and the anterior–posterior diameter of the PUJ is > 30 mm on the affected side.11 An Anderson–Hynes pyeloplasty is the standard surgical procedure for PUJ obstruction. When there remains uncertainty, for example when the divided renal function is > 40% and the anterior–posterior diameter of the PUJ is < 30 mm, a conservative approach is usually adopted; the majority of these patients improve or remain stable.
The management of a poorly or nonfunctioning kidney (divided renal function < 15%) is more controversial; recommendations vary from an initial nephrostomy to monitor any functional improvement to immediate nephrectomy.11 A clinical decision must be based on serial functional assessment with 99mTcMAG3 renography. When bilateral PUJ obstruction is present, it is important to exclude voiding dysfunction as a cause. Radioisotope scans are unreliable in patients with bilateral PUJ obstruction.
In adults, unilateral hydronephrosis may be a result of an undetected, congenital PUJ obstruction. However, it is important to exclude other acquired causes, as 5% of transitional cell carcinomas (TCCs) occur in the upper urinary tract and may present as urinary tract obstruction. IVU is performed more frequently in adults than in children with upper urinary tract obstruction along with other investigations such as urine cytology, retrograde pyelography, and CT or magnetic resonance (MR) imaging of the retroperitoneum.
Bilateral upper urinary tract obstruction in the elderly male patient occurs most frequently due to high-pressure chronic retention as a result of bladder out-flow obstruction. Locally advanced bladder, prostate, or gynecological tumors will occasionally cause bilateral ureteric invasion, resulting in upper urinary tract obstruction with the patients usually presenting with acute renal failure. Percutaneous nephrostomy insertion under radiological guidance is the initial management, although it is best practice to insert either a double-J stent or a metallic stent across the obstruction as soon as possible.
Summary points:
• Due to calculi, sloughed papillae, blood clots, renal pelvis debris, and retro-peritoneal abnormalities
• Acute and chronic upper urinary tract obstruction may result in loss of renal function
• Percutaneous nephrostomy insertion is possible, although it is best practice to insert a double-J stent across the obstruction as first line management
• In children PUJ obstruction is common
Hematuria
The presentation of hematuria may vary from microscopic hematuria detected on urine analysis, visible blood in the urine, to blood clots causing urinary retention. The importance of careful assessment in these patients cannot be underestimated; underlying urinary tract pathology occurs frequently. Urinary tract malignancy is found four times more often in patients with macroscopic than those with microscopic hematuria; studies have suggested a 5% and 22% incidence of bladder carcinoma with microscopic and macroscopic hematuria, respectively.13–15
There are a number of causes for hematuria which include both medical and surgical diseases. Glomerulonephritis, UTIs, and childhood tumors are the main differential diagnoses in the pediatric age group. Uri-nary stones, trauma, atypical glomerulonephritis, UTI, and IgA nephropathy should be considered in young adults, whereas over age 40 urinary tract malignancy is the main concern. Complications of benign prostatic hypertrophy, such as UTI and bladder calculi, are also a common cause in older men.
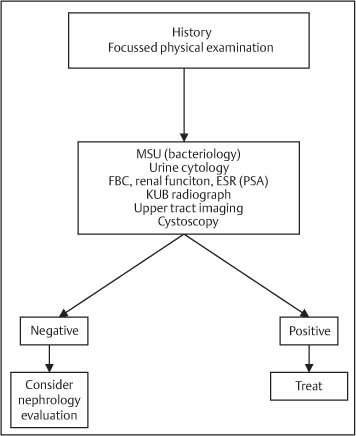
Clinical assessment of patients with any form of hematuria includes a focused history and examination, urine testing, cystoscopy, and urinary tract imaging (Fig. 7.2). There remains some dispute as to the benefits of investigating microscopic hematuria in younger patients to the same extent as macroscopic hematuria in all age groups; the risk of cancer in young patients with microscopic hematuria is low.14,16 However, a study has suggested significant disease in 18.5% of patients with microscopic hematuria, which implies that a thorough evaluation is always indicated.14
Fig. 7.2 Investigation of hematuria
(MSU = midstream urine; FBC = full blood count; ESR = erythrocyte sedimentation rate; PSA = prostate-specific antigen; KUB= kidneys, ureter, bladder)
From the clinician’s point of view, the most appropriate technique for imaging the upper renal tracts remains debatable. Ultrasound is the favored option as it is as accurate as an IVU for hydronephrosis and superior at imaging bladder tumors.1,16 When further imaging is indicated, CT has the advantage of tumor staging and detecting nonurological causes of hematuria.13 The most reliable method of assessing the lower urinary tract is cystoscopy.17 If a bladder tumor is demonstrated on cystoscopy, a transurethral resection (TUR) of the tumor for histological confirmation and staging may be performed. The presence of hydronephrosis, secondary to a tumor at the ureteric orifice, is frequently a sign of muscle-invasive disease.17 Over 90% of bladder tumors are TCCs, of which over 70% are superficial in nature.17 The risk of recurrence varies from 50–70% according to tumor grade.17,18 Superficial TCC may progress to invasive disease; carcinoma in situ and poorly differentiated (Grade 3) tumors pose by far the highest risk. Intravesical therapy, with either mitomycin C or epirubicin, may reduce the frequency of recurrent disease, whereas intravesical bacille Calmette–Guérin (BCG) reduces the overall recurrence rate, as well as the risk of tumor progression.19 Patients are monitored with regular cystoscopy to detect and treat any tumor recurrence. When muscle-invasive disease is present, staging with CT or MR imaging is performed. A radio-nucleotide bone scan should be requested with advanced disease. Treatment options for invasive bladder carcinoma include radical cystectomy or radiotherapy, with platinum-based chemotherapy being used in advanced disease.
Summary points:
• Causes for hematuria include both medical and surgical diseases
• Urinary tract malignancy is 4x more common in macroscopic than in microscopic hematuria
• There is dispute as to the benefits of investigating microscopic hematuria in younger patients
• Assessment of any form of hematuria includes a focused history and examination, urine testing, cystoscopy, and urinary tract imaging
Lower Urinary Tract Symptoms and Benign Prostatic Hyperplasia
Lower urinary tract symptoms (LUTS), affecting up to 30% of males over age 65, are a heterogeneous group of symptoms related to the bladder and bladder outflow, further divided into storage (filling) or voiding (obstructive) symptoms (Table 7.4). There are numerous causes for LUTS (Table 7.5), of which benign prostatic hyperplasia (BPH) is the commonest. The aim of assessing patients with BPH/LUTS is to establish the diagnosis, identify the cause, and manage treatment appropriately (Fig. 7.3). Patients with troublesome symptoms have a substantial improvement in quality of life following successful treatment.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree