Breast magnetic resonance (MR) imaging is increasingly performed for a variety of indications, most commonly with the goal of detecting breast cancer. Percutaneous biopsy (usually under MR guidance or ultrasound if there is a correlating finding) is commonly used to evaluate suspicious imaging findings detected on MR imaging with the goal of identifying malignancy. It is important to be familiar with the characteristics and management of high-risk lesions detected or biopsied under MR guidance. This review focuses on the appearance of a variety of breast lesions detected on MR imaging that require excision with focus on pathologic correlation.
Key points
- •
Morphologic features of masses seen on magnetic resonance (MR) imaging, such as spiculated margins and rim enhancement, are highly predictive of malignancy.
- •
High-risk lesions found on MR-guided biopsy may be upgraded to malignancy at surgical excision; however, upgrade rate and management of these lesions should be addressed on a lesion-by-lesion basis.
- •
A clearly thought-out MR-guided biopsy technique with checks for appropriate and accurate targeting of lesions, including postbiopsy marker clip placement, is crucial for evaluating radiologic-pathologic correlation.
- •
There are no morphologic or kinetic MR imaging characteristics that have been shown to definitively predict high-risk lesion upgrade.
- •
Atypical ductal hyperplasia is the most frequently encountered high-risk lesion encountered on MR-guided biopsy and has the highest likelihood of being upgraded to malignancy of the high-risk lesions.
Introduction
Breast magnetic resonance (MR) imaging is increasingly being performed for a variety of indications, most commonly with the goal of detecting breast cancer. Percutaneous biopsy (usually under MR guidance or under ultrasound if there is a correlating finding) is commonly used to evaluate suspicious imaging findings detected on MR imaging with the goal of identifying malignancy. In addition to invasive and in situ breast cancer, there are several other lesions that are generally excised when encountered on percutaneous biopsy; this group includes discordant lesions and high-risk lesions. Discordant lesions are those where pathologic abnormality is not consistent with the expected result based on the radiologic appearance and excision is advised to exclude the possibility of a missed cancer. High-risk lesions include atypical ductal hyperplasia, lobular carcinoma in situ, atypical lobular hyperplasia, radial scar, papillary lesions, and flat epithelial atypia; excision is often recommended for these lesions because of the possibility of their upgrade to malignancy when more tissue is sampled.
In addition to understanding imaging characteristics and management of malignancy detected on MR imaging, it is also important to be familiar with the characteristics and management of high-risk lesions detected or biopsied under MR guidance. In this review, the appearance of a variety of breast lesions detected on MR imaging that require excision with a focus on pathologic correlation is described. In particular, the focus is high-risk lesions detected on MR imaging with a review of the pathologic features of these lesions. The current work on high-risk lesions seen on MR imaging is also reviewed and appropriate management of these lesions is discussed.
Introduction
Breast magnetic resonance (MR) imaging is increasingly being performed for a variety of indications, most commonly with the goal of detecting breast cancer. Percutaneous biopsy (usually under MR guidance or under ultrasound if there is a correlating finding) is commonly used to evaluate suspicious imaging findings detected on MR imaging with the goal of identifying malignancy. In addition to invasive and in situ breast cancer, there are several other lesions that are generally excised when encountered on percutaneous biopsy; this group includes discordant lesions and high-risk lesions. Discordant lesions are those where pathologic abnormality is not consistent with the expected result based on the radiologic appearance and excision is advised to exclude the possibility of a missed cancer. High-risk lesions include atypical ductal hyperplasia, lobular carcinoma in situ, atypical lobular hyperplasia, radial scar, papillary lesions, and flat epithelial atypia; excision is often recommended for these lesions because of the possibility of their upgrade to malignancy when more tissue is sampled.
In addition to understanding imaging characteristics and management of malignancy detected on MR imaging, it is also important to be familiar with the characteristics and management of high-risk lesions detected or biopsied under MR guidance. In this review, the appearance of a variety of breast lesions detected on MR imaging that require excision with a focus on pathologic correlation is described. In particular, the focus is high-risk lesions detected on MR imaging with a review of the pathologic features of these lesions. The current work on high-risk lesions seen on MR imaging is also reviewed and appropriate management of these lesions is discussed.
Imaging findings and pathologic abnormality
Malignancy
Invasive carcinoma
The MR imaging characteristics of invasive breast cancer presenting as a mass have been well described. In addition to lesion size, certain specific morphologic features, such as smooth or lobulated margins with no or low enhancement, can be predictive of benign disease. The presence of nonenhancing internal septations in a smooth or lobulated mass is highly specific for the diagnosis of fibroadenoma (specificity 93%–97%). In contrast, certain morphologic features are very suggestive of malignancy, such as spiculated margins (positive predictive value = 76%–88%) and rim enhancement (positive predictive value = 79%–92%). Schnall and colleagues noted that the most important feature was the presence of enhancement of any type; however, the absence of enhancement did not exclude invasive cancer. As Gutierrez and colleagues demonstrated, masses of 1 cm or larger with heterogeneous enhancement and irregular margins have a high (68% in their study) probability of malignancy. On the other hand, masses of 1 cm or larger with smooth margins and homogeneous enhancement have a very low probability of malignancy (3% in their cohort). Mahoney and colleagues recently analyzed the cancers detected in the ACRIN 6667 trial and noted that masses with irregular shapes and irregular or spiculated margins had the highest likelihood of malignancy.
As has been well demonstrated, kinetic analysis may also aid in cancer detection. Qualitative assessment of kinetic curve parameters is used in most clinical scenarios with initial enhancement defined as slow, medium, or rapid and the shape of the time signal intensity curve defined as type I persistent, type 2 plateau, and type 3 wash-in/wash-out curves. The curves indicating likelihood of benignity or malignancy were shown in the 1999 study by Kuhl and colleagues of signal intensity time course data; a type 3 curve should raise suspicion for a malignant lesion. However, there is overlap within these categories and kinetics should not be used independently of morphology. Mahoney and colleagues showed that rapid initial enhancement was also associated with carcinomas. Currently, the assessment of the morphologic features is more predictive in the diagnosis of breast cancer than characterization of the kinetic curves.
In situ carcinoma
Recent studies evaluating detection of DCIS on MR imaging have shown that MR imaging is the most sensitive modality currently available for identifying DCIS and is more accurate in evaluating the extent of DCIS than mammography. MR imaging is particularly sensitive for identifying high-grade and intermediate-grade DCIS. The imaging appearance of ductal carcinoma in situ (DCIS) on MR imaging has been well described. DCIS may vary in terms of morphology, but nonmass enhancement (NME) morphology is the most common presentation. Rosen and colleagues reported that the most common pattern of NME for pure DCIS was segmental distribution and clumped enhancement. DCIS may also present as a mass on MR imaging, most likely an irregular mass. Although the kinetics of DCIS are variable, a fast uptake and plateau curve are reported as the most common kinetic pattern.
High-risk Lesions
High-risk lesions may demonstrate an upgrade to malignancy after further surgical excision. An initial underestimation at biopsy may occur for a variety of reasons including undersampling at biopsy and difference of opinion among pathologists over the criteria for defining a high-risk lesion versus a cancer. In addition, it is always possible that the targeted concerning finding may have been missed at biopsy. Overall for all types of high-risk lesions, the current literature suggests frequencies of between 3% and 21%. Underestimation rates for all high-risk lesions combined on MR imaging range from 13% to 57%. Given the high potential for underestimation of malignancy, radiologic-pathologic correlation after biopsy is extremely crucial. As well, if the pathologic abnormality appears discordant with radiologic imaging, investigation and repeat biopsy or surgical excision is necessary.
Review of the MR imaging high-risk lesion literature
To the authors’ knowledge, excluding review articles, pictorial essays, technical notes, case reports, non-English articles, and animal studies, there are 27 articles on PubMed reporting frequency of high-risk lesions on MR imaging. Of this group, 20 articles detail frequency and upgrade rates of the lesions and include complete excision data (ie, complete accounting of imaging or surgical follow-up for all lesions) for at least some of the identified types of high-risk lesions. At present, most high-risk data are reported as part of work evaluating outcomes of MR imaging–guided biopsies; only a few studies focus on high-risk lesions outcomes specifically or on outcomes of a specific type of high-risk lesion on MR imaging (for example, one article on atypical ductal hyperplasia [ADH] and one on papillary lesions). There is also a recent review of frequency and underestimation of lobular carcinoma in situ (LCIS) and atypical lobular hyperplasia (ALH), although MR imaging detected and MR-guided biopsied lesions comprise only a subset of the cohort.
Imaging and biopsy techniques
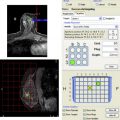
Appropriate MR imaging technique is essential with adequate temporal and spatial resolution needed for optimal assessment of detected lesions. A few of the MR biopsy and high-risk studies have been performed at 3 T, but most of the available studies have been performed at 1.5 T field strengths. The studies are united in using dynamic contrast-enhanced MR sequences and usually include fat-suppression sequences as well as subtraction images but show variation with respect to plane of acquisition, spatial resolution, and kinetic resolution.
Most of the studies evaluate lesions identified and biopsied under MR guidance. However, some studies also include lesions identified on MR but biopsied under other modalities. If a lesion in need of biopsy is detected on MR imaging (BI-RADS 4 or 5), it is the authors’ practice to recommend targeted ultrasound if the lesion is a mass or if is of a large enough size. As several recent articles have discussed, the likelihood of identifying a lesion on targeted ultrasound is greater if it is a mass or of larger size. In the metareview of 13 studies, Leung found that between 23% and 89% of lesions had an ultrasound correlate depending on the study and between 15% and 57% of cancers were identified on second-look ultrasound. Biopsy may then be pursued under ultrasound guidance if there is clear correlation. If there is no mammographic or sonographic correlate, or the lesion is thought unlikely to have a correlate, MR-guided biopsy is recommended.
A clearly thought-out biopsy technique with checks for appropriate and accurate targeting of a lesion is crucial for evaluating radiologic-pathologic correlation. Studies use different means of verifying accuracy of tissue sampling and establishing radiologic-pathologic correlation once biopsy has been performed ( Table 1 ). Most studies rely on postbiopsy markers and postmarker MR and mammographic imaging after biopsy (see Table 1 ). There are also multiple studies where radiologic-pathologic correlation is difficult to prove; for example, if final surgical pathologic abnormality is based on mastectomy without localization of the original lesion (see Table 1 ).
| Study | Biopsy Gauge | Verification of Biopsy Site | Surgical Excision Details and Verification of Surgical |
|---|---|---|---|
| Liberman et al, 2003 | 9 G | Clip and postbiopsy imaging | All to needle localization and surgery |
| Chen et al, 2004 | 14 G | Marker clip in “most patients”; final postbiopsy image | 4/5 ADH 2 surgical excisions 2 mastectomies |
| Lehman et al, 2005 | No data | Clip and postbiopsy imaging | Biopsy cavity in surgical specimen for one upgraded high-risk lesion |
| Liberman et al, 2005 | 9 G | Clip “may be” placed; assessment of degree of removal of target | “Surgery” |
| Orel et al, 2006 | 9 G | 75/85 clip, postbiopsy imaging, mammogram in 20/75 | “Surgical excision or mastectomy” |
| Perlet et al, 2006 | 11 G | Postbiopsy imaging, evaluation of partial or complete removal, radiologic-pathologic review | Surgical excision after ultrasound, mammogram, or MR wire localization of cavity |
| Liberman et al, 2007 | 9 G | Clip and postbiopsy mammogram | “Surgery” |
| Han et al, 2008 | 9 G or 10 G | Clip in all except 13; postbiopsy imaging if clip; radiologic-pathologic review | “Surgery performed” |
| Mahoney, 2008 | 10 G | Clip placed; postbiopsy imaging and mammogram; radiologic-pathologic review | “Surgical excision” |
| Perretta et al, 2008 | 10 G | 35/47 clip Imaging 1 wk post, radiologic-pathologic correlation | Excisional biopsy |
| Malhaire et al, 2010 | 10 G | Clip, postbiopsy MR imaging and mammogram; multidisciplinary radiologic-pathologic review | “Surgical” |
| Noroozian et al, 2010 | 9 G | Clip, postbiopsy mammogram, radiologic-pathologic review | “Surgical” |
| Strigel et al, 2010 | 14-G core needle OR 9 G vacuum-assisted biopsy OR Ultrasound 14-G OR 9-G or 11-G stereotactic | No data | No data |
| Tozaki et al, 2010 | 11 G and 10 G | Clip placed; postbiopsy imaging | “Surgery performed” |
| Crystal et al, 2011 | 9 G | Clip at discretion of radiologist; postclip mammogram; multidisciplinary review of rad-path concordance | All excisional biopsies guided via clip |
| Rauch et al, 2012 | 9 G | Clip placed; postbiopsy imaging | Surgical excision |
| Oxner et al, 2012 | 10 G | Not stated | Final surgical pathologic abnormality compared with MR-guided biopsy lesion |
| Dogan et al, 2012 | 9 G | Clips placed, but not stated if performed for all MR biopsies | Mastectomy or segmentectomy |
Pathology and radiologic-pathologic correlation
Overall the literature is consistent in demonstrating no morphologic or kinetic features that can be used to identify high-risk lesions on MR imaging with the exception of papillary lesions discussed later in this article. In addition, there are no imaging characteristics that have been shown to predict upgrade of a lesion to malignancy. There are some studies that use MR imaging as a problem-solving mechanism to evaluate high-risk lesions identified through biopsy under other modalities and have suggested that a lack of enhancement on MR imaging may suggest lack of underestimation of an invasive malignancy, but more studies are needed to verify this possibility.
Not all high-risk lesions are the same in terms of likelihood of upgrade to malignancy ( Tables 2 and 3 ): upgrade rate and management of these lesions should be addressed on a lesion-by-lesion basis. In the rest of the article, therefore, the focus is on the pathologic abnormality, radiologic-pathologic correlation, and management of specific high-risk lesions when detected on MR imaging.
| Author | Total No. of Lesions | Total High-risk Lesions | Frequency of ADH/Total (%) | No. ADH Excised | ADH Upgrade (%) | Final Surgical Histology |
|---|---|---|---|---|---|---|
| Liberman et al, 2003 | 27 | 1 | 1/27 (3.7) | 1 | 1/1 (100) | DCIS |
| Chen et al, 2004 | 35 | 5 | 5/35 (14) | 4 | 2/4 (50) | 2 IDC |
| Liberman et al, 2005 | 95 | 10 | 4/95 (4) | 4 | 2/4 (50) | 2 DCIS |
| Lehman et al, 2005 | 38 | 2 | 2/38 (5.3) | 2 | 1/2 (50) | DCIS |
| Ghate et al, 2006 | 19 | 4 | 2/19 (11) | 2 | 1/2 (50) | 1 IDC |
| Perlet et al, 2006 | 517 | 17 | 17/517 (3.3) | 17 | 5/17 (29.4) | 5 DCIS |
| Orel et al, 2006 | 85 | 18 a | 8/85 (9.4) | 8 | 2/8 (25) | 2 DCIS |
| Liberman et al, 2007 | 237 | 15 | 15/237 (6.3) | 13 | 5/13 (38.5) | 5 DCIS |
| Mahoney et al, 2008 | 55 | 7 | 3/55 (5.5) | 3 | 2/3 (66.7) | 2 IDC |
| Malhaire et al, 2010 | 72 | 10 | 1/72 (1) | 1 | 1/1 (100) | 1 DCIS |
| Perretta et al, 2008 | 47 | 4 a | 4/47 (8.5) | 4 | 1/4 (25) | 1 DCIS |
| Noroozian et al, 2010 | 75 | 7 | 2/75 (2.7) | 2 | 0/2 (0) | NA |
| Strigel et al, 2010 | 482 | 51 | 51/482 (10.6) | 34 | 11/34 (32.4) | 1 ILC 4 IDC 6 DCIS (1 ADH and LCIS) |
| Tozaki et al, 2010 | 102 | 9 a | 4/102 (4) | 2 | 2/2 (100) | 2 DCIS |
| Crystal et al, 2011 | 161 | 31 | 6/161 (3.7) | 6 | 3/6 (50) | 2 DCIS 1 IC, NOS |
| Rauch et al, 2012 | 218 | 37 a | 13/218 (6) | NS | 4/NS | 3 DCIS 1 IDC |
| Oxner et al, 2012 | 187 | 16 a | 15/187 (8) | NS (data on 3 cases) | 2/NS | 2 DCIS |
| Dogan et al, 2012 | 199 | 31 | NS | NS (data on 2 cases) | 2/NS | 1 DCIS 1 IDC |
| Range (%) | 19–572 | 1–51 | 1–19 | 0–100 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree