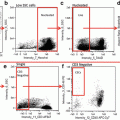
Fig. 1
Glovebox used for sample deoxygenation. The apparatus is shown with the location of valves and gauges for the gas cylinders and vacuum pump indicated
6.
500 μl, 96-well micro-titer plates with adhesive cover.
7.
Table-top centrifuge.
8.
1.5 mL polypropylene tubes.
9.
200 μL and 1 mL pipettes and tips.
2.2 Imaging Flow Cytometry Run and Analysis Components
1.
PBS 1×.
2.
ImageStreamX or MK II imaging flow cytometer (Amnis Corporation, Seattle, WA, USA) equipped with Amnis INSPIRE and Amnis IDEAS softwares.
3.
Amnis Speed Beads suspension.
3 Methods
Carry out all procedures at room temperature unless otherwise specified.
3.1 Blood Sample Collection and Preparation
1.
Collect 4 mL of peripheral venous blood into Vacutainer EDTA tubes. Sample can be stored at 4 °C for up to 24 h.
2.
If sample was stored at 4 °C, place tube on a horizontal shaker at 80–100 rpm until sample is homogenized (approximately 2–5 min).
3.
Pipette 10 μL of whole blood into a 1.5 mL polypropylene tube containing 990 μL of Hemox Buffer and mix. This makes a 1 % suspension of blood.
4.
Pipette 400 μL duplicates of the 1 % suspension into adjacent wells of a 96-well plate. If more than one sample will be incubated in the same plate, place sample doubles one well apart in both directions, and preferably away from the edges of the plate (Fig. 2).
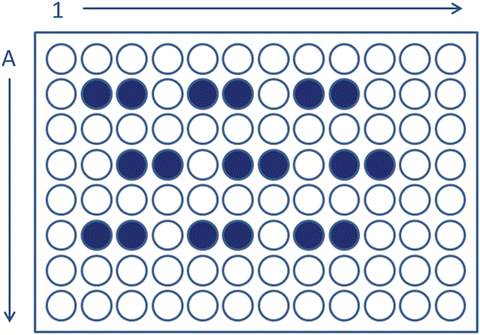

Fig. 2
Schematic representation of sample arrangement in a 96-well plate
5.
Seal the 96-well plate with an appropriate self-adhesive plastic cover.
3.2 Blood Sample Deoxygenation
1.
Before starting, make sure the glovebox is airtight, the gas cylinder contains enough of a 95 % nitrogen/5 % hydrogen mixture, and that the vacuum pump and the oximeter are both operating. Place a 200 μL pipette, 200 μL pipette tips, a new self-adhesive plastic cover, and a proper waste disposal bag for pipette tips and used microplate covers inside the glovebox.
2.
Thaw a vial of frozen glutaraldehyde 25 % to room temperature. Total volume of glutaraldehyde should be at least 10 % of the total sample volumes (i.e., 80 μL for two 400 μL samples).
3.
Decrease the oxygen concentration inside the glovebox to 2 % by injecting N2/H2 mixture and aspirating with the vacuum pump (see Note 1 ).
4.
Place the 96-well plate inside the glovebox on top of a shaker and remove adhesive cover.
5.
6.
Upon opening the glovebox to place the plate and vial, some oxygen may enter the glovebox, so it will be necessary to readjust the oxygen concentration to 2 %.
7.
Incubate the 96-well plate at room temperature and 2 % oxygen for 2 h under constant shaking at 300 rpm.
8.
Once the incubation is done, turn off the shaker and pipette 40 uL of glutaraldehyde 25 % into each well containing 400 μL of 1 % blood suspension. Mix between seven and ten times with the pipette to sufficiently mix the glutaraldehyde and incubate for 5–10 min.
9.
Reseal the plate with a new cover seal and remove from glovebox.
10.
Transfer the contents of each well to a properly identified 1.5 mL polypropylene tube.
11.
Wash each sample by adding 1 mL of PBS 1×.
12.
Centrifuge at 2000 × g for 2 min at 4 °C.
13.
Discard supernatant and wash two more times.
14.
Resuspend the cell pellet in 100 μL of PBS 1× and store at 4 °C until it is run.
3.3 Imaging Flow Cytometer Sample Run
1.
In INSPIRE, for the first run, select the appropriate parameters from the menus to the right: In the Illumination menu, ensure that Brightfield is on and set to 800 for channels 1 and 9, and that the 785 nm laser is set to 0.5; in the Magnification and EDF menu, set the magnification to 60×.
2.
Click on “Load”—the equipment will open the sample portal to load the vial containing the sample.
4.
Click on “OK” when the tube is correctly in place.
5.
Once the run starts, wait until the images appear to be in focus and centered (about 30–60 s).
6.
Click on “New Histogram”.
7.
Click on the “All” population to select it.
8.
Choose “Area_Ch01” as the X Axis Feature.
9.
This generates a new histogram in the analysis area whose title bar says “All”.
10.
Click on “Create Line Region” and draw a line that selects all events with an area between 20 and 350. This eliminates the acquisition of small particles and debris.
11.
Name this population “R1.”
12.
At this point, if this is the first sample run, save this layout as a template that can be loaded for future runs.
13.
Type the name of the file to which images should be saved. Make sure that the number of events acquired is set to 20,000, and that the events to be acquired come from “R1,” not “All,” to minimize acquisition of noise. Designate the destination folder into which files will be saved.
14.
Click on “Acquire” to start acquisition.
15.
Once acquisition is done, click on “Load” to discard the remainder of the sample and proceed to the next tube. The equipment will return the empty tube before allowing loading of the next sample. Alternatively, click on “Return” to save the remainder of the sample. The equipment will return the tube containing the remaining cell suspension. Click on “Load” again after removing the tube to allow the next sample to be loaded.
3.4 Imaging Flow Cytometry Analysis: Mask Definition
1.
Inside the IDEAS software, click on the “Guided Analysis” menu.
2.
3.
To define new masks, select the “Analysis” menu, then click on “Masks…” to open a dialogue box (see Note 6 ).
4.
On the left side, a box will show a list of masks already available. To create a new mask, click on “New.”
5.
Click on “Function” to open a dialogue box called “Define Mask Function”.
6.
Under “Function,” select “Object.”
7.
Under “Mask,” select “M01.”
8.
Under “Channel,” select “Ch01.”
9.
Under “Image,” select “Ch01.”
10.
The name of the mask will, by default, become the definition of the mask just created.
11.
Click on “OK.”
12.
This returns you to the previous box, where it will show the definition of the mask. It should show “Object(M01, Ch01, Tight)”.
13.
Click on “OK” to include the new mask. It will show up as the last mask on the list to your left.
14.
Click on “Function” to open the dialogue box called “Define Mask Function.”
15.
De-select “Link Inputs.”
16.
Under “Function,” select “System.”
17.
Under “Mask,” select “Object(M01, Ch01, Tight).”
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree