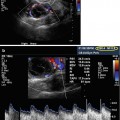
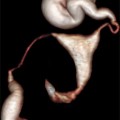
Fig. 13.1
2D longitudinal (upper image) and transverse (lower image) images of the uterus showing adequate distention of the endometrial canal with saline during SHG
SHG for Congenital Uterine Anomalies
SHG is a cost-effective method available in an outpatient setting which is highly accurate in identifying uterine anomalies, especially septate and bicornuate uterus [44]. Mullerian anomalies are congenital defects in the development of the uterus and the upper vagina. The ability of 2D US to distinguish between different types of uterine anomalies is limited and operator dependent. The finding of a uterine anomaly may affect the management of the infertile and/or pregnant woman and the pregnancy outcome. In a recent meta-analysis [39], including 94 observational studies comprising 89,861 women, the prevalence of uterine anomalies diagnosed by optimal tests was 5.5 % (95 % CI, 3.5–8.5) in unselected population, 8.0 % (95% CI, 5.3–12) in infertile women, 13.3 % (95 % CI, 8.9–20.0) in women with a history of miscarriage, and 24.5 % (95 % CI, 18.3–32.8) in women with miscarriage and infertility.
Congenital uterine anomalies are associated with poor reproductive outcome [40]. All uterine anomalies are associated with an increase incidence of fetal malpresentations at delivery. Unification defects do not reduce fertility but some defect, in particular bicornuate uteri, are associated with aberrant outcomes throughout the course of pregnancy. Canalization defects appear to reduce the chance of clinical pregnancy and to increase risk of preterm delivery. These are more profound in cases of septate uteri. Arcuate uteri, while previously considered to have no reproductive sequelae, are specifically associated with poor outcomes in late pregnancy, i.e., second-trimester miscarriage and malpresentation [40].
Uterine anomalies are defined by the criteria outlined by the American Society of Reproductive Medicine [41]. The visualization of the uterine fundus at the coronal plane is necessary for classifying uterine shape. SHG has been shown to have superior diagnostic ability with compared with HSG and 2D US for the evaluation of uterine malformation. Tur-Kaspa et al. [14] studied prospectively the prevalence of uterine anomalies diagnosed by SHG in 600 consecutive infertile patients compared to 409 patients with AUB. While the prevalence of septate uterus was 3 % in each group, arcuate uterus was significantly more common among the infertile patients (15 % vs. 6 %, respectively). All other anomalies had <1 % frequency in either group. Tur-Kaspa et al. [14], as well as others [7–11], concluded that SHG is an excellent method for the evaluation of congenital uterine anomalies. 3D SHG may be needed in some cases to assist in the final diagnosis.
SHG for Acquired Uterine Abnormalities
SHG can serve as a first-line test for the evaluation of acquired intrauterine abnormalities such as adhesions (Fig. 13.2), polyps (Fig. 13.3), and fibroids. Tur-Kaspa et al. [14] have documented that intracavitary abnormalities are significantly more frequent among patients with AUB than with infertility. Polyps were the most common abnormal finding among patients with AUB or infertile women (30 and 13 %, respectively) [14].
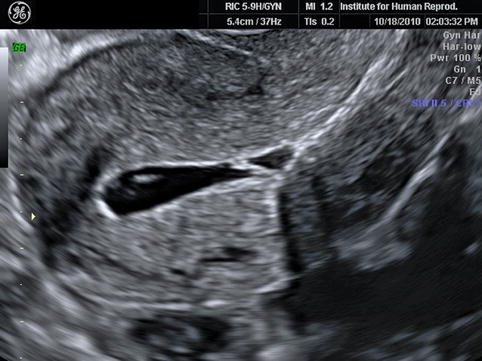
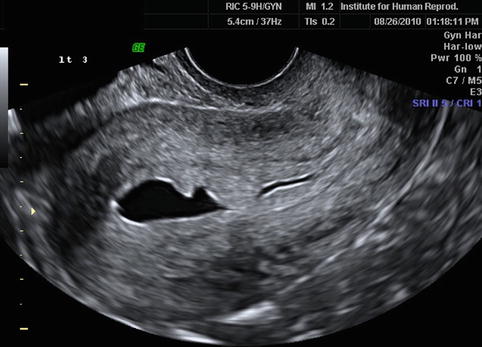

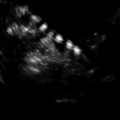
Fig. 13.2
2D longitudinal image of SHG demonstrating intrauterine adhesion at the lower uterine segment, connecting the anterior and the posterior walls of the uterus

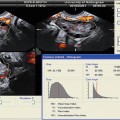
Fig. 13.3
2D longitudinal image of SHG demonstrating two polyps protruding into the uterine cavity
Submucosal fibroids were found in 9 % of the AUB group and 3 % among infertile women [14]. Submucosal fibroids have been shown by meta-analysis to significantly lower pregnancy rates in ART and should be removed by operative hysteroscopy [4, 6]. Besides infertility, the submucosal fibroids may cause bleeding and miscarriages. The European Society of Hysteroscopy has developed a classification system for fibroids which can also assist in the surgical approach. A Type 0 submucosal fibroid has no myometrial invasion, while a T1 has <50 % extension and T2 has more than 50 % extension into the myometrium. The TO and T1 are appropriate for the hysteroscopic approach, while the T2 may require more than one procedure or be removed laparoscopically.
2D vs. 3D SHG
When the option of having a 3D SHG scan is available, it may shorten the procedure and the volume of the saline used [42]. 3D SHG vs. 2D SHG is more accurate for diagnosing congenital uterine anomalies. For acquired uterine anomalies, in experienced hands, 3D will not improve the accuracy, but may assist it for better imaging (Figs. 13.4 and 13.5) [43, 44]. For the evaluation of postmenopausal bleeding, 2D and 3D SHG have similar diagnostic accuracy as hysteroscopy with higher patient acceptability of SHG [45, 46].
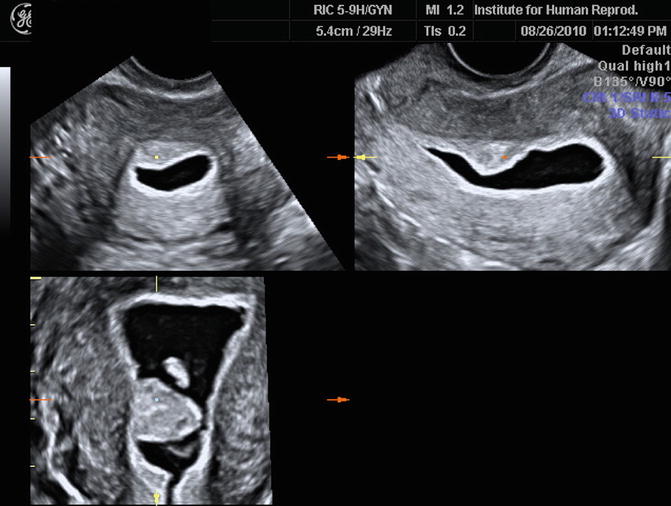
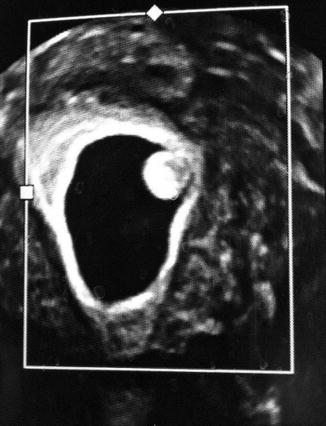

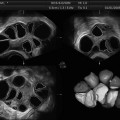
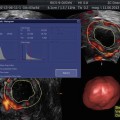
Fig. 13.4
3D SHG images of a uterine polyp. They are able to show the size and location of the stalk of the polyp more accurately in preparation for operative hysteroscopy and for consulting the patient

Fig. 13.5
3D SHG image of a corneal uterine polyp providing excellent information for the practitioner and patient on the size and location of the polyp
A 3D US, in comparison to a 2D US, allows for the visualization of the entire uterine cavity in the coronal view; it can detect the exact placement of uterine fibroids, polyps, and synechiae in the cavity, as well as the mean diameter of different tissues. A 3D US examination comprises approximately four steps: (1) data acquisition, (2) volume calculation, (3) image animation, and (4) data storage and transfer. The scans can be obtained either by freehand, by manual movement through the region of interest (ROI), or, automatically, by sweeping through the ROI. 3D US needs post-processing of the received data. Data can be stored and visualized in various displays such as multiplanar with navigation through the planes or surface rendering mode. For more details on 3D US technique, the reader may refer to Chap. 2.
A saline infusion enhances the contrast in a 3D US and can facilitate the accurate diagnosis of congenital uterine anomalies, especially the arcuate uterus (Fig. 13.6) compared with the septate uterus (Fig. 13.7) and the bicornuate uterus. The serosal edge and the fundal indentation can be clearly seen. Through TUI tomographic imaging, a series of images can visualize the leiomyomata protruding into the uterine cavity vs. deviating the endometrial cavity.
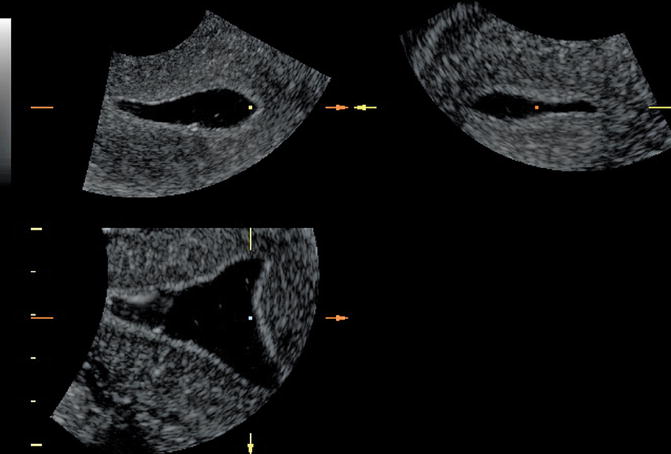
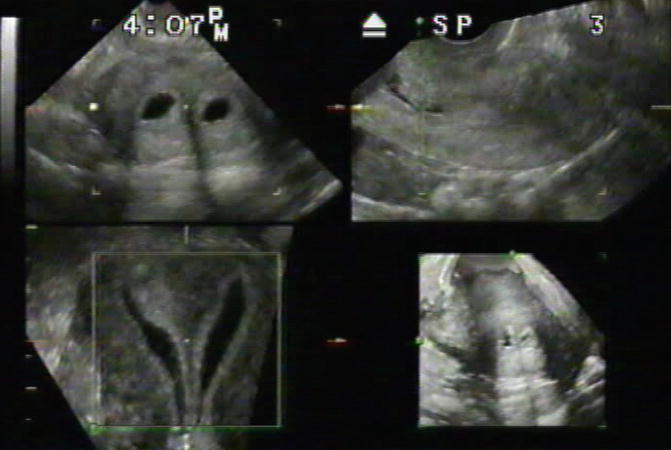

Fig. 13.6
3D SHG demonstrating an arcuate uterus. The visualization of the fundal area at the coronal plane and the ability to measure the depth of the anomaly can easily define arcuate uterus and rule out a septum

Fig. 13.7
3D SHG demonstrating a completely septated uterus. The 3D reconstruction at the coronal plane leaves no space for imagination, providing definite diagnosis and assisting in planning the surgical treatment needed as well as consulting with the patient
3D adds value to 2D SHG by improving with visualization of the uterine fundus [47]. Others suggest that when the SHG is performed by an experienced examiner, 3D does not add additional value to the 2D SHG [48]. It is the opinion of the authors that adding a 3D US to a 2D SHG will allow the exam to be completed faster with the same or better accuracy. Still in most cases, 2D SHG is adequate for diagnosing abnormal intracavitary finding.
Gel Instillation SHG
Gel SHG uses hydroxyethylcellulose gel instead of saline as its medium. This is done in order to try to simplify the technique of artificial uterine cavity distension for SHG [49]. The gel provides a more stable filling of the uterine cavity, allowing a high-quality ultrasonographic visualization of intrauterine pathology by 2D and 3D US [50– 55]. Still, most centers will use saline for SHG.
No Pain with SHG
Tur-Kaspa [37] has recently summarized data supporting that SHG, as well as HSG and HysteroContrastSonography (HyCoSy), should be considered pain-free procedures. Hysterosalpingography (HSG) has a long-standing reputation of being a painful procedure. The use of modern thin catheters and nonionic media that significantly reduced pain during and after HSG [56–60] was unable to affect significantly HSG’s “reputation.” SHG and HyCoSy, the modern ultrasound-based procedures that are currently used instead of HSG for the evaluation of the uterine cavity and/or the fallopian tubes, “inherited” this high level of fear of pain. It is possible that this stigma discourages patients and makes them believe that the procedure should be painful when it does not have to be. Several recent randomized controlled trials (RCT) have failed to demonstrate a significant benefit of various pharmacological strategies available to reduce pain during these procedures, suggesting that the pain is more psychological than physical [34–36]. It is the author’s opinion, based on evidence data and the experience of performing thousands of these tests, that they can be pain free for women.
One of the primary ways to make SHG a pain-free procedure is using gentle movements with a thin flexible catheter. Using a rigid catheter, which requires grasping the cervix with a tenaculum, will promote pain. If a balloon catheter is used, it is preferred to inflate the balloon intracervically rather than intrauterine, and the appropriate position of the catheter may be confirmed by pulling it slightly. An RCT recently showed a significantly less fluid used for SHG and significantly less pain felt by patients when the balloon was inflated inside the cervix rather than in the lower uterine segment [38]. Warming the saline solutions to body temperature before instillation is another way of reducing patients’ discomfort. It is crucial to introduce the saline solution slowly into the cavity to prevent abrupt overdistention of the uterus, which would induce immediate pain. While women naturally may feel embarrassed, stressed, and discomfort, as with any medical and gynecological examinations, there should be no more fear of pain from procedures such as SHG, HyCoSy, and HSG [37].
Conclusions
SHG is a simple, cost-effective, safe, and easy-to-perform procedure for the evaluation of congenital and acquired uterine abnormalities. Published guidelines by AIUM and ACOG are easy to implement in routine gynecological and reproductive medicine practice. While using thin flexible catheters, placing them inside the cervix, and injecting the saline slowly, this procedure can be pain free. SHG can serve as a first-line test for screening and evaluation of the uterine cavity for the diagnosis of infertility and before ART.
References
1.
Randolph JR, Ying YK, Maier DB, Schmidt CL, Riddick DH. Comparison of real-time ultrasonography, hysterosalpingography, and laparoscopy/hysteroscopy in the evaluation of uterine abnormalities and tubal patency. Fertil Steril. 1986;46:828–32.PubMed
2.
Syrop C, Sahakian V. Transvaginal sonographic detection of endometrial polyps with fluid contrast augmentation. Obstet Gynecol. 1992;79:1041–3.PubMed
3.