Grade of injury
Description
I
Hematoma: subcapsular with spleen surface involvement <10%
Laceration: capsular tear, <1 cm parenchymal depth
II
Hematoma: subcapsular, interest 10–50% of surface area
Hematoma: intraparenchymal, <5 cm in diameter
Laceration: capsular injury, 1–3 cm parenchymal depth that does not involve a trabecular vessel
III
Hematoma: subcapsular, interest >50% of surface area or expanding; ruptured subcapsular or parenchymal hematoma
Hematoma: intraparenchymal, >5 cm or expanding
Laceration:>3 cm parenchymal depth or involving trabecular vessels
IV
Laceration involving segmental or hilar vessels producing major devascularisation (>25% of spleen)
V
Laceration: complete shattered spleen
Vascular: hilar vascular injury with devascularized spleen
However, this surgical classification internationally used is not perfectly comparable to the real CT-findings: the differences could be attributable in some cases to the evolution of the injury by the time of surgery after the CT scanning, but the major limitation of the AAST scaling system was that it has not been reliable in predicting the outcome and guiding the management of blunt splenic injury [11]. In fact, in the last years targeted classification to define a CT-scoring system has been created. The most widely used classification based on CT is the Mirvis’ one [12, 13]. This classification includes the distinction in four grades:
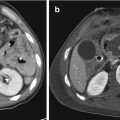
Grade 1 includes capsular avulsion, superficial laceration, and subcapsular hematoma smaller than 1 cm (Fig. 16.1);
Grade 2 includes parenchymal lesions from 1 to 3 cm, or parenchymal and subcapsular hematoma smaller than 3 cm (Fig. 16.2);
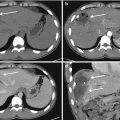
Grade 3 defines splenic laceration deep more than 3 cm (Fig. 16.3); at least
Grade 4 defines the fragmentation of the splenic parenchyma (“shattered spleen”) or its devascularization, what is called “non-enhanced spleen” (Fig. 16.4).

Fig. 16.1
Grade 1. Contrast-enhanced CT scan shows a small splenic laceration: (a) arterial and (b) venous phase. The portal-venous phase is better for the illustration of parenchymal damage (arrow)

Fig. 16.2
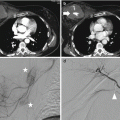
Grade 2: Not replenished parenchymal hematoma (smaller than 3 cm) associated with a small peri-splenic hemoperitoneum. CE-MDCT axial images in baseline (a), arterial (b), and venous phase (c)

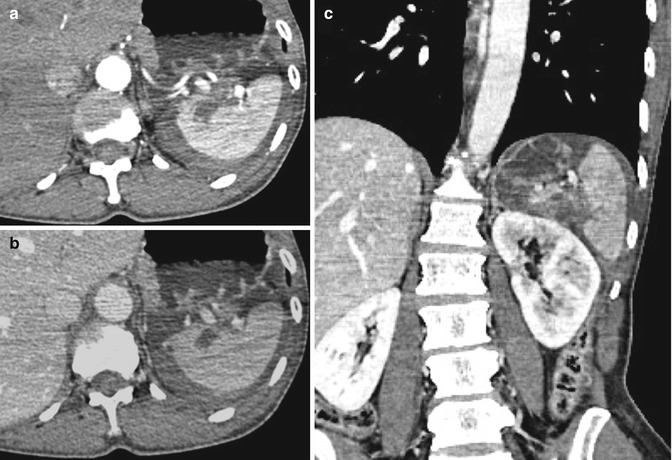
Fig. 16.3
Grade 3: These CT images show a deep hilar splenic laceration with rupture of the capsule. Multiphasic CT scans in arterial (a–c) and venous phase in (b)

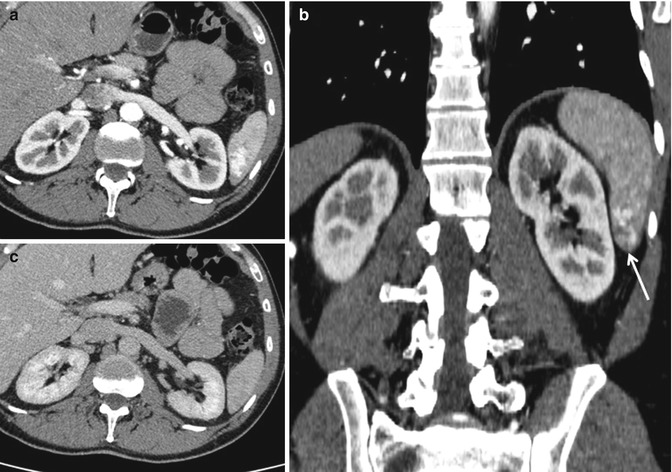
Fig. 16.4
Grade 4: Fragmentation of the splenic upper pole parenchyma, what is called “shattered spleen” (arrow). (a) Axial CT scan and (b) coronal reconstruction of the venous phase
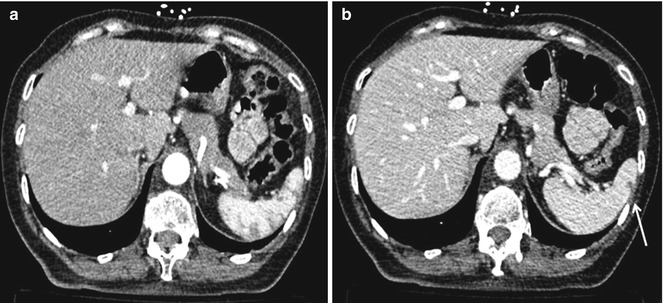
While severity of splenic injury according to the AAST does correlate with outcome, this classification, with the Mirvis’ one, does not take account of active extravasation or contained vascular injury such as arteriovenous fistula or pseudoaneurysm formation (Figs. 16.5 and 16.6). In fact, the last CT-based classification was written by Marmery in 2007 [14] and recalls Mirvis’, while adding vascular lesions (Grades 4a–4b), which are predictive of the necessity (or lack thereof) of surgical treatment or splenic embolization (Fig. 16.7).
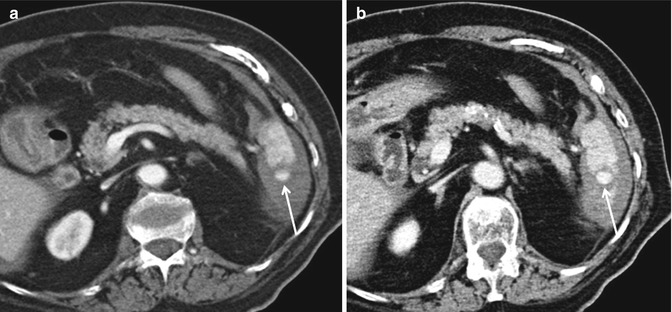
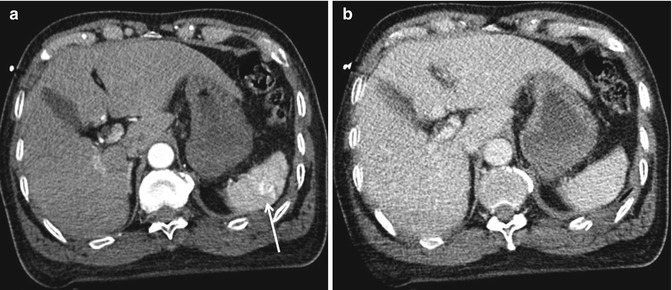
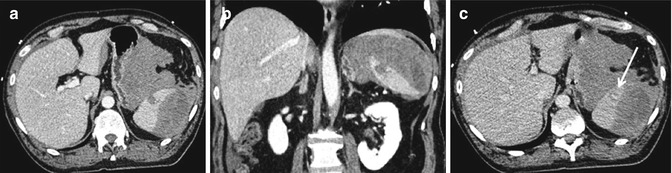

Fig. 16.5
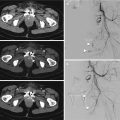
Grade 4a: CE-MDCT in arterial (a) and venous (b) phases allows to make a diagnosis of lower splenic pole pseudoaneurysm (arrows), associated with hemoperitoneum in peri-splenic and peri-hepatic spaces

Fig. 16.6
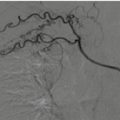
Arteriovenous fistula. Multiphasic CT study shows a small arteriovenous fistula at the upper splenic pole (arrow): the dynamic study is essential as it shows the classical sign of a contained vascular lesion that is a focal area of parenchymal hyperdensity in the arterial phase (a), followed by a typical “wash-out” isodensity in the portal phase (b)

Fig. 16.7
Grade 4a: Subcapsular and parenchymal replenished hematoma associated with “large” hemoperitoneum. CT scan: the venous phase (a, b) allows to identify the parenchymal lesion and the blood spreading in the abdomen; (c) the late phase shows a small blood supply within the lesion (white arrow)
16.4 Radiological Diagnosis
The radiologist has the essential task of providing an adequate diagnosis and classification of a splenic injury in order to refer the patient to a conservative treatment or to a surgical one.
The poly-injured patient’s radiological study is different according to the dynamics of trauma and their clinical conditions upon arrival at the emergency room.
Poly-traumatized hemodynamically unstable patient: Basic diagnostic tests are performed (chest and pelvis X-Rays, US e-FAST) to find pneumothorax, hemothorax, and hemoperitoneum. If one of these three is diagnosed and the patient is still hemodynamically unstable, he is directed to the surgical room. In the emergency room, the doctors try to stabilize the patient (with plasma expanders, hemothorax and pneumothorax drainage, pelvis fracture stabilization) and, if they achieve a hemodynamic stability, the patient is subjected to a total-body contrast-enhanced MDCT scan (CE-MDCT).
Poly-traumatized hemodynamically stable patient: Contrast-enhanced CT scan is always the best choice in major trauma.
In case of a localized low energy trauma, it is preferred to do an ultrasound exam (US), possibly complemented with the use of intravenous contrast (Contrast Enhanced UltraSonography—CEUS). If a lesion or free fluid (suspected for hemoperitoneum) are detected, a MDCT exam is requested for a complete evaluation of the patient.
US and CT have a key role in the management of blunt abdominal trauma. Studies have shown that US has sensitivity of 97% and specificity of 100% in predicting the presence of hemoperitoneum: in fact, US is the first choice technique for early screening, allowing to demonstrate the presence of hemoperitoneum in which patients that are hemodynamically unstable, and refer them to embolization or surgery [15]. Ultrasound should be the initial investigation of choice when examining children or in young woman [16]. However, US has low sensitivity in diagnosing an organ injury [17]. Over the last few years, with the introduction of Contrast Enhanced UltraSonography (CEUS) the sensitivity is greatly increased, with 96% sensitivity and 99% specificity rates compared to CT. CEUS shows a high sensitivity both in lesion detection and grading [18, 19] but it has a lower accuracy in the evaluation of prognostic factors; CEUS plays an important role also in blunt abdominal trauma in pediatric patients [20–22]. CT is undeniably the gold standard for the identification, diagnosis, and staging of abdominal organs in trauma patients: CT also shows all non-parenchymal organ lesions that US and CEUS are not capable of detecting. CT must always be performed in CEUS-positive patients to exclude active bleeding [23].
16.5 Computed Tomography
Computed tomography (CT) is the gold standard for patient evaluation in case of blunt injury and for the detection and staging of splenic lesions.
This technique is widely used in Emergency Radiology Departments, due to increasing diffusion of multi-detector CT (MDCT) and because it’s able to meet the main targets of Emergency Surgery which are early lesion identification, the assessment of the presence of hemoperitoneum and any further injury in whole body. Computed tomography study, due to its panoramicity, allows to identify any damage to every organ beyond the spleen. The aim of a correct and prompt diagnosis is to refer the patient to a surgical or conservative treatment, already in the acute phase, and avoid the misdiagnosis of the most insidious injuries, that may show signs only hours or days later.
In a patient with left-quadrant isolated trauma, after US and CEUS, CT can be used as a second choice, to clarify any questionable sonographic findings [16]; instead in a poly-traumatized hemodynamically stable patient, MDCT is the first choice and helps in differentiation of patients with active splenic bleeding (who risk hemorrhagic shock and which could indicate a need for surgery or embolization) from patients with stable non-bleeding injuries, who are candidates for conservative management, with the valuation of vital parameters and a close follow-up, to reduce surgical morbidity and preserve immune competence [24].
Total-body MDCT is always performed in basal phase and after intravenous administration of contrast, with dual-phase (acquisitions in the arterial and portal-venous phases): it is vital that the amount of contrast is adequate and customized to the patient’s weight, and that the flow must be high, because those parameters greatly influence the quality of the examination.
The multiphasic protocol provides a better overall diagnostic tool for the evaluation of the splenic lesions: in particular, the arterial phase is more sensitive for the evaluation of vascular injury including active bleeding and vascular lesions, while the portal-venous phase is better for the illustration of parenchymal damage. Several types of splenic injury can occur: intraparenchymal and subcapsular hematoma, contusion, laceration, active extravasation, and contained vascular injury [25]. Clotted blood has an attenuation of approximately 45–70 Hounsfield Units (HU); it is better seen in basal phase as a slightly hyperdense area, while unclotted blood has an attenuation of 30–45 HU.
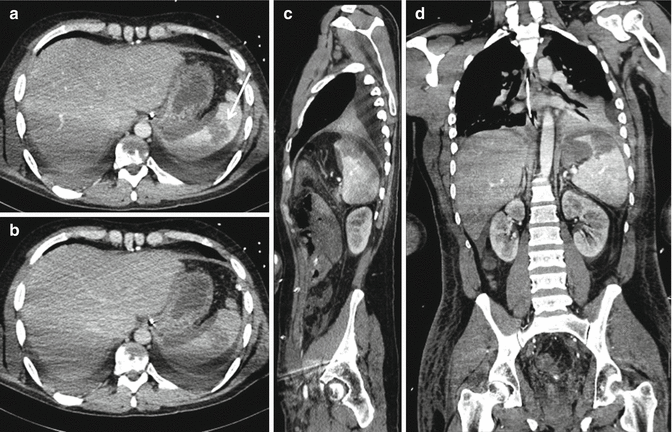
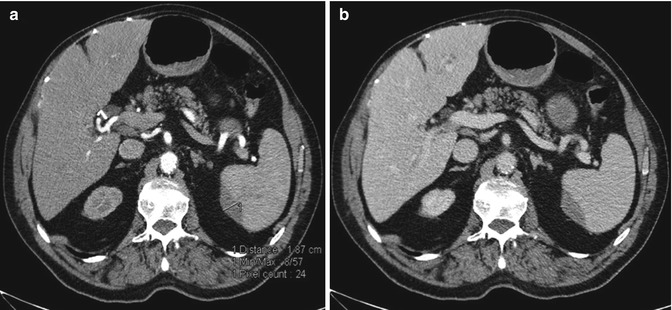
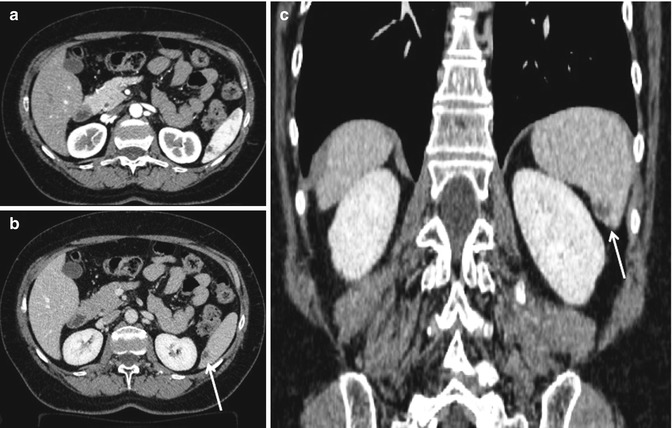
Parenchymal hematoma: At basal exam, a parenchymal hematoma may appear to be an area with defined margins, if it is made up of recent blood it is hyperdense but it can be iso-hypo-hyperdense compared to the surrounding parenchyma in relation to the time elapsed since the blood collected; during the dynamic phases, it is hypodense compared to the remaining parenchyma (Fig. 16.8).
Subcapsular hematoma: Similarly, at basal exam, subcapsular hematoma has the same semeiotic of a parenchymal hematoma and appears like a peripheral crescent collection that displaces and marks the parenchyma the splenic parenchyma (Fig. 16.9): it can even grow to considerable size, it is always hypodense during dynamic phases, and it can have a multi-stratified appearance due to repeated bleeding.
Splenic contusion: It is not well defined at baseline; instead at the contrast-enhanced exam, it appears as a small hypodense area with blurred margins caused by perilesional interstitial edema and local blood suffusion.
Laceration: It appears as a linear lack of parenchymal opacification with an irregular shape that may extend to the capsule (Fig. 16.10). It is important to report the laceration site, which can be superficial, if it interests only the capsule, or deep, involving vascular structures of splenic hilum.
Severe disruption of splenic parenchyma can result in a “shattered” spleen. Vascular hilum injuries usually result in significant hemorrhage and cardiovascular instability [26].

Fig. 16.8
Grade 3: Not replenished parenchymal hematoma of the upper splenic pole associated with a small subcapsular hematoma (arrow), as shown in portal-venous phase (a, axial scan, arterial phase, b, axial scan, late phase, c, sagittal and d, coronal reconstruction)

Fig. 16.9
Grade 2: these CT axial pictures show a subcapsular hematoma, smaller than 3 cm, studied in both arterial (a) and venous phase, (b). The arterial phase is more sensitive for the evaluation of vascular injury including active bleeding and vascular lesions: in fact, it is a not replenished hematoma as we cannot see any sign of active bleeding

Fig. 16.10
Grade 1. This dual-phase CT shows a small subcapsular laceration to the lower pole of the spleen (arrows): axial images in arterial (a) and venous phase (b). Coronal reconstruction in the venous phase (c)
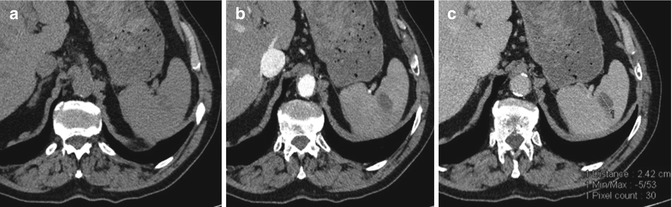
According to Marmery, the splenic lesion is classified from Grade 1 to Grade 3, and the grade increases according to the depth of the laceration [14, 27]. Concerning vascular lesions, this classification adds grade 4 injury, that is divided into subgroups: 4a and 4b. In fact, a laceration that compromises the vascularization of the spleen (vascular hilar damage) coincides with a severe injury (Grade 4). A contrast-medium extravasation is an active sign of bleeding, which can be subcapsular/parenchymal (Grade 4a) (Fig. 16.11) or endo-peritoneal/peri-splenic (Grade 4b) (Fig. 16.12) and almost always requires immediate surgical treatment. When the exam detects a bleeding, a vascular study is essential, as it allows to detect focal areas of high attenuation, which often is evident in the arterial phase and then spreads in portal-venous phase, this change in morphology allows to make a differential diagnosis between active bleeding and a contained vascular lesion (such as pseudoaneurysm and arteriovenous fistula) that does not change in its morphology. On single-phase scanning, it can be difficult to differentiate active extravasation from contained vascular injury; however, on equilibrium phase scans, an area of active extravasation will remain hyperdense and enlarged, whereas a contained vascular injury will typically “wash out” and be isodense or slightly hypodense relative to splenic parenchyma (Fig. 16.13). These contained vascular lesions are recognized as a well-defined margin in intraparenchymal areas, with high attenuation in arterial phase and isodensity in portal-venous phase (Fig. 16.14). The distinction between fistula and post-traumatic pseudoaneurysm is easier to identify with arteriography. In the pseudoaneurysm there is a tear in the artery, but bleeding is typically limited by the arterial adventitia.
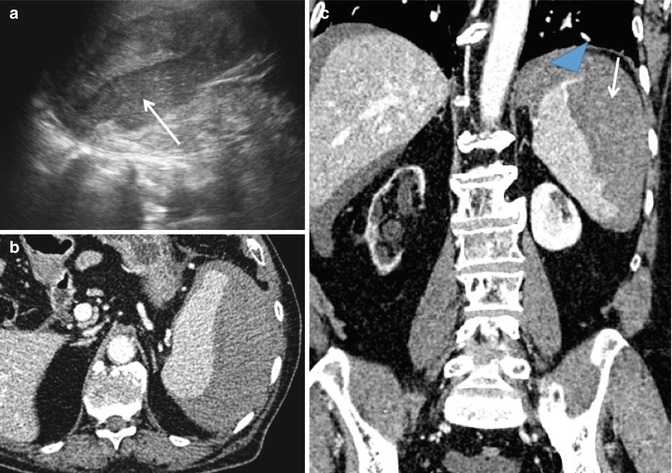
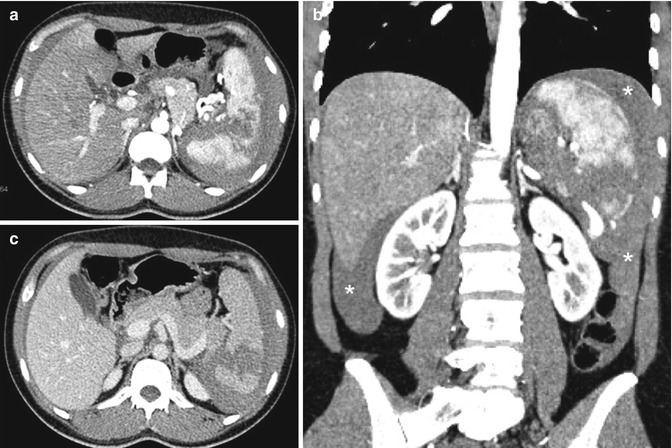
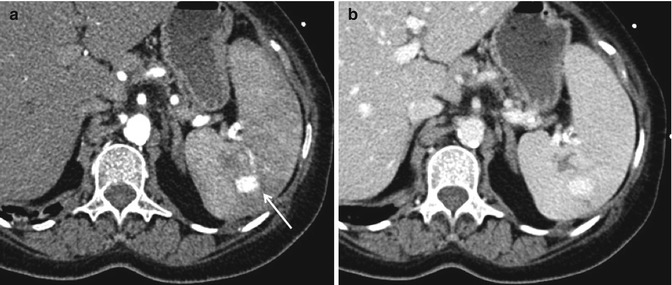
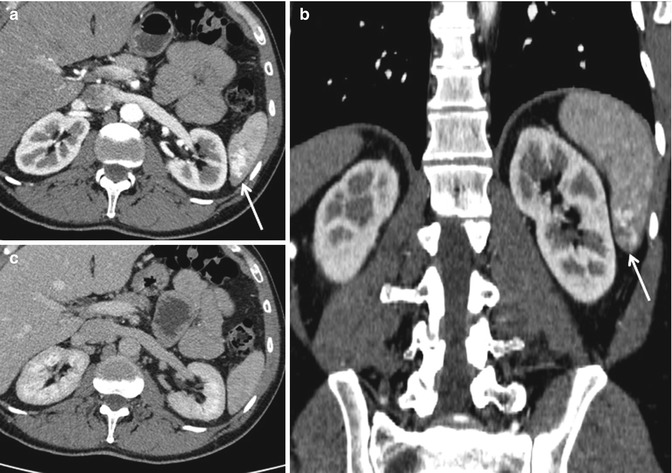

Fig. 16.11
Grade 4a: replenished subcapsular hematoma (arrows). (a) The US image shows a hematoma that is more hypoechoic than the splenic parenchyma. (b) Axial CT scan and (c) coronal reconstruction in the venous phase demonstrate a blood spreading at the upper splenic pole (blue arrowhead)

Fig. 16.12
Grade 4b: This vascular CT study shows a splenic fracture associated with “large” hemoperitoneum (asterisks) in peri-hepatic, peri-splenic and both parieto-colic spaces. There is no evidence of active blood extravasation: arterial (a, b) and venous phases (c)

Fig. 16.13
Grade 4a: These arterial (a), and venous (b), axial CT images show a pseudoaneurysm (arrow) associated with a splenic parenchymal laceration: in this case, a vascular study is essential as it allows to make a differential diagnosis between active bleeding and a contained vascular lesion (such as pseudoaneurysm and arteriovenous fistula). On single-phase scanning, it can be difficult to differentiate active extravasation from contained vascular injury; however, on delayed scans, an area of active extravasation will remain hyperdense and enlarged, whereas a contained vascular injury will typically “wash out” and be isodense or slightly hypo/hyperdense relative to splenic parenchyma

Fig. 16.14
Grade 4a: Arteriovenous fistula of the lower splenic pole: typical hyperdensity in the arterial phase (arrows) (a, b), isodensity in the venous phase (c)
Active bleeding increases in size on delayed-phase images unlike a contained vascular lesion [14]. Pseudoaneurysm and arteriovenous fistula are conditions predisposing a late-breaking spleen and typically are managed through embolization. Pseudoaneurysm breaks in a percentage that ranges from 3 to 46% of cases.
When the capsule is break, typically, hemoperitoneum is associated. According to the Federle semi-quantitative scale [28], hemoperitoneum is divided into small (when the blood is in only one endo-peritoneal space, as the peri-splenic, peri-hepatic, in the parieto-colic or Morrison spaces, with an estimated blood loss of about 250–500 mL) or large (with blood effusion in two or more endo-peritoneal/pelvic spaces, with blood extravasation greater than 500 mL) (Fig. 16.12).
16.6 Ultrasonography
Ultrasonography is used first in hemodynamically stable patients with minor trauma, because it is easy to perform, quick, affordable, and noninvasive. However, even in expert hands, basic US has too low sensitivity to diagnose organ injuries (no more than 50%) especially in case of spleen injury because the “fresh blood” has similar echogenicity as a recent parenchymal lesion. For this reason, CT was used excessively over the years so as not to miss potentially dangerous splenic injuries [3].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree