Fig. 5.1
Normal tendon. (a) Long-axis ultrasound image of the supraspinatus tendon demonstrates the normal hyperechoic appearance (arrows). (b) Short-axis ultrasound image of the Achilles tendon demonstrates normal hyperechoic appearance with visualization of individual tendon fibrils (arrows)
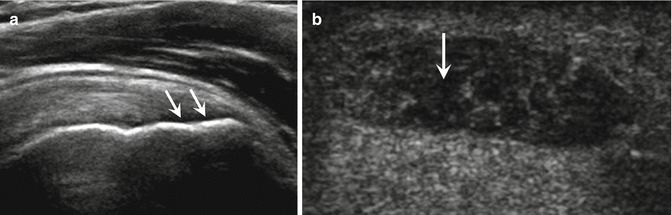
Fig. 5.2
Anisotropy. (a) Long-axis ultrasound image of the supraspinatus tendon shows insertional fibers are oblique to the ultrasound beam resulting in artificially decreased echogenicity (arrows). (b) Short-axis ultrasound image of the Achilles tendon with ultrasound beam angled obliquely to the tendon shows a falsely pathological, hypoechoic appearance (arrow)
Normal muscle appears hypoechoic with internal hyperechoic septations representing the perimysium that surrounds the bundles of muscle fibers (Fig. 5.3) (van Holsbeeck and Introcaso 2001). Muscles can present a characteristic multipennate appearance when viewed in long axis. Ligaments are typically hyperechoic and demonstrate a fibrillar structure similar to that of tendons (Fig. 5.4); however, the individual fibers are more tightly packed. Like tendons, ligaments demonstrate anisotropy when the ultrasound beam is not perpendicular to the structure. When surrounded by echogenic fat, ligaments may appear relatively hypoechoic (Jacobson 2002).
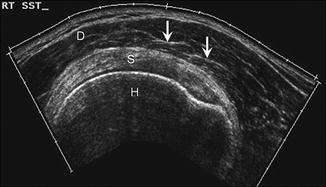
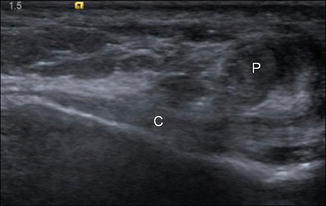

Fig. 5.3
Normal muscle. Short-axis ultrasound image using extended field of view of the deltoid muscle (D) shows hypoechoic appearance with echogenic striations reflecting the perimysium (arrows); humeral head (H), supraspinatus tendon (S)

Fig. 5.4
Normal ligament. Normal echogenic ultrasound appearance of the calcaneofibular ligament (C) in long axis adjacent to peroneal tendons (P)
Peripheral nerves demonstrate a fascicular pattern with hypoechoic nerve fascicles surrounded by a hyperechoic connective tissue known as the epineurium (Fig. 5.5) (Silvestri et al. 1995). Only the surface (cortex) of the bone is visible with ultrasound. The cortex is smooth and hyperechoic with posterior acoustic shadowing (Fig. 5.6) (Jacobson 2002).
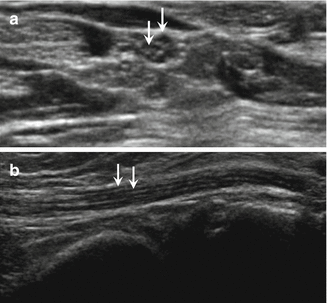
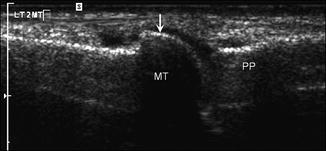

Fig. 5.5
Normal nerve. (a) Short-axis and (b) long-axis ultrasound images of a normal peripheral nerve demonstrating the hypoechoic fascicles surrounded by the echogenic epineurium (arrows)

Fig. 5.6
Normal bone. Long-axis ultrasound image shows echogenic cortex (arrow) of the second metatarsal (MT) at the level of the metatarsophalangeal joint with posterior acoustic shadowing; proximal phalanx (PP)
There are two types of cartilage within the musculoskeletal system, hyaline cartilage that lines the articular surfaces of the joints and fibrocartilage that comprises the labrum of the shoulder and hip and the menisci of the knee. Hyaline cartilage is hypoechoic (Fig. 5.7), while the normal fibrocartilage is hyperechoic (Fig. 5.8) (Kazam et al. 2011; Lee and Bouffard 2001).
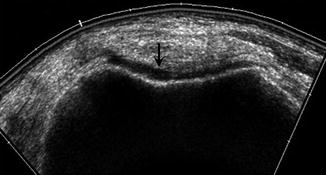
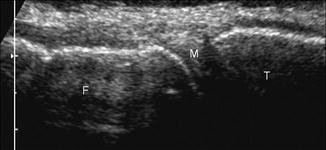

Fig. 5.7
Normal hyaline cartilage. Normal ultrasound appearance of the hypoechoic hyaline cartilage (arrow) lining the femoral trochlea

Fig. 5.8
Normal fibrocartilage. Long-axis ultrasound image of medial knee shows hyperechoic fibrocartilage of the meniscus of the knee (M); femur (F), tibia (T)
5.3 Ultrasound of Pathology in the Musculoskeletal System
5.3.1 Tendons
The pathology of tendons includes tendinosis or tendinopathy, partial tearing, and full-thickness tearing as well as the development of calcific deposits as seen in calcific tendinitis. Tendinosis manifests as abnormalities of both tendon morphology and echogenicity (Adler and Sofka 2003; Kainberger et al. 1990; Sell et al. 1996). There may be hypoechoic linear regions of diminished echogenicity as well as mucoid degeneration manifested by more globular regions of decreased echogenicity (Fig. 5.9). The tendon may be enlarged. In addition, areas of dystrophic calcification or ossification may also be seen (Adler and Finzel 2005).
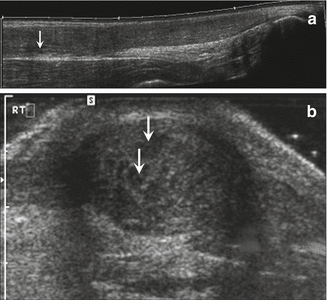

Fig. 5.9
Tendinosis. (a) Long- and (b) short-axis ultrasound images of the Achilles tendon show the tendon is enlarged and heterogeneous in echotexture with areas of decreased echogenicity (arrows)
Tears appear as discretely marginated defects within the tendon involving a portion of the tendon width (partial tear) (Fig. 5.10) or the entire width (full-thickness tear). The defects are well defined and hypoechoic or anechoic in appearance. Because tears and tendinosis can both appear hypoechoic, secondary signs may be necessary in differentiating these two entities (to be discussed later in this chapter) (Teefey et al. 2000a, b, c; Wiener and Seitz 1993).
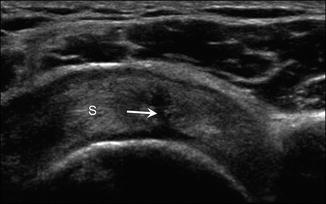

Fig. 5.10
Partial tear. Short-axis ultrasound image of the supraspinatus (S) demonstrates a discretely marginated linear defect (arrow) partially traversing the tendon width
Occasionally, areas of dystrophic calcification or ossification may develop within a tendon. These calcifications present as linear or globular foci of increased echogenicity with variable degrees of posterior acoustic shadowing (Fig. 5.11) (Adler and Finzel 2005; Farin and Jaroma 1995). The calcification represents deposits of calcium hydroxyapatite.
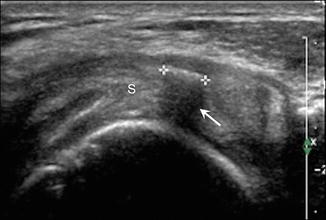

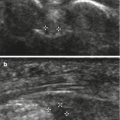
Fig. 5.11
Calcific tendinitis. Short-axis ultrasound image shows linear echogenic calcification (++) with posterior acoustic shadowing (arrow) within the supraspinatus tendon (S)
5.3.2 Muscles
Abnormalities of the muscle in the setting of sports medicine may result from direct or indirect injury. Indirect injury may be caused by stretching of the muscle fibers during muscle contraction or may be due to excessive stretch alone (Speer et al. 1993). There are three grades of injury that may result in this setting. Grade 1 refers to muscle strain without frank tissue disruption. Grade 2 refers to partial-thickness tearing of the muscle. Grade 3 refers to a full-thickness tear of the muscle (Palmer et al. 1999). These types of injuries typically occur at the myotendinous junction and affect muscles that traverse two joints such as the hamstring muscles in the thigh or the gastrocnemius muscle in the calf (Garrett 1996).
Acute muscle injury may be associated with hemorrhage that appears hyperechoic (Fig. 5.12) in the acute setting (Blankenbaker and Tuite 2010; Lee and Healy 2004). Over time, the hematoma becomes more hypoechoic in appearance. Most frequently, hematomas are heterogeneous in appearance with areas of increased and decreased echogenicity (Lee and Healy 2004). Diffuse muscle edema also appears hyperechoic (Adler and Garofalo 2009). Partial tears manifest as incomplete disruption (Fig. 5.12) of the muscle fibers, while complete disruption of the muscle fibers reflects a full-thickness tear (Peetrons 2002). Over time, the muscles may develop granulation tissue that displays vascularity on color or power Doppler flow imaging.
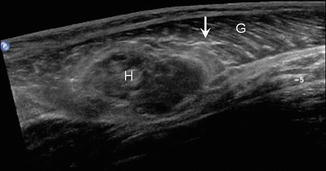

Fig. 5.12
Partial muscle tear. Long-axis ultrasound image shows grade 2 injury of the medial gastrocnemius muscle (G) with partial disruption of the normal muscle fibers (arrow) and an associated hematoma (H)
A special subtype of injury occurs when hemorrhage occurs between the deep surface of the subcutaneous fat and the subjacent musculature due to a shear injury, most frequently at the level of the hip. This is known as a Morel-Lavallee lesion (Mellado and Bencardino 2005). On ultrasound, these lesions are heterogeneous in the early stages, with irregular margins. Chronic lesions are smoother in contour and more homogeneous. They may contain hypoechoic or anechoic fluid and may contain echogenic globules of fat (Fig. 5.13). No flow should be demonstrated with color or power Doppler interrogation (Mukherjee et al. 2007; Neal et al. 2008; Parra et al. 1997).

Fig. 5.13
Morel-Lavallee lesion. Long-axis ultrasound image of the lateral thigh shows a hypoechoic fluid collection (H) with lobular echogenic foci reflecting fat globules (F). No flow is present with power Doppler interrogation
Occasionally, following trauma, the injured muscle may ossify. This appears as a region of increased echogenicity that may show posterior acoustic shadowing (Fig. 5.14). Ultrasound may demonstrate early mineralization before changes are visible on plain radiography (Tyler and Saifuddin 2010).
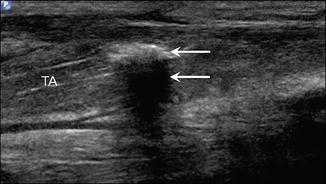

Fig. 5.14
Myositis ossificans. Long-axis ultrasound image of the tibialis anterior (TA) muscle several months following direct trauma shows linear echogenic focus with posterior acoustic shadowing (arrows) reflecting myositis ossificans at the site of prior injury
Finally, a previously injured muscle or a muscle with an injury to its nerve supply may atrophy. This results in decreased muscle volume and increased echogenicity of the affected muscle (Fig. 5.15). Atrophy may also occur in the setting of chronic tear of the associated tendon (Peetrons 2002).
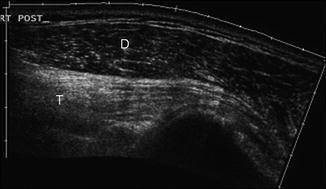

Fig. 5.15
Muscle atrophy. Long-axis ultrasound image of the shoulder shows denervation atrophy of the teres minor (T) with increased echogenicity of the muscle reflecting fatty replacement. Deltoid (D) demonstrates normal hypoechoic appearance of muscle with the normal echogenic perimysium
5.3.3 Bones
As previously mentioned, the normal cortex of the bone is smooth and hyperechoic with posterior acoustic shadowing. In the setting of an acute fracture, there is focal discontinuity of the cortex (Fig. 5.16) with or without an associated cortical step-off (Crag et al. 1999). The sonographic appearance of stress fractures includes periosteal reaction with hyperemia on color or power Doppler interrogation. There may hypoechoic periosseous fluid and/or hemorrhage and possible cortical discontinuity (Bodner et al. 2005; Drakonaki and Garbi 2010). Patients will typically report point tenderness with transducer pressure. The presence of modular increased echogenic soft tissue about the fracture site represents callous formation.
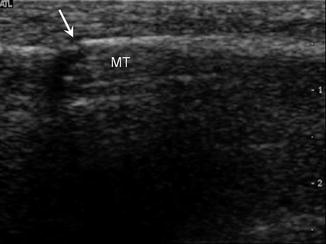

Fig. 5.16
Fracture. Long-axis ultrasound image shows focal cortical disruption (arrow) in a patient with a nondisplaced fracture of the fifth metatarsal (MT)
5.3.4 Ligaments
Abnormalities of the ligaments include partial and complete tears. Partial tears appear as areas of decreased echogenicity within a thickened ligament with some fibers remaining in continuity. A complete tear manifests as disruption of the ligament fibers or replacement of the fibers by heterogeneous, hypoechoic tissue representing fluid and/or hemorrhage and residual ligament (Fig. 5.17). Occasionally, a hyperechoic fleck of bone can be seen in the setting of an avulsion fracture. Remote sprains are characterized by thickening and enlargement of the ligament possibly associated with focal ossification (Peetrons et al. 2004). Scar remodeling or granulation tissue may appear as hypervascular soft tissue at the site of the tear.
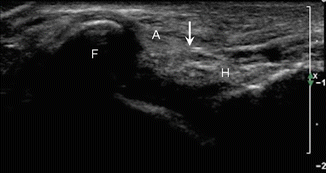

Fig. 5.17
Ligament tear. Long-axis ultrasound image shows the anterior talofibular ligament (A) with complete rupture with torn fibers (arrow) attached to the fibula (F) and hemorrhage (H) in gap
5.3.5 Nerves
Abnormalities of the nerves may be caused by entrapment secondary to ligamentous or fibro-osseous structures or secondary to mass effect from anomalous muscles, tumors, or fluid collections. Alternatively, abnormal motion of a nerve can result in nerve pathology as seen in ulnar nerve subluxation. The abnormal nerve appears enlarged and hypoechoic with loss of the normal fascicular pattern (Fig. 5.18) (Bianchi 2008; Jacobson et al. 2010; Kermarrec et al. 2010). In the setting of a localized nerve injury, a posttraumatic neuroma may form. These appear as discrete hypoechoic nodules.
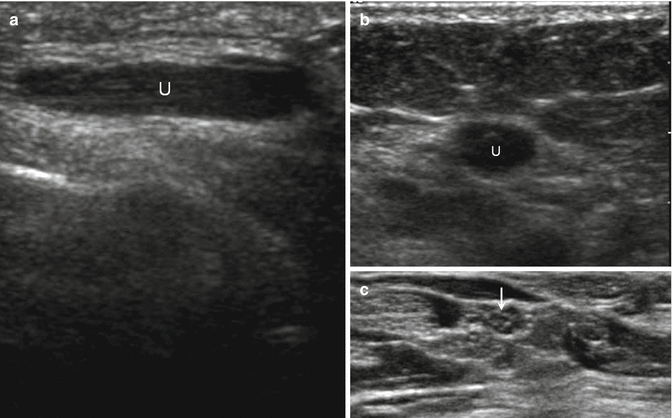

Fig. 5.18
Ulnar neuritis. (a) Long- and (b) short-axis ultrasound images of the ulnar nerve (U) in a patient with ulnar neuritis. The nerve is enlarged and hypoechoic with loss of the normal fascicles. (c) Compare to a normal peripheral nerve in short axis with hypoechoic fascicles surrounded by hyperechoic epineurium (arrow)
5.4 Shoulder Ultrasound
5.4.1 Rotator Cuff
The shoulder is the most frequently evaluated joint with sonography. The routine shoulder examination includes the assessment of the rotator cuff tendons (supraspinatus, infraspinatus subscapularis, teres minor), long head of the biceps brachii, and the acromioclavicular joint as well as the performance of dynamic maneuvers to assess for subacromial impingement (Jacobson 2013a). The most commonly affected tendon is the supraspinatus tendon. Pathology of the tendons ranges from tendinosis to full-thickness, full-width tears.
Tendinosis manifests as focal or diffuse decreased echogenicity of the tendon with possible enlargement of the affected tendon (Fig. 5.19) (Adler and Sofka 2003; Kainberger et al. 1990; Sell et al. 1996). Partial tears appear as focal hypoechoic or anechoic defects within the tendon that involve only the bursal or articular surface of the tendon (Fig. 5.20) (Jacobson et al. 2004; van Holsbeeck et al. 1995). Partial tears may also be intrasubstance or interstitial, not extending to the articular or bursal surfaces.
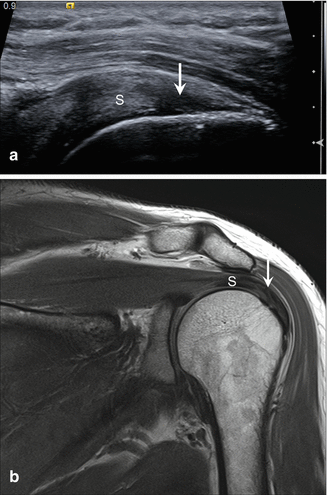
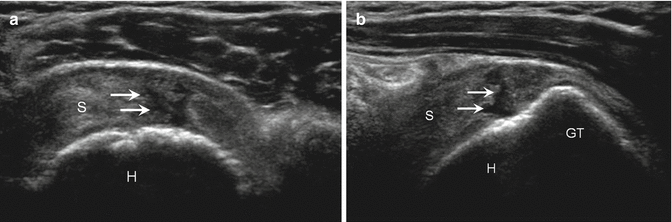

Fig. 5.19
Rotator cuff tendinosis. (a) Longitudinal ultrasound image of the supraspinatus (S) demonstrates decreased echogenicity and loss of the normal fibrillar pattern of the tendon (arrow). (b) Corresponding coronal proton density-weighted MRI demonstrates increased signal (arrow) in the supraspinatus (S) tendon due to tendinosis

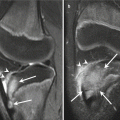
Fig. 5.20
Supraspinatus partial tear, articular surface. (a) Short- and (b) long-axis ultrasound images demonstrate a high-grade partial articular-sided tear (arrows) of the supraspinatus (S) tendon; humerus (H), greater tuberosity (GT)
The most commonly encountered partial, articular surface tear involves the anterior supraspinatus tendon distally at its insertion on the greater tuberosity (Fig. 5.20) (Schaeffeler et al. 2011; Tuite et al. 1998). Cortical irregularity or enthesopathic change involving the greater tuberosity may be present related to chronic rotator cuff degeneration (Fig. 5.21). This will not be present in the setting of an acute tear of an otherwise normal tendon or a tear involving the more proximal non-insertional tendon fibers (Jacobson et al. 2004; Wohlwend et al. 1998). In the setting of a partial, articular surface tear, the superficial surface of the tendon may maintain its normal convexity on ultrasound, without associated volume loss (Fig. 5.20) (Jacobson 2013a).
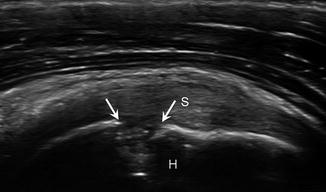

Fig. 5.21
Enthesopathy. Short-axis ultrasound image of the supraspinatus (S) tendon insertion demonstrates enthesopathic cyst formation (arrows) in the humeral head (H)
A special subtype of partial articular surface tear involving the most distal, insertional fibers of either the infraspinatus or supraspinatus is known as a rim-rent tear or PASTA lesion when it involves the supraspinatus (partial articular-sided supraspinatus tendon avulsion) (Fig. 5.22). This more frequently affects younger patients, such as athletes (Tuite et al. 1998; Vinson et al. 2007).
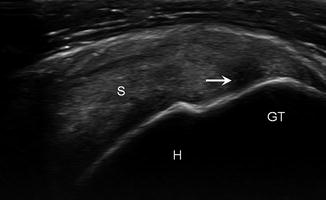

Fig. 5.22
Supraspinatus tear, rim rent. Long-axis ultrasound image of the supraspinatus (S) demonstrates a partial, rim-rent-type tear (arrow) at the insertion on the greater tuberosity (GT); humerus (H)
Like partial articular surface tears, bursal surface tears also appear hypoechoic or anechoic; however, these tears occur along the more superficial aspect of the tendon (Fig. 5.23) (Jacobson et al. 2004). Tears extending from the bursal surface through the tendon footprint to the greater tuberosity are still considered bursal surface tears (Jacobson 2013a).
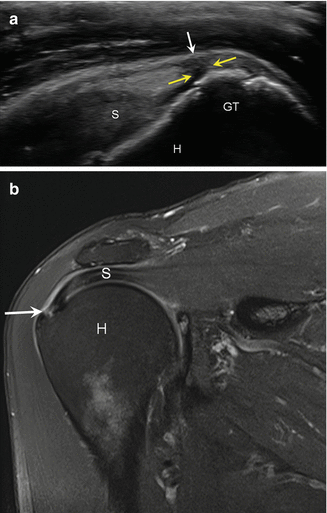

Fig. 5.23
Supraspinatus tear, bursal surface. (a) Long-axis ultrasound image of the supraspinatus (S) insertion shows a high-grade bursal surface tear (yellow arrows) extending from the collapsed subacromial bursa (white arrow) nearly to the greater tuberosity (GT); humerus (H). (b) Corresponding coronal fluid-sensitive MRI demonstrates the bursal tear (white arrow) of the supraspinatus (S); humerus (H)
Since tendon tears and tendinosis both may appear hypoechoic, secondary findings will be helpful in differentiating these entities. Indirect signs of a tear include diminished thickness of the tendon. In addition, the hypoechoic tear tends to be a more focal and well-defined defect, rather than the ill-defined hypoechoic areas seen in tendinosis (Teefey et al. 2000a, b, c; Wiener and Seitz 1993). The cartilage interface sign may assist in the diagnosis of partial articular surface tears. This finding occurs when the tear contacts the hypoechoic hyaline cartilage along the humeral head, appearing as a hyperechoic curvilinear interface between the tear and the cartilage (Fig. 5.24) (Bouffard et al. 1993). Secondary signs of bursal surface partial tears include tendon thinning and herniation of the subacromial–subdeltoid bursa and peribursal fat into the defect with resultant loss in the normal convexity of the tendon (Fig. 5.25) (Bouffard et al. 1993; Thain and Adler 1999; Wiener and Seitz 1993). Tears may be accentuated in real time by direct compression with the transducer. This maneuver may be helpful when complex material fills the tear or in differentiating partial from full-thickness tears.
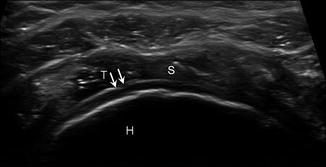
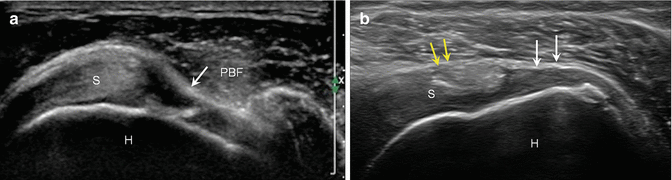

Fig. 5.24
Cartilage interface sign. Short-axis ultrasound image of the supraspinatus (S) demonstrates the cartilage interface sign (arrows) indicating extension of the tear (T) to the articular surface; humerus (H)

Fig. 5.25
Secondary sign, bursal tears. (a) Long-axis ultrasound image of the supraspinatus (S) shows herniation of the peribursal fat (PBF) and subacromial–subdeltoid bursa (arrow) into the bursal surface tear (T). (b) Long-axis ultrasound image of the supraspinatus (S) shows loss of the normal tendon convexity (white arrows) in a subtle bursal surface tear. Normal convex surface of tendon (yellow arrows); humerus (H)
Tendinosis and tears of the subscapularis tendon are less common than supraspinatus tears. Partial- and full-thickness tears have similar sonographic appearances as tears of the supraspinatus and infraspinatus tendon. The more common type of tear involves the superior fibers of the subscapularis associated with a tear of the anterior fibers of the supraspinatus. In the setting of a partial-thickness subscapularis tear, the long head biceps brachii tendon may subluxate or dislocate into the substance of the tear (Fig. 5.26). Full-thickness tears of the subscapularis may allow the biceps brachii tendon to dislocate into the glenohumeral joint (Fig. 5.27) (Farin and Jaroma 1996; Morag et al. 2011; Thain and Adler 1999). The multipennate nature of the subscapularis tendon seen in short axis should not be confused with tendon pathology.
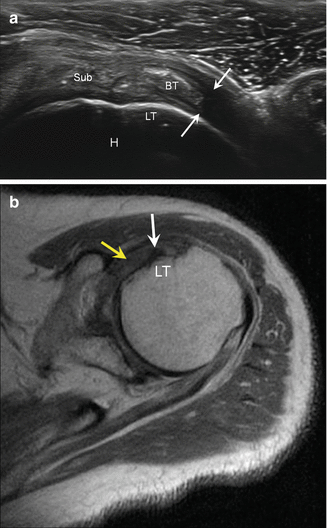
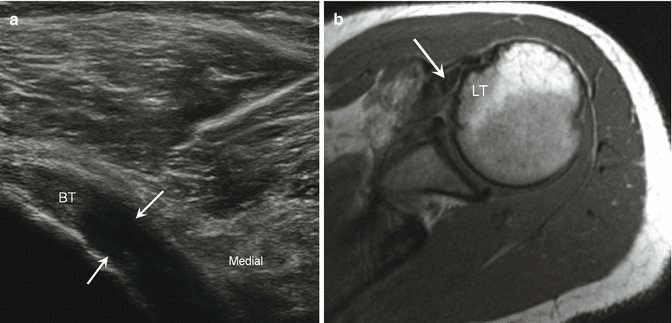

Fig. 5.26
Subscapularis partial tear. (a) Long-axis ultrasound image of the subscapularis (Sub) demonstrates mild medial subluxation of the biceps tendon (BT) into the high-grade tear (arrows); biceps is perched on lesser tuberosity (LT); humerus (H). (b) Corresponding axial proton density-weighted MRI shows medial subluxation of the biceps (white arrow) into a subscapularis tear (yellow arrows); lesser tuberosity (LT)

Fig. 5.27
Subscapularis complete tear. (a) Long-axis ultrasound image shows anechoic defect due to complete subscapularis tendon tear (arrows) with complete medial dislocation of the biceps (BT). Corresponding axial proton density-weighted MRI (b) shows medial dislocation of the biceps (arrow) beyond the lesser tuberosity (LT) due to the complete subscapularis tear
5.4.2 Biceps
Biceps tendinosis appears diffusely hypoechoic on ultrasound (Fig. 5.28) (Adler and Sofka 2003; Kainberger et al. 1990; Sell et al. 1996). The biceps tendon may be enlarged, and there may be associated calcification manifesting as echogenic foci with posterior acoustic shadowing (Adler and Finzel 2005). Small calcifications may not shadow and may just appear as discrete echogenic foci. Partial or complete disruption of the tendon may occur. Additionally, linear regions of decreased echogenicity may also be present reflecting interstitial split tears (Thain and Adler 1999). As mentioned above, the biceps tendon may subluxate or dislocate secondary to partial- or full-thickness tears of the subscapularis tendon (Figs. 5.26 and 5.27). The biceps tendon may be perched on the lesser tuberosity in the setting of subluxation or translate more medially in the setting of a full-thickness subscapularis tear. In dynamic ultrasound imaging, placing the shoulder in external rotation can accentuate subluxation and dislocation of the biceps tendon (Farin et al. 1995a). Complete rupture of the biceps tendon results in an empty sheath (Fig. 5.29) (Thain and Adler 1999). Although a trace amount of fluid may normally be seen adjacent to the biceps tendon, when a greater amount of fluid is seen surrounding a focal segment of the tendon, this suggests tenosynovitis (Jacobson 2013a). These findings can be further confirmed with the use of power Doppler that will demonstrate hyperemia in the setting of inflammation as seen in tendinitis and tenosynovitis (Fig. 5.30) (Thoirs et al. 2008). Normal tendons and synovium about the shoulder should demonstrate minimal or no flow with power Doppler interrogation. A branch of the anterior humeral circumflex artery adjacent to the biceps tendon will cause a focus of blood flow on power Doppler imaging that should not be confused with tenosynovitis (Fig. 5.31) (Thain and Adler 1999). Because the biceps tendon sheath communicates with the glenohumeral joint, the joint should be assessed for an effusion so as not to mistake biceps tenosynovitis for normal decompression of joint fluid into the tendon sheath (Zubler et al. 2011).
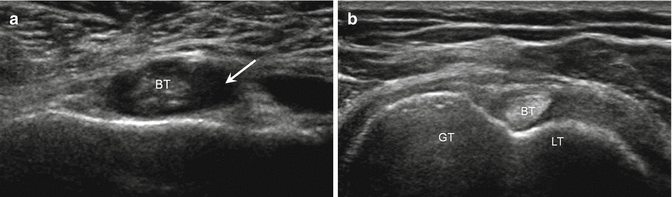
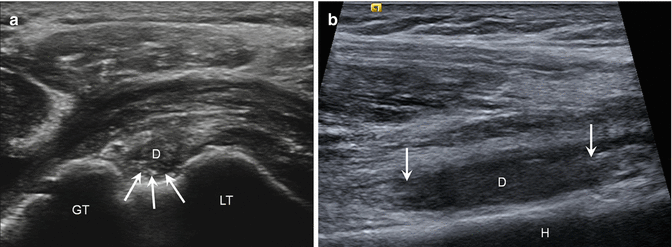
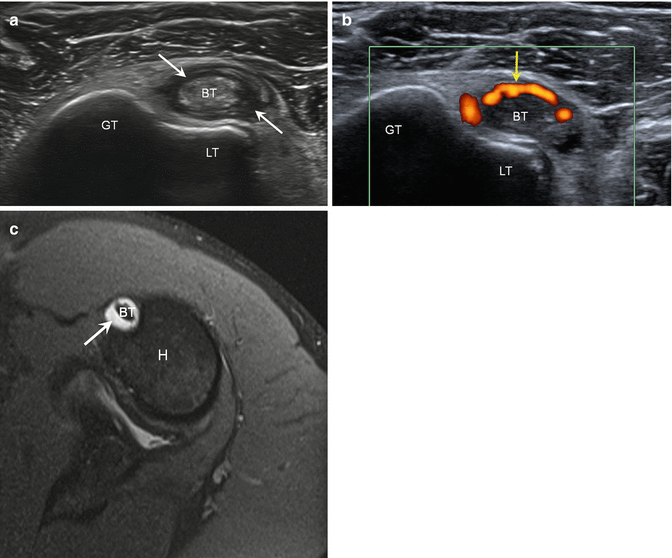
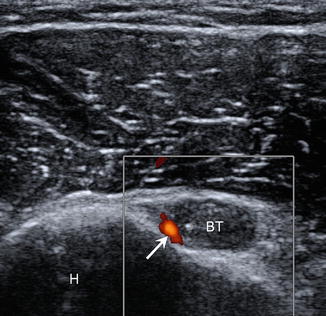

Fig. 5.28
Biceps tendinosis. (a) Short-axis ultrasound image of the biceps tendon (BT) shows an enlarged tendon with loss of the echogenic, fibrillar appearance; tendon sheath effusion (arrow). In (b), normal ellipsoid, echogenic appearance of the biceps tendon (BT) in short axis; greater tuberosity (GT), lesser tuberosity (LT)

Fig. 5.29
Proximal biceps rupture. (a) Short-axis ultrasound image shows empty bicipital groove (arrows) due to rupture of the biceps with debris (D) in the sheath; greater tuberosity (GT), lesser tuberosity (LT). (b) Long-axis ultrasound image demonstrates retracted ends of torn tendon (arrows) with debris (D) in sheath; humerus (H)

Fig. 5.30
Biceps tenosynovitis. (a) Short-axis ultrasound image of the biceps tendon (BT) shows an enlarged, heterogeneous tendon surrounded by hypoechoic fluid (arrows); greater tuberosity (GT), lesser tuberosity (LT). (b) Power Doppler ultrasound image demonstrates hyperemia (yellow arrow) due to tenosynovitis of the biceps tendon (BT); greater tuberosity (GT), lesser tuberosity (LT). (c) Corresponding axial fluid-sensitive MRI shows biceps tendon (BT) surrounded by fluid (arrow); humerus (H)

Fig. 5.31
Circumflex artery branch. Short-axis ultrasound image of the biceps tendon (BT) with normal branch of the anterior humeral circumflex artery (arrow) that should not be confused with hyperemia; humerus (H)
5.4.3 Calcific Tendinitis
Calcific tendinitis develops due to the deposition of calcium hydroxyapatite in the rotator cuff tendons. The supraspinatus is by far the most frequently affected tendon (80 %); however, the other rotator cuff tendons may also be affected (Bianchi and Martinoli 2007; Bosworth 1941; Mole et al. 1997). It is thought to be the result of local hypoxia within the tendon that results in metaplasia of the tendon and resultant calcification (Uhthoff and Sarkar 1989). Areas of calcification may appear as linear echogenic foci with posterior acoustic shadowing or more globular areas of echogenicity without shadowing (Fig. 5.32). These latter calcifications are typically more symptomatic and associated with hyperemia on power Doppler interrogation (Chiou et al. 2002). These calcifications may be treated with ultrasound-guided aspiration that will be discussed later in this chapter (Farin et al. 1995b; Howard et al. 1993).
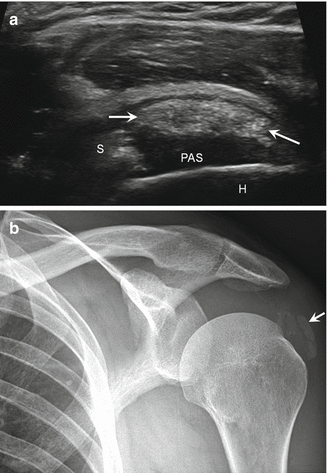

Fig. 5.32
Supraspinatus calcific tendinitis. Long-axis ultrasound image of the supraspinatus tendon (S) shows an ovoid echogenic focus (arrows) with posterior acoustic shadowing (PAS) reflecting calcific tendinitis; humerus (H). (b) Anteroposterior radiograph shows calcific deposit (arrow)
5.4.4 Impingement
Impingement syndromes in the shoulder are readily evaluated with dynamic ultrasound imaging. Impingement occurs when the acromiohumeral interval through which the supraspinatus tendon passes is narrowed. This may be due to a thickened coracoacromial ligament, an acromioclavicular joint arthrosis, or a subacromial spur (Farin et al. 1990). On ultrasound, the supraspinatus tendon and subacromial–subdeltoid bursa become entrapped between the greater tuberosity and the structures of the coracoacromial arch (Farin et al. 1990). As the arm is abducted, there may be “bunching” of the supraspinatus tendon and subacromial–subdeltoid bursa as these structures attempt to pass under the coracoacromial arch (Fig. 5.33). There may also be pooling of fluid within the bursa with subacromial–subdeltoid bursitis (Farin et al. 1990). Abnormalities of the supraspinatus tendon may be seen in association with impingement including tendinosis and tears. Similar findings can be seen in subcoracoid impingement where there is narrowing of the coracohumeral interval, resulting in the entrapment of the subscapularis tendon during internal rotation of the shoulder (Troelsen et al. 2007).
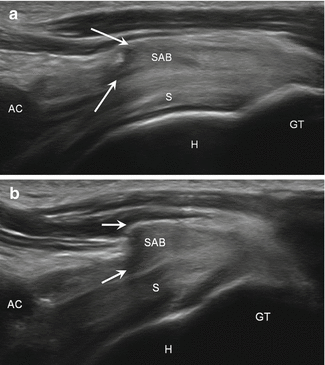

Fig. 5.33
Subacromial impingement. Long-axis ultrasound images of supraspinatus tendon (S) with (a) mild shoulder abduction show thickened subacromial bursa (SAB) becoming “bunched” (arrows) due to impingement under acromion (AC). With increased abduction in (b), the bursa demonstrates increased “bunching” (arrows); humerus (H), greater tuberosity (GT)
5.4.5 Glenoid Labrum
As previously described, the normal glenoid labrum, composed of fibrocartilage, is hyperechoic (Fig. 5.34). The labrum attaches to the adjacent glenoid and appears as a triangular structure (Talijanovic et al. 2000). Degeneration of the labrum appears as diffuse hypoechogenicity, while a tear appears as a discrete hypoechoic or anechoic defect (Tirman et al. 1994). While a portion of the posterior labrum can be visualized when scanning the infraspinatus tendon and the posterior joint recess, the anterior labrum is difficult to assess due to overlying structures. Therefore, MRI with or without intra-articular gadolinium should be considered to evaluate the labrum.
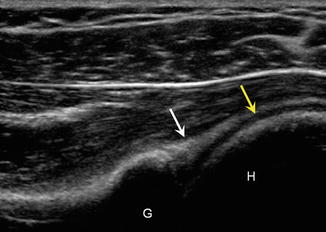

Fig. 5.34
Normal glenoid labrum. Longitudinal ultrasound image shows normal posterior labrum (white arrow) which appears as an echogenic triangle attached to the glenoid (G); normal hypoechoic articular cartilage (yellow arrow), humerus (H)
A paralabral cyst is a fluid collection that may develop when fluid from the glenohumeral joint extends through a labral tear into the surrounding tissues (Hashimoto et al. 1994). Around the shoulder, this may result in compression of the suprascapular nerve within the suprascapular or the spinoglenoid notch (Fehrman et al. 1995; Tirman et al. 1994; Tung et al. 2000). The cysts appear as round, anechoic sometimes multiloculated collections that can contain echogenic debris (Fig. 5.35). A communication through the associated labral tear may also be demonstrated. There may be associated atrophy of the infraspinatus or supraspinatus and infraspinatus muscles in the region of the spinoglenoid notch or suprascapular notch cysts, respectively (Fig. 5.36). These cysts may be aspirated under ultrasound guidance (see Sect. 5.10.1) often producing thick, clear material, although the cysts can recur if the underlying labral tear is not repaired.
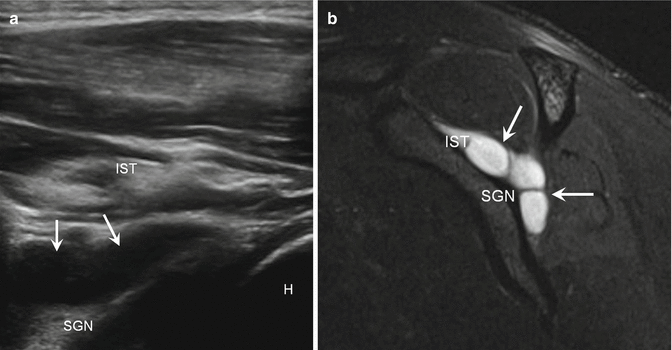
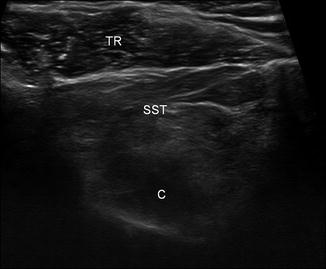

Fig. 5.35
Paralabral cyst. (a) Long-axis ultrasound image along the posterior shoulder at the spinoglenoid notch (SGN) demonstrates an anechoic collection (arrows) consistent with a paralabral cyst; infraspinatus (IST), humerus (H). In (b), corresponding sagittal fluid-sensitive MRI shows the spinoglenoid notch (SGN) with the paralabral cyst (arrows) and mild denervation edema in the infraspinatus muscle (IST)

Fig. 5.36
Muscle atrophy. Short-axis ultrasound image of the supraspinatus muscle (SST) shows increased echogenicity due to denervation from the suprascapular notch paralabral cyst (C). Compare to normal echogenicity of the adjacent trapezius (TR)
5.4.6 Fractures
Fractures of the greater tuberosity can be diagnosed during evaluation of the shoulder following trauma. Nondisplaced fractures may be missed at plain radiography. Fractures appear as a focal disruption of the echogenic cortex with an associated step-off (Fig. 5.37) (Patten et al. 1992). There will typically be point tenderness on exam over the fracture site.
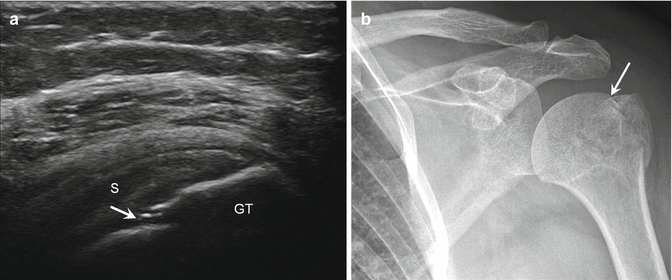

Fig. 5.37
Greater tuberosity fracture. (a) Long-axis ultrasound image of the supraspinatus tendon (S) and greater tuberosity (GT) demonstrates focal cortical disruption (arrow) due to a minimally displaced fracture. (b) Corresponding anteroposterior radiograph shows the fracture (arrow)
5.4.7 Pectoralis Major
Although not routinely evaluated during a shoulder ultrasound examination, the pectoralis major muscle and tendon may be assessed as directed by the patient’s clinical history or symptoms. The normal pectoralis major muscle has three heads, the clavicular, sternal, and abdominal heads. The tendons converge into a single tendon unit to insert on the bicipital groove of the humeral shaft, just lateral to the biceps tendon. The insertion extends over 5 cm in craniocaudal dimension (Rehman and Robinson 2005). Injury of the pectoralis muscle has become more common in high-performance athletes and is seen almost exclusively in men (Weaver et al. 2005). Injuries to the pectoralis include avulsion injuries at the tendon insertion with or without osseous avulsion or injury to the myotendinous junction or muscle belly (Miller 2003). As in the rotator cuff, injuries may be partial or full thickness. The most common injuries are partial tears involving both the sternal and clavicular heads (Connell et al. 1999; Lee et al. 2000). Injuries manifest as hypoechoic or anechoic regions within the tendon, at the myotendinous junction, or within the muscle belly with disruption of the tendon and/or muscle fibers (Fig. 5.38). Retraction indicates a full-thickness tear. There may be associated hematoma that can be hypoechoic or anechoic (Weaver et al. 2005). There may be edema within the more proximal muscle bellies. Dynamic examination may accentuate the site of the tear.
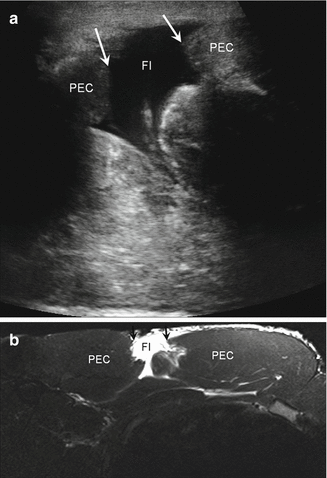

Fig. 5.38
Pectoralis muscle tear. (a) Long-axis ultrasound image of the pectoralis major muscle (PEC) demonstrates a myotendinous junction tear (arrows) with intervening fluid/hemorrhage (Fl). (b) Corresponding axial fluid-sensitive MRI shows the tear (black arrows) of the pectoralis (PEC) with a fluid-filled gap (Fl)
5.5 Elbow Ultrasound
5.5.1 Biceps Brachii
Because many of the ligaments and tendons of the elbow are relatively superficial in location, they are readily evaluated with ultrasound. Evaluation of the distal biceps, however, can be difficult due to its oblique course, anisotropy, and the deeper location of the insertion on the radial tuberosity. Provocative maneuvers and dynamic imaging may be used to better visualize the insertion and to eliminate anisotropy (Fig. 5.39) (Chew and Giuffrè 2005; Miller and Adler 2000). Injuries of the biceps tendon typically occur 1–2 cm proximal to the insertion on the radial tuberosity. Tears can be complete or partial and may be associated with pre-existing tendinosis due to relative hypovascularity of the tendon in this region (Chew and Giuffrè 2005; Vardakas et al. 2001). Mechanical impingement between the radial tuberosity and proximal ulna during pronation may also predispose to biceps tendon tears (Fig. 5.40) (Chew and Giuffrè 2005; Seiler et al. 1995). There may be associated bicipitoradial bursitis manifesting as a hypoechoic fluid collection that may contain internal debris or echoes that insinuates between the insertional fibers of the biceps tendon and the adjacent radial tuberosity (Fig. 5.41) (Sofka and Adler 2004).
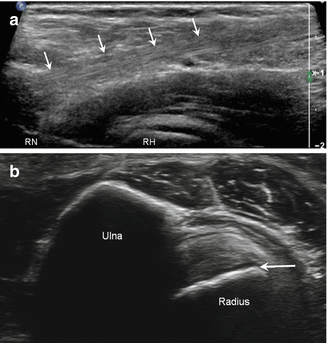
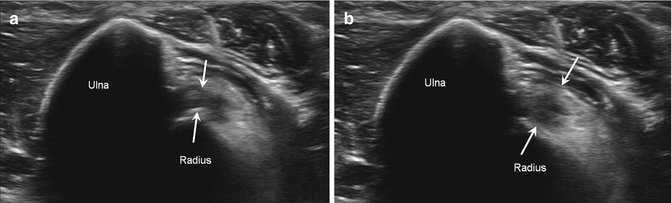
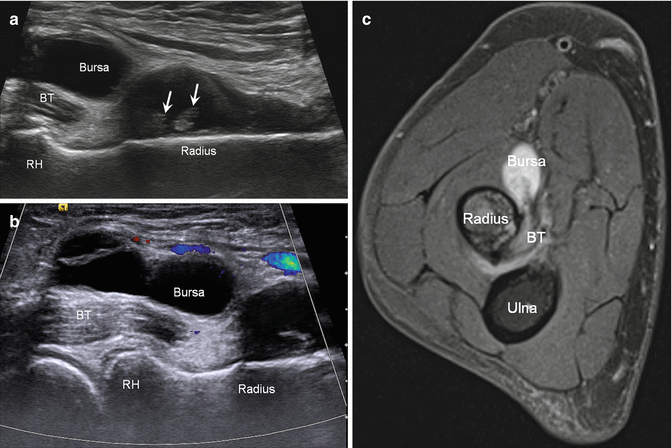

Fig. 5.39
Normal biceps insertion. (a) Long-axis ultrasound image shows the normal echogenic appearance of the biceps tendon insertion (arrows); radial neck (RN), radial head (RH). (b) Short-axis ultrasound image of the posterior forearm just below the elbow joint with the wrist in pronation shows the normal insertion of biceps (arrow); radius and ulna

Fig. 5.40
Biceps impingement at insertion. Short-axis ultrasound images of the posterior forearm just below the elbow in (a) pronation and (b) supination show the biceps tendon insertion (arrows) becoming “bunched” in supination (b) due to impingement; radius and ulna

Fig. 5.41
Bicipitoradial bursitis. Long-axis ultrasound images without (a) and with (b) color Doppler demonstrate distension of the bicipitoradial bursa (Bursa) at the level of the biceps tendon (BT) insertion on the radius; radial head (RH), synovial hypertrophy (arrows). (c) Corresponding axial fluid-sensitive MRI shows the distended bicipitoradial bursa (Bursa) where biceps tendon (BT) inserts on the radius; ulna
With partial tears, there is thickening of and decreased echogenicity within the tendon (Fig. 5.42). This may be difficult to differentiate from tendinosis. There may also be attenuation of the distal tendon in the setting of partial tearing. Tendon retraction will be absent. Dynamic imaging can help elucidate partial tears from tendinosis and nonretracted full-thickness tears (Chew and Giuffrè 2005; Hayter and Adler 2012; Kalume et al. 2009). With full-thickness tears, there will be complete disruption of the tendon fibers with a hypoechoic defect. There may be surrounding edema or hemorrhage in the acute setting (Fig. 5.43). If there is tearing of the bicipital aponeurosis, there will be proximal retraction of the tendon into the arm. This usually occurs in the setting of a single, forceful traumatic injury. Ultrasound may be used to locate the retracted tendon stump (Chew and Giuffrè 2005; Hayter and Adler 2012; Miller and Adler 2000).
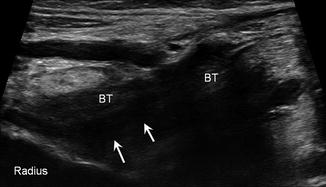
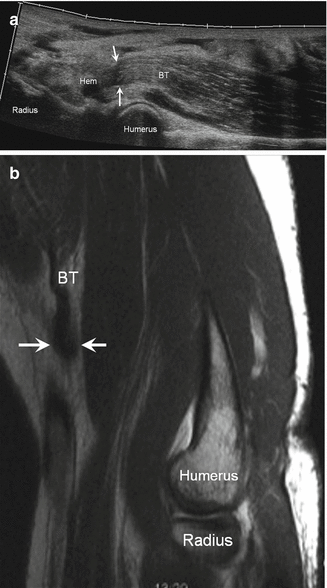

Fig. 5.42
Partial tear distal biceps. Long-axis ultrasound image of the biceps tendon (BT) insertion shows a hypoechoic defect (arrows) due to partial tearing; radius

Fig. 5.43
Complete rupture distal biceps. (a) Longitudinal ultrasound image of the distal biceps tendon (BT) demonstrates complete rupture at the insertion on the radius (Rad) with retraction of the tendon (arrows) and intervening hemorrhage (Hem); humerus. (b) Sagittal proton density-weighted MRI shows the ruptured biceps tendon (BT) that is proximally retracted (arrows); humerus, radius
5.5.2 Triceps Brachii
Triceps injury may result from direction impaction or tendon avulsion from the olecranon. Tears may be partial or complete, and there may be pre-existing tendinosis or enthesopathy (Fig. 5.44), or there may be associated olecranon bursitis. Tears appear as hypoechoic or anechoic defects in the tendon involving some (partial) or all (complete) of the tendon fibers (Fig. 5.45). There may be tendon retraction in full-thickness tears, and a linear focus of increased echogenicity reflecting an osseous fragment may be present in the setting of bone avulsion (Fig. 5.46). Tendinosis appears as tendon enlargement with decreased echogenicity of the tendon fibers without disruption (Downey et al. 2011; Hayter and Adler 2012; Jacobson 2013b).
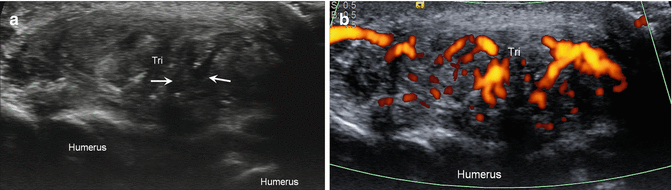
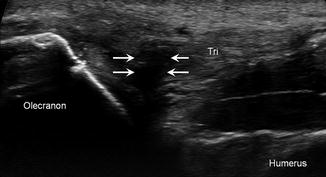
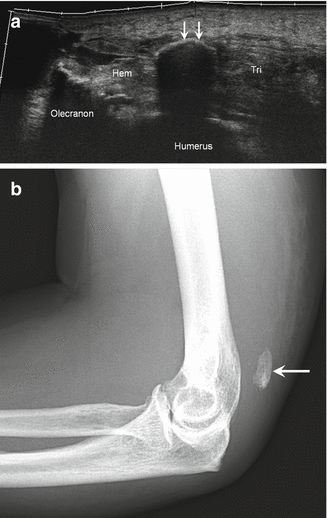

Fig. 5.44
Triceps tendinosis. (a) Transverse ultrasound image of the distal triceps tendon (Tri) shows enlargement and heterogeneity of the tendon with interstitial tearing (arrows). (b) Power Doppler image shows extensive hyperemia due to neovascularity in the setting of tendinosis; humerus

Fig. 5.45
Triceps tendon tear. Longitudinal ultrasound image demonstrates a high-grade partial, deep surface tear (arrows) of the distal triceps tendon (Tri); humerus (Hum), olecranon (Olec)

Fig. 5.46
Triceps tendon avulsion. (a) Longitudinal ultrasound image shows a complete avulsion of the distal triceps tendon (Tri) from the olecranon with an ossific avulsion (arrows) and hemorrhage (Hem); humerus. (b) Lateral radiograph of a different patient with similar findings of an avulsion fracture (arrow) due to a triceps tendon tear
5.5.3 Common Flexor and Extensor Tendons
The origins of the flexor and extensor tendons of the hand and wrist originate from the medial and lateral epicondyles, respectively. Disorders of the tendons including tendinosis and tearing are the sequelae of acute, traumatic, and chronic overuse injuries (Hayter and Adler 2012). The term lateral epicondylitis or “tennis elbow” refers to pathology of the common extensor tendons. Although acute or chronic inflammation is lacking, the term epicondylitis continues to be used. The underlying pathology involves tendinosis and tearing of the tendons (Nirschl and Pettrone 1979; Potter et al. 1995). At ultrasound, the tendons appear enlarged and hypoechoic (Connell et al. 2000). There may be foci of internal calcification within the tendon and enthesopathic change at the lateral epicondyle (Hayter and Adler 2012). With power Doppler interrogation, there may be increased vascularity (Tran and Chow 2007). The most commonly and severely affected tendon is the extensor carpi radialis brevis. In the setting of tears, there is partial- or full-thickness discontinuity of the tendon fibers (Fig. 5.47) (Levin et al. 2005).
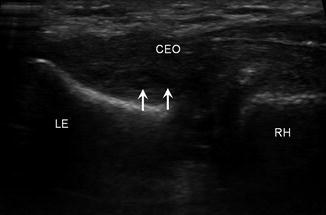

Fig. 5.47
Lateral epicondylitis. Longitudinal ultrasound image of the lateral elbow shows thickening of the common extensor tendon origin (CEO) with a partial deep surface tear (arrows); lateral epicondyle (LE), radial head (RH)
Medial epicondylitis or “golfer’s elbow” refers to tendinopathy of the common flexor origin. This entity is much less common than lateral epicondylitis and is most frequently seen in overhead, throwing athletes such as baseball pitchers (Kaplan and Potter 2004). The findings of medial epicondylitis are similar to those of lateral epicondylitis (Fig. 5.48) (Kijowski and De Smet 2005; Miller et al. 2002). Ultrasound is useful in assessing both the findings of medial epicondylitis and concomitant pathology of the medial collateral ligament and the ulnar nerve that will be further described below (Martin and Schweitzer 1998).
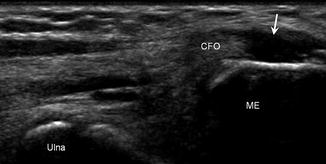

Fig. 5.48
Medial epicondylitis. Longitudinal ultrasound image of the medial elbow shows an anechoic defect (arrow) in the common flexor tendon origin (CFO) reflecting a high-grade tear; medial epicondyle (ME), ulna
5.5.4 Medial (Ulnar) Collateral Ligament and Lateral (Radial) Collateral Ligament Complex
The primary stabilizer of the medial elbow against valgus instability is the medial collateral ligament (MCL), composed of anterior, posterior, and transverse/oblique bundles (Morrey and An 1983). The anterior bundle is the most functionally important and frequently evaluated component of the MCL complex, extending from the undersurface of the medial humeral epicondyle to the sublime tubercle of the ulna (Hayter and Adler 2012). This segment of the MCL is the most commonly injured ligament in the elbow and may be injured acutely, or more commonly, as a result of chronic overuse as seen in overhead throwing athletes (Jobe and Nuber 1986). The normal appearance of the anterior bundle of the MCL is a smooth, hyperechoic band of tissue (Fig. 5.49). Due to anisotropy, the ligament may appear hypoechoic (Jacobson et al. 2003; Ward et al. 2003). Sprains of the ligament appear as decreased echogenicity with loss of the normal band-like appearance of the ligament. Partial- and full-thickness tears appear as hypoechoic or anechoic areas of discontinuity in the ligament fibers (Fig. 5.49). In the setting of hemorrhage, it may be difficult to distinguish partial- and full-thickness tears (Miller et al. 2004). Dynamic imaging is helpful in these situations by exerting valgus stress on the elbow in partial flexion and assessing for separation of the torn fibers as well as widening of the medial joint line. The contralateral side may be imaged concurrently to assess for asymmetry (De Smet et al. 2002; Nazarian et al. 2003). In the setting of chronic MCL pathology due to overuse in overhead athletes, the ligament may be thickened and hypoechoic or may become remodeled and elongated due to the repetitive stress (De Smet et al. 2002; Nazarian et al. 2003). In the chronic setting, there may be associated signs of valgus extension overload or posteromedial impingement that can be seen in the throwing athlete. These include common flexor tendinopathy, medial joint line osteophytes, intra-articular bodies, or ulnar neuropathy. Due to increased compressive forces on the lateral joint line, there may be associated radiocapitellar arthritis (Gaary et al. 1997; Hayter and Adler 2012; Kijowski et al. 2005; Wilson et al. 1983).
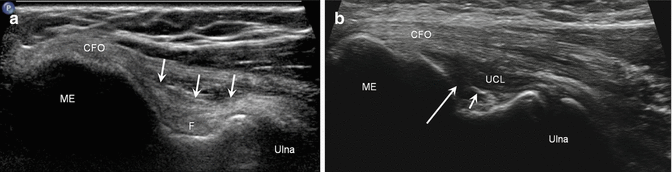

Fig. 5.49
Normal and torn ulnar collateral ligament. (a) Longitudinal ultrasound image of the medial elbow demonstrates the normal ulnar collateral ligament (arrows). (b) Long-axis ultrasound image of the medial elbow in a different patient shows an anechoic area reflecting a tear (long arrow) of the ulnar collateral ligament (UCL) at the origin with a small avulsion fracture (short arrow); common flexor origin (CFO), medial epicondyle (ME), ulna, fat (F)
The lateral collateral ligament complex is composed of the radial collateral ligament (RCL), the lateral ulnar collateral ligament (LUCL), and the annular ligament. The LUCL is the most important stabilizer against varus stress and posterolateral rotatory instability (Olsen et al. 1996). The LUCL originates from the posterior aspect of the lateral humeral epicondyle and curves around the posterior aspect of the radial head to insert on the supinator crest of the ulna. The RCL originates at the lateral humeral epicondyle and inserts along the annular ligament (Hayter and Adler 2012). The RCL and common extensor origin may be difficult to separate when both structures are normal. The normal ligaments should be echogenic and band-like in appearance (Fig. 5.50) (Jacobson 2013b).
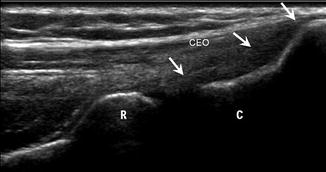

Fig. 5.50
Normal lateral structures of the elbow. Longitudinal ultrasound image of the lateral elbow shows the common extensor origin (CEO) superficial to, and nearly inseparable from, the radial collateral ligament (arrows). The lateral ulnar collateral ligament (not shown) is slightly posterior to the radial collateral ligament; radius (R), capitellum (C)
The LUCL may be injured acutely following varus extension injury or a traumatic elbow dislocation, such as after a fall on an outstretched hand (Nestor et al. 1992). The ligament may also be injured iatrogenically following overaggressive debridement of the common extensor tendon for the treatment of lateral epicondylitis. In addition, severe cases of lateral epicondylitis may extend into and involve the lateral collateral ligament complex. Disruption of the LUCL results in posterolateral rotatory instability with posterior subluxation of the radial head with respect to the capitellum (O’Driscoll et al. 1992; Walz et al. 2010). Like the UCL, tears of the lateral ligamentous structures manifest as areas of decreased echogenicity with partial or complete disruption of the ligament fibers (Fig. 5.51).
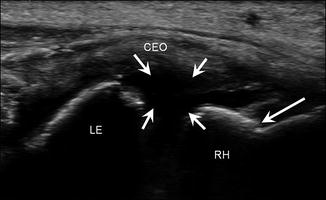

Fig. 5.51
Tear of lateral ligaments of the elbow. Longitudinal ultrasound image of the lateral elbow following dislocation shows complete disruption of the lateral ligaments (short arrows) with a focal cortical disruption at the radial head (RH)/neck junction (long arrow) indicating a fracture; common extensor origin (CEO), lateral epicondyle (LE)
5.5.5 Ulnar Neuropathy
The ulnar nerve traverses the posterior elbow within the cubital tunnel. The cubital retinaculum forms the roof of this tunnel, keeping the nerve within the tunnel through the range of motion of the elbow. Localized ulnar neuropathy at the elbow is known as cubital tunnel syndrome (Hayter and Adler 2012). Causes of ulnar neuropathy include acute trauma, chronic repetitive injury during elbow flexion, ulnar nerve subluxation/dislocation, or the presence of an anconeus epitrochlearis muscle, an anatomic variant present in up to 23 % of the population (Husarik et al. 2009; Jacobson 2013b). With ulnar neuritis, the nerve is abnormally enlarged and diffusely decreased in echogenicity with loss of the normal fascicular pattern (Fig. 5.52) (Hayter and Adler 2012). A cross-sectional area of greater than 9 mm2 is considered abnormal (Thoirs et al. 2008). Measurements, however, are not well established for the ulnar nerve.
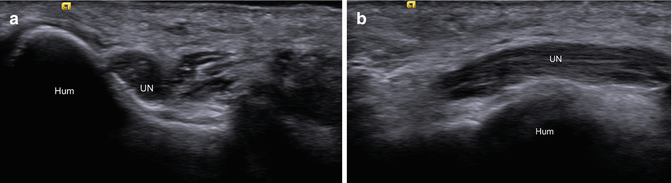

Fig. 5.52
Ulnar neuropathy. (a) Short- and (b) long-axis ultrasound images of the posterior elbow demonstrate enlargement of and decreased echogenicity within the ulnar nerve (UN) in the setting of ulnar neuropathy; humerus (Hum)
The role of ultrasound is to evaluate for a potentially treatable cause of ulnar neuritis, to assess for subluxation or dislocation, and to provide guidance for therapeutic injection. Although frequently no cause of compression is seen, other causes besides an anconeus epitrochlearis include synovitis, soft tissue masses, and findings related to valgus extension overload as seen in the throwing athlete, including osteophytes and joint bodies (Wilson et al. 1983).
The advantage of ultrasound over MRI is the ability to perform dynamic imaging. This is particularly important in evaluating for ulnar nerve subluxation where the nerve abnormally dislocates medially over the medial epicondyle during flexion (Fig. 5.53) and “snapping triceps” where the medial head of the triceps subluxates medially during flexion, resulting in medial dislocation of the ulnar nerve. This latter entity has been described in weight lifters (Husarik et al. 2009; Jacobson et al. 2010; O’Driscoll et al. 1991; Okamoto et al. 2000).
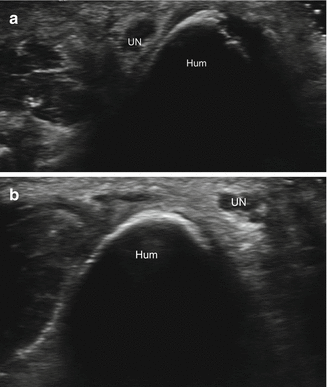

Fig. 5.53
Ulnar nerve subluxation. Short-axis ultrasound images of the posterior elbow in extension (a) and flexion (b) demonstrate subluxation of the ulnar nerve (UN) from lateral (a) to medial (b) with respect to the distal humerus (Hum)
5.5.6 Olecranon Bursitis
In addition to bicipitoradial bursitis, olecranon bursitis is readily assessed with ultrasound. In the athlete, this is frequently secondary to trauma. The collapsed bursa is not easily identified; however, in the setting of olecranon bursitis, there will be distension of the bursa with anechoic or hypoechoic fluid that may contain internal debris (Fig. 5.54). If the fluid is more hyperechoic, a hemorrhagic bursitis should be considered (Jacobson 2013b).
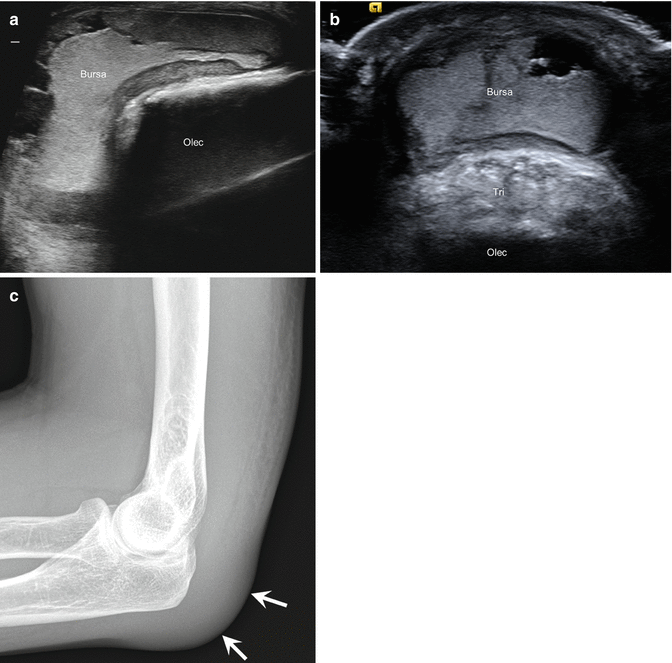

Fig. 5.54
Olecranon bursitis. (a) Long-axis extended field of view and (b) short-axis ultrasound images of the posterior elbow demonstrate marked olecranon bursitis with distension of the bursa (Bursa) with echogenic, hemorrhagic fluid; triceps tendon (Tri), olecranon (Olec). (c) Lateral radiograph of the elbow shows soft tissue swelling (arrows) in the location of the olecranon bursa
5.6 Wrist and Hand (Including Finger) Ultrasound
Ultrasound is an ideal modality for the evaluation of sports-related injuries in the hand and wrist because of the development of high-frequency transducers and the superficial location of the structures (Teefey et al. 2000a, b, c). In addition, dynamic imaging may be performed to identify abnormalities of tendon motion or joint instability.
5.6.1 Scapholunate Ligament
The dorsal component of the scapholunate ligament is the more clinically relevant portion of the ligament and an important stabilizer of the carpus. The dorsal fibers are readily assessed with sonography appearing as a hyperechoic, fibrillar band. Tears appear as focal disruptions of the normal hyperechoic ligament with a focal area of decreased echogenicity (Jacobson et al. 2002). In the setting of chronic, repetitive injury, the ligament may be abnormally thickened and hypoechoic (Jacobson et al. 2002; Renoux et al. 2009). There may be associated widening of the scapholunate interval that may be accentuated with clenching of the fist. A ganglion cyst (Fig. 5.55) may also develop adjacent to the tear with fluid decompressing from the joint through a tear (Cardinal et al. 1994). This may cause compression of the distal portion of the posterior interosseous nerve.
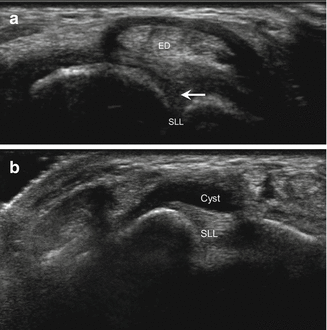

Fig. 5.55
Scapholunate ligament pathology. (a) Short-axis ultrasound image of the dorsal wrist demonstrates a hypoechoic tear (arrow) of the scapholunate ligament (SLL); extensor digitorum tendon (ED). (b) Short-axis ultrasound image of the scapholunate ligament (SLL) with an associated ganglion (Cyst)
5.6.2 Scaphoid Fracture
5.6.3 Extensor Tendons
Abnormalities of the tendons can be readily assessed with ultrasound including tenosynovitis, tendinosis, and tendon tears. Tenosynovitis refers to distension of the sheath surrounding a tendon with fluid and/or soft tissue (Fig. 5.56). The fluid may be anechoic or more heterogeneous and complex due to synovial hypertrophy, hemorrhage, or debris (Daenen et al. 2004; Teefey et al. 2000a, b, c). In the setting of synovial hypertrophy, there may be hyperemia as demonstrated in color or power Doppler interrogation (Fig. 5.56) (Breidahl et al. 1998; Teefey et al. 2000a, b, c).
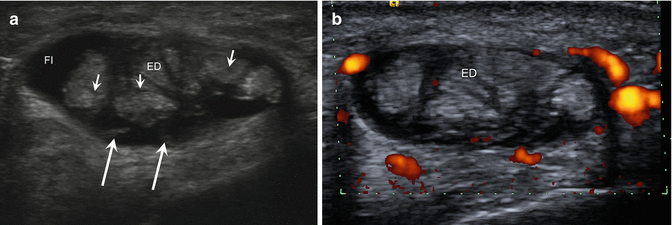

Fig. 5.56
Extensor tendinosis/tenosynovitis of the wrist. (a) Short-axis ultrasound image of the extensor digitorum tendon (ED) demonstrates mild tendinosis of the tendons with hypoechoic areas (short arrows) with fluid (Fl) distension of the tendon sheath and synovitis (long arrows). (b) Same image with power Doppler interrogation shows peritendinous hyperemia in the setting of inflammation
Tendinosis manifests as focal or diffuse enlargement of the affected tendon with areas of decreased echogenicity (Fig. 5.56). Foci of calcification may be present (Daenen et al. 2004). Tendon rupture may be partial or complete and appears as a fluid-filled cleft within the affected tendon with disruption of the normal fibers (Fig. 5.57) (Daenen et al. 2004). The fluid may be anechoic or hypoechoic and complex in the setting of hemorrhage due to acute injury. With complete rupture, there may be retraction and separation of the torn ends of the tendon (Daenen et al. 2004).
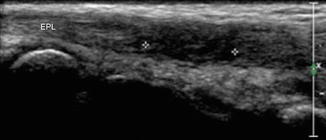

Fig. 5.57
Extensor pollicis longus tear. Longitudinal ultrasound image of the wrist shows a high-grade partial tear (++) of the extensor pollicis longus (EPL)
De Quervain’s tenosynovitis is a common disorder that describes tendinosis and tenosynovitis of the first extensor compartment of the wrist that includes the abductor pollicis longus (APL) and extensor pollicis brevis (EPB) tendons. It is frequently seen due to chronic repetitive stress and overuse in athletes, such as tennis players (Daenen et al. 2004). At sonography, there will be tendinosis of the APL and EPB tendons with tendon enlargement and decreased echogenicity with loss of the normal fibrillar pattern. There may be linear foci of decreased echogenicity reflecting interstitial microtears (Daenen et al. 2004). There will be distension of the tendon sheath with anechoic or hypoechoic fluid (Fig. 5.58) (Daenen et al. 2004). This is often accompanied by thickening of the corresponding extensor retinaculum.
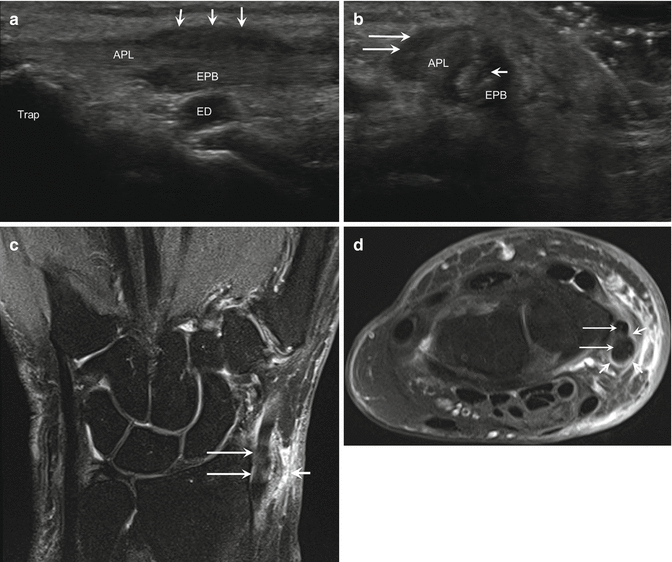

Fig. 5.58
De Quervain’s tenosynovitis. (a) Long- and (b) short-axis ultrasound images of the first extensor compartment of the wrist show tendinosis of the abductor pollicis longus (APL) and extensor pollicis brevis (EPB) with a split tear (short arrow) of the EPB and synovial thickening (long arrows); trapezium (Trap), radial artery (RA). (c) Coronal and (d) axial fluid-sensitive MRIs of the same patient show findings of De Quervain’s tenosynovitis including enlargement of the abductor pollicis longus and extensor pollicis brevis tendons (long arrows) with surrounding fluid (arrow)
The extensor hood stabilizes the extensor tendons of the hand at the level of the metacarpophalangeal (MCP) joints, maintaining them in place through the range of motion during flexion and extension (Kichouh et al. 2011). The sagittal bands are the most important stabilizing structures of the extensor hood (Clavero et al. 2003; Drapé et al. 1994; Scott 2000; Young and Rayan 2000). Ruptures of the sagittal bands can occur following trauma to the MCP joint as seen in “boxer knuckle.” This can result in subluxation or dislocation of the extensor tendon at the level of the MCP joint (Kichouh et al. 2011). On short-axis sonographic imaging, the normal sagittal bands appear as linear hyperechoic bands along both sides of the extensor tendon at the level of the MCP joint (Fig. 5.59) (Kichouh et al. 2011). Injury to the sagittal bands may appear as focal disruption of the normal hyperechoic band and/or focal hypoechoic thickening (Fig. 5.59) (Kichouh et al. 2011). There may be subluxation of the extensor tendon due to the injury. There may also be associated injuries to the extensor tendon itself (Kichouh et al. 2011).
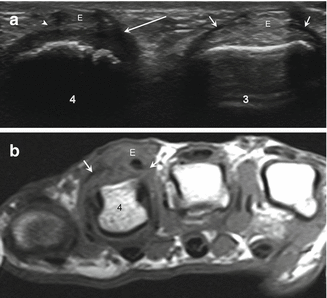

Fig. 5.59
Normal and torn extensor hood. (a) Short-axis ultrasound image shows disruption of the radial (long arrow) and ulnar (arrowhead) sagittal bands of the fourth finger (4) extensor hood. Normal-appearing sagittal bands (short arrows) are seen in the third finger (3); extensor tendon (E). (b) Axial proton density-weighted MRI shows disruption of the sagittal bands of the extensor hood (arrows) with subluxation of the extensor tendon (E) of the fourth finger (4)
5.6.4 Flexor Tendons
Abnormalities of the flexor tendons of the wrist and hand are similar to those affecting the extensor tendons, including tenosynovitis, tendinosis, and tendon tears. The sonographic findings of flexor tendon pathology are similar to those previously described in the extensor tendons (Fig. 5.60).
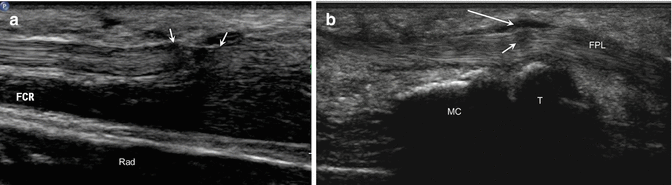

Fig. 5.60
Wrist flexor tendon pathology. (a) Longitudinal ultrasound image of the flexor carpi radialis tendon (FCR) shows a high-grade partial tear (arrows); radius (Rad). (b) Long-axis ultrasound image of the wrist in a different patient shows tendinosis and tenosynovitis (long arrow) of the flexor pollicis longus tendon (FPL) with interstitial tearing (short arrow); metacarpal (MC), trapezium (T)
5.6.5 Triangular Fibrocartilage
The normal triangular fibrocartilage complex (TFCC) is a hyperechoic structure extending from the base of the ulnar styloid process to the radius. The deeper, radial attachment may be more difficult to see, although utilizing a lower-frequency transducer may help (Chiou et al. 1998; Jacobson 2013c; Taljanovic et al. 2011). The ulnar attachment of the TFCC is located subjacent to the extensor carpi ulnaris tendon. Tears of the TFCC appear as areas of hypoechogenicity, typically linear, or areas of thinning of the TFCC (Fig. 5.61) (Keogh et al. 2004).
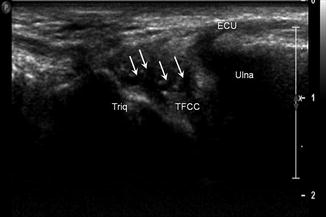

Fig. 5.61
Triangular fibrocartilage tear. Longitudinal ultrasound image of the medial wrist shows irregular hypoechoic tearing (arrows) of the triangular fibrocartilage complex (TFCC); triquetrum (Triq), extensor carpi ulnaris (ECU)
5.6.6 Ulnar Collateral Ligament of the Thumb
Injury to the ulnar collateral ligament (UCL) of the thumb is known as gamekeeper’s thumb or skier’s thumb (Moschilla and Breidahl 2002). The normal ulnar collateral ligament of the thumb appears as a hyperechoic fibrillar band along the ulnar aspect of the metacarpophalangeal joint of the thumb (Fig. 5.62). The ligament is located deep to the adductor aponeurosis that appears as a smooth, thin echogenic band. Tears of the UCL appear as partial or complete hypoechoic disruptions with loss of the normal ligament architecture (Fig. 5.62). The ligament may also appear enlarged and hypoechoic (Ebrahim et al. 2006; Jacobson 2013c; Moschilla and Breidahl 2002). The torn ligament may be proximally retracted appearing as a round hypoechoic mass adjacent to the metacarpal head (Hergan et al. 1995). There may be an associated hyperechoic avulsion fragment from the base of the proximal phalanx that remains attached to the torn ligament (Fig. 5.62) (Moschilla and Breidahl 2002).
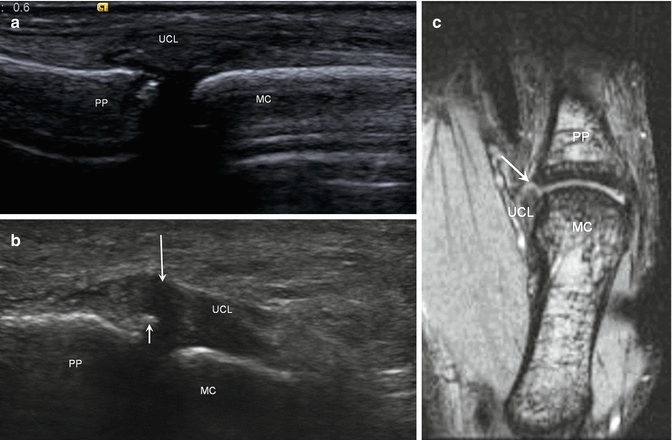

Fig. 5.62
Ulnar collateral ligament, thumb. (a) Longitudinal ultrasound image of the thumb shows a normal, echogenic, and intact ulnar collateral ligament (UCL). (b) Longitudinal ultrasound image shows a complete, nonretracted tear (long arrow) of the UCL with a small avulsion fracture (short arrow). (c) Coronal gradient echo MRI shows a tear (arrow) at the insertion of the UCL; proximal phalanx (PP), metacarpal (MC)
A subtype of gamekeeper’s thumb is a Stener lesion that is defined as a full-thickness and displaced tear of the UCL. A Stener lesion is present when the torn, retracted UCL displaces superficial to the adductor aponeurosis (Ebrahim et al. 2006). This distinction is clinically relevant as Stener lesions are usually treated with surgery (O’Callaghan et al. 1994). Sonographic images demonstrate a nodular focus of tissue of mixed echogenicity at the level of the metacarpal head reflecting the proximally retracted UCL. The retracted ligament is displaced superficial to the adductor aponeurosis that becomes distorted and bulges with a convex surface due to the overlying retracted ligament (Fig. 5.63) (Ebrahim et al. 2006). The appearance has been referred to as a yo-yo on a string (Ebrahim et al. 2006).
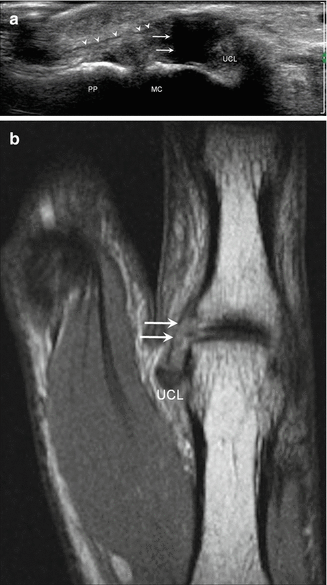

Fig. 5.63
Stener lesion. (a) Longitudinal ultrasound image of the thumb shows a complete tear (arrows) of the ulnar collateral ligament (UCL) with proximal retraction and partial displacement superficial to the adductor aponeurosis (arrowheads); proximal phalanx (PP), metacarpal (MC). (b) Coronal proton density-weighted MRI of the thumb demonstrates a Stener lesion with a torn, proximally retracted UCL superficial to the adductor aponeurosis (arrows)
Dynamic imaging with slight valgus stress placed on the MCP joint may help elucidate complete, nonretracted tears from partial tears (Jacobson 2013c).
5.6.7 Finger Pulleys
Ultrasound may also be used to evaluate the pulley system of the fingers (Boutry et al. 2005). The pulleys help to stabilize the flexor tendons during finger flexion and prevent subluxation or bowing (Boutry et al. 2005). The normal pulley (Fig. 5.64) appears as a linear hyperechoic band located superficial to the flexor tendon, although it may appear relatively hypoechoic adjacent to fat (Boutry et al. 2005; Martinoli et al. 2000). They may demonstrate anisotropy (Boutry et al. 2005). The A2 pulley located along the proximal phalanx is the most commonly injured pulley. This injury is typically seen in rock climbers but may be seen in other athletes such as football players (Moschilla and Breidahl 2002). Ultrasound shows focal hypoechoic disruption of the normal hyperechoic pulley. There may be thickening of and diffuse hypoechogenicity within the injured pulley. There will be volar displacement of the flexor tendon that becomes more pronounced during finger flexion known as bowstringing (Fig. 5.64) (Jacobson 2013c; Klausner et al. 2002; Martinoli et al. 2000; Moschilla and Breidahl 2002).
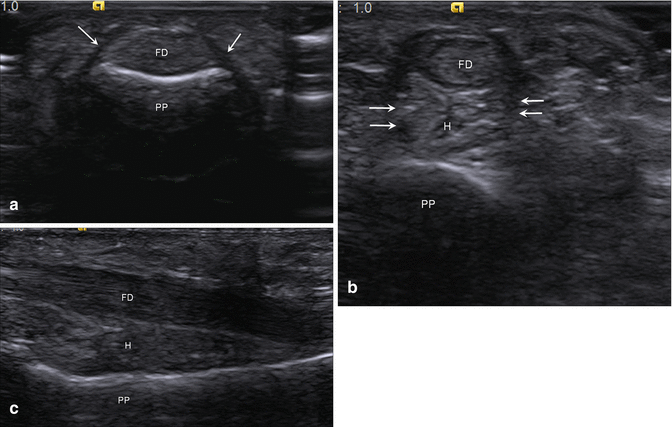

Fig. 5.64
Pulley apparatus of the finger. (a) Short-axis ultrasound image of the index finger shows the normal A2 pulley (arrows) maintaining the flexor digitorum tendons (FD) adjacent to the proximal phalanx (PP). (b) Short- and (c) long-axis ultrasound images in the setting of a completely disrupted A2 pulley (arrows) with displacement of the flexor tendons (FD) away from the proximal phalanx (PP) with intervening hemorrhage (H)
5.6.8 Carpal Tunnel
The carpal tunnel may also be assessed sonographically. Carpal tunnel syndrome is an entrapment neuropathy of the median nerve within the fibro-osseous carpal tunnel (Sernik et al. 2008). Anything that results in decreased size of the tunnel or increase in the tunnel contents may result in compression of the median nerve. These include tenosynovitis of the flexor tendons, masses, or trauma although frequently a cause is not found (Chen and Liang 1997; Duncan et al. 1999).
On imaging, there is enlargement of the median nerve within the carpal tunnel with decreased echogenicity and loss of the normal honeycomb appearance. Enlargement of the nerve of greater than 12 mm2 is considered abnormal (Fig. 5.65) (Chen et al. 1997; Klauser et al. 2009). Values between 10 and 12 mm2 are indeterminate. There may be flattening of the nerve with bowing of the overlying flexor retinaculum (Chen et al. 1997).
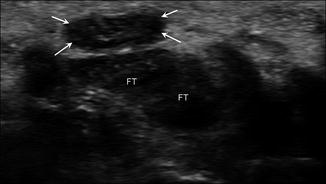

Fig. 5.65
Carpal tunnel syndrome. Short-axis ultrasound image of the wrist demonstrates an enlarged, hypoechoic median nerve (arrows) within the carpal tunnel adjacent to the flexor tendons (FT)
5.6.9 Ganglia of the Wrist and Hand
Ganglion cysts frequently occur in the hand and wrist and may be evaluated sonographically. Most commonly, they occur along the dorsal aspect of the wrist, superficial to the scapholunate ligament (Fig. 5.66) (Bianchi et al. 1994a). Along the volar aspect of the wrist, ganglion cysts commonly occur between the radial artery and flexor carpi radialis tendon (Fig. 5.67) (Bianchi et al. 1994a). Within the fingers, ganglia may occur as retinacular cysts related to the A1 or A2 pulleys. Ultrasound may be used to guide aspiration and therapeutic injection with corticosteroid as a treatment for ganglion cysts (Breidahl and Adler 1996).
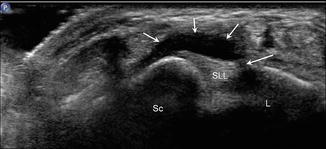
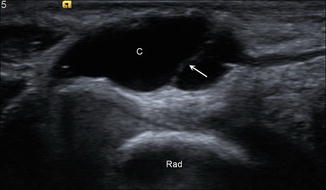

Fig. 5.66
Dorsal wrist ganglion cyst. Short-axis ultrasound image of the dorsal wrist demonstrates a ganglion cyst (short arrows) arising from a tear (long arrow) in the scapholunate ligament (SLL); scaphoid (Sc), lunate (L)

Fig. 5.67
Volar wrist ganglion cyst. Short-axis ultrasound image of the volar wrist shows a slightly complex ganglion cyst (C) with a thin internal septation (arrow) superficial to the radius (Rad)
5.7 Hip and Thigh Ultrasound
5.7.1 Snapping Hip Syndrome
Snapping hip syndrome is an abnormal snapping or clicking sensation associated with certain hip motions. It is frequently seen in young, athletic adults and is more common in women. There may be pain associated with the snapping sensation (Deslandes et al. 2008). The syndrome can be further subdivided into intra-articular causes, including labral tears, chondral abnormalities, or joint bodies (Fig. 5.68), and extra-articular causes. The extra-articular causes may involve the iliopsoas tendon anteriorly (internal snapping hip) or the iliotibial band and gluteus maximus tendons laterally (external snapping hip) (Choi et al. 2002; Pelsser et al. 2001; Schaberg et al. 1984). Although there may be bursitis and tendinopathy associated with the affected tendons, frequently these findings are absent, and the only abnormality is the pathologic motion of the tendons. The kinematic nature of snapping hip limits the utility of MRI in these patients, making dynamic ultrasound critical to the diagnosis of snapping hip syndromes (Pfirrmann et al. 2008).
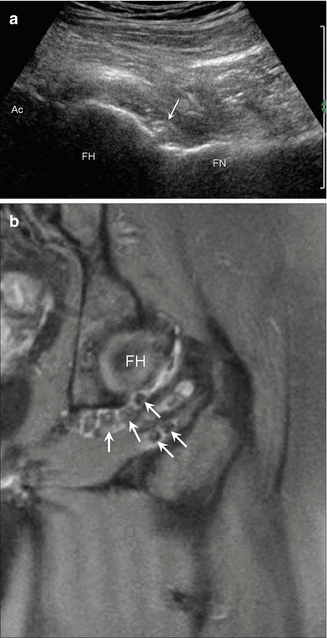

Fig. 5.68
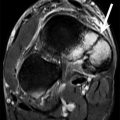
Hip intra-articular bodies. (a) Long-axis ultrasound image along the anterior hip shows an echogenic intra-articular body (arrow) adjacent to the femoral head (FH)/femoral neck (FN) junction; acetabulum (Ac). (b) Coronal fluid-sensitive MRI in a different patient with the same pathology shows multiple hypointense joint bodies (arrows) adjacent to the femoral head (FH)
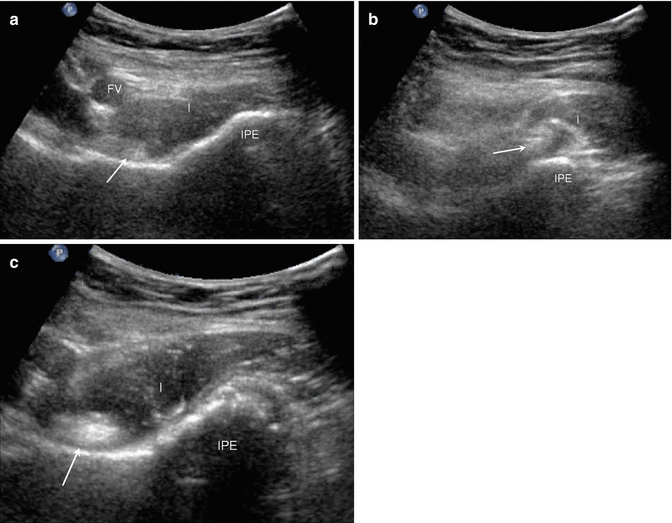
Under normal circumstances, the psoas muscle glides laterally, rolling over the medial border of the iliacus muscle. With internal snapping hip, during abduction/flexion/external rotation, the iliopsoas tendon moves abruptly over the iliacus muscle, which becomes entrapped between the tendon and the iliopectineal eminence. As the hip returns to the neutral position, the tendon “snaps” over the muscle onto the subjacent pubic bone (Fig. 5.69) (Deslandes et al. 2008). The tendon may also abruptly flip over the femoral vessels during abduction/flexion/external rotation (Fig. 5.70). In addition to the findings at dynamic imaging, there may be iliopsoas tendinosis with enlargement and decreased echogenicity of the tendon with fluid distension of the bursa (Pelsser et al. 2001).