Fig. 1
Theories of blunt traumatic aortic injuries (From ‘Blunt Aortic Injury’ Neschis DG et al. (2008) NEJM 359:1708–1716. Copyright © (2008) Massachusetts Medical Society. Reprinted with permission of the Massachusetts Medical Society)
Depending on the forces in play, the aorta injury site can be the root, ascending aorta, arch, isthmus, descending aorta or even abdominal aorta (less common). Multiple studies in deceased and surviving patients show the aortic isthmus is the most common location of injury. However, since studies are not inclusive of all blunt trauma-related fatalities, there may be a shift in numbers with more injuries potentially occurring at the aortic root. A considerable number of patients has aortic injury at multiple sites, in the autopsy series by Teixeira et al. amounting up to 18 % (Teixeira et al. 2011) (Fig. 2). In that study, 2 % of injuries were located at the aortic root, 3 % at the ascending aorta and 11 % at the arch. Mention is warranted for the other upper thoracic major vessels, especially the aortic arch branches, which can be injured in isolation. In a small sample study by Chen et al., 38 % of injuries involved the proximal aortic arch branching vessels (Chen et al. 2001). Even though the percentage likely is lower, injury to these vessels is important to diagnose and treat if necessary.
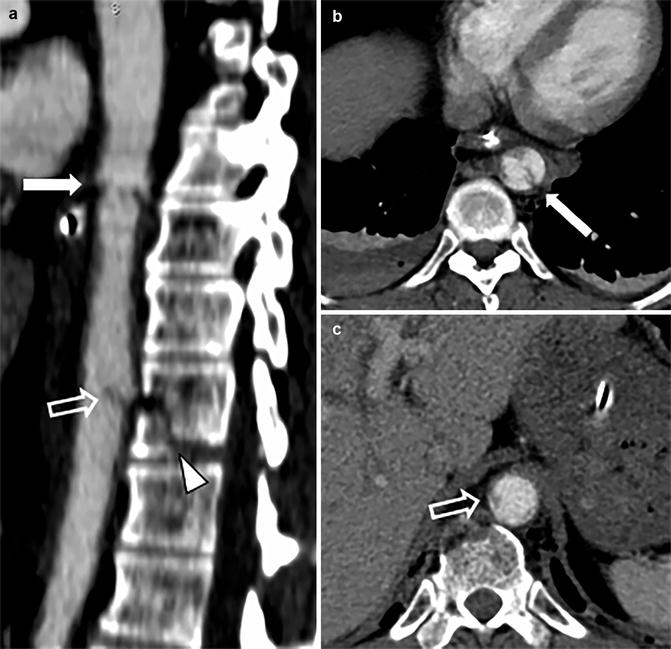

Fig. 2
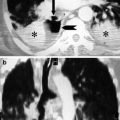
Example of multilevel blunt traumatic aortic injury on sagittal CTA (a) with corresponding axial CTA slides (b, c). The proximal intimal injury (solid white arrow) was most likely caused by a stretch mechanism; the distal intimal flap (open white arrow) may have been caused by stretch and/or direct impact from the nearby vertebral fracture (arrow head)
3 Terminology and Clinical Issues
The diagnostic and treatment process in polytrauma patients usually takes place in multidisciplinary teams with members of varying level of experience. In combination with stress because of time concerns and multitude of injuries, clear communication is paramount. Therefore, the most important point to make about terminology is that all members of the resuscitation team, including radiology staff, should align the vocabulary used throughout the institution. This also includes which of the many classification schemes is used to grade the injury.
As described in the introduction, many different terms are used in literature to indicate traumatic injury to the aorta after blunt trauma. Further confusion may arise with other terminologies in this setting. Specific terms to be aware of with regard to diagnosis and treatment are mediastinal haematoma, peri-aortic haematoma, minimal aortic injury, pseudo-aneurysm and secondary signs of injury (SSI). We will describe these terms here to minimize confusion.
Mediastinal haematoma is used to describe haematoma within the mediastinum with a fat plane preserved around the aortic adventitia. These haematomas are not considered to be associated to aortic injury, but originate from other sources of bleeding, like mediastinal veins or fractures of vertebrae or sternum (Forman et al. 2013; Raptis et al. 2015) (Fig. 3).
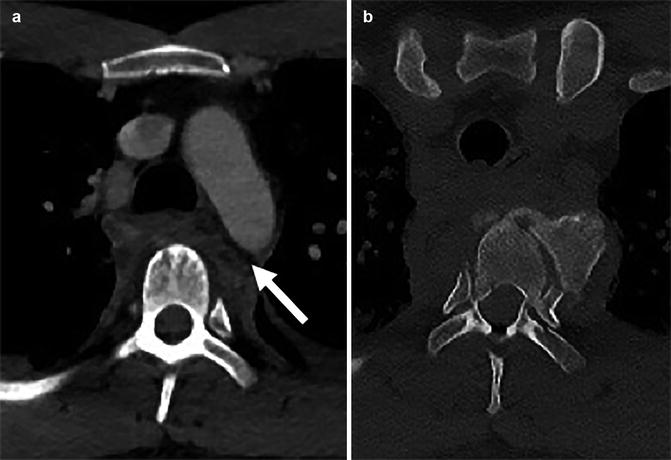

Fig. 3
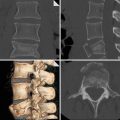
Example of mediastinal haematoma (a) due to thoracic vertebral body fracture dislocation (b). Note the fat plane between the aorta and mediastinal haematoma (arrow). The fat plane allows to differentiate more distant mediastinal haematoma from peri-aortic haematoma
Peri-aortic haematoma is used to describe haematoma directly bordering the aortic adventitia (i.e. loss of peri-aortic fat plane) and either confined to the direct peri-aortic region or extending beyond into the mediastinum (Fig. 4). Although previously reported to occur as a false-positive indirect sign of aortic injury (Steenburg and Ravenel 2008), advances in modern CT techniques pick up previously undetected small injuries. If peri-aortic haematoma is present, the aorta should be scrutinized for presence of injury. If peri-aortic haematoma is absent, this nearly excludes higher-grade aortic injury. However, lesser grade injury is still possible if peri-aortic haematoma is absent, around 21–22 % of such cases occurring without surrounding haematoma (Forman et al. 2013; Aladham et al. 2010).
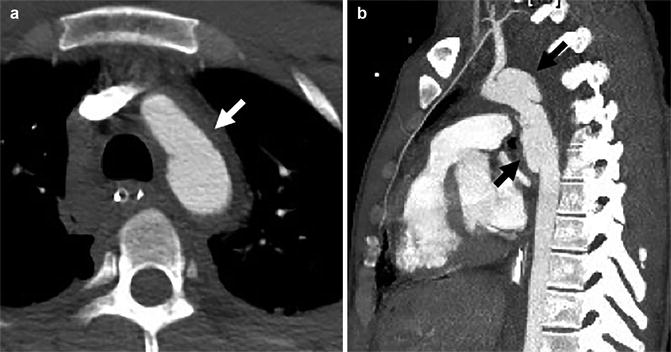

Fig. 4
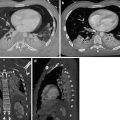
Axial (a) and sagittal-oblique (b) CTA demonstrating peri-aortic haematoma (solid white arrow) directly abutting the aortic adventitia (loss of peri-aortic fat plane), caused by a long, bilobar pseudo-aneurysm (solid black arrows)
Minimal aortic injury (MAI, also known as minor aortic injury) is a term used to describe a lesser grade of aortic injury that may resolve with conservative measures, including monitoring, blood pressure control and repeat CT. However, multiple groups that publish on the topic define the term differently. The definition ranges from injuries in which outer aortic wall contour is preserved to those including pseudo-aneurysms of up to 50 % of the normal aorta diameter (Forman et al. 2013; Caffarelli et al. 2010; Rabin et al. 2014a).
Pseudo-aneurysm relates to an aortic injury in which there is (near) complete loss of aortic wall integrity resulting in a regular or an irregular deformity of the outer wall of the aorta (Fig. 5). Especially if there is surrounding haematoma with mass effect on the mediastinum, there is a high chance of rupture (Rabin et al. 2014a, b).
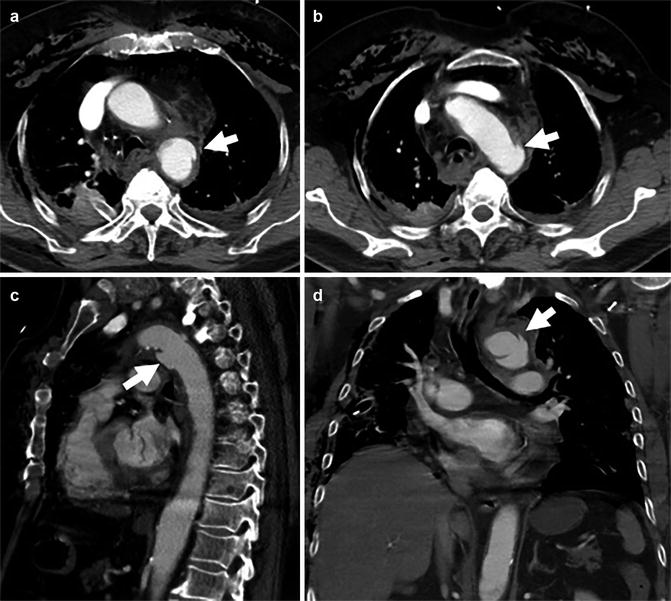

Fig. 5
Axial (a, b), sagittal (c) and coronal (d) CTA of a 57-year-old man involved in an MVA. Images depict a proximal thoracic aortic pseudo-aneurysm (arrows). Moderate peri-aortic haematoma and extensive subcutaneous emphysema are also present (not marked)
Secondary signs of injury (SSI) were recently defined in a study that sought parameters for successful nonoperative management of BTAI as pseudo-coarctation, extensive mediastinal haematoma (with mass effect) and large left haemothorax (Rabin et al. 2014a, b) (Fig. 6). They concluded that grade 3 aortic injuries (pseudo-aneurysms involving more than 50 % the circumference of the aorta in their study) with SSI needed urgent repair, whereas those without SSI could undergo delayed repair (Rabin et al. 2014a). The use of SSI therefore seems to aid management decisions.
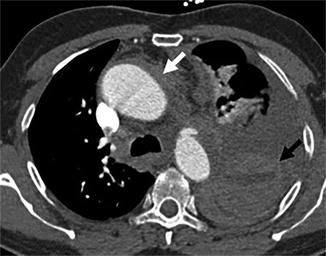

Fig. 6
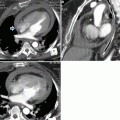
Example of BTAI with peri-aortic haematoma bordering the ascending (solid white arrow) and descending thoracic aorta with large left-sided haemothorax, caused by BTAI at the aortic isthmus. Note the different densities indicating cloth formation (solid black arrow)
3.1 Classification of BTAI
To grade blunt traumatic aortic injury, several classification systems have been published, the first of which was by Parmley et al. in 1958 on the basis of a large autopsy study (Parmley et al. 1958). Gavant et al. published the first classification system based on CT images in 1999, very comprehensive but somewhat difficult to use (Gavant and Helical 1999). In 17 years since, six more groups published classification systems with different degree of variation, namely, Simeone et al. (2006), Azizzadeh et al. (2009), Caffarelli et al. (2010), Lamarche et al. (2012), Starnes et al. (2012) and Rabin et al. (2014a). The multitude of different classification systems likely reflects local experiences with treatment choices, as well as the rapid improvement of CT technology and endovascular stent grafts used for treatment. In 2011, the Society for Vascular Surgery published clinical practice guidelines for endovascular repair of BTAI (Lee et al. 2011), using the classification system by Azizzadeh et al. (2009). However, this was before other classification systems were published. The Vancouver Simplified classification system in the study by Lamarche et al. found better interobserver agreement than the Simeone and Gavant classifications, the reason for which we like to use it (Lamarche et al. 2012). As mentioned before, the most important issue for all team members involved in caring for BTAI patients remains to make sure to use the same terminology and classification.
The Vancouver classification, like most other classifications, has four grades (Fig. 7). Grade 1 and grade 2 essentially have preserved outer aortic contours and consist of luminal thrombus, intimal flap and/or intramural haematoma, these lesions in grade 1 being smaller than 1 cm and in grade 2 larger than 1 cm. Grade 3 and grade 4 lesions demonstrate abnormality of the outer wall of the aorta, with grade 3 lesions being pseudo-aneurysms of any size and grade 4 lesions showing frank contrast extravasation. Given the fact that some publications show good results of conservative management as treatment for smaller pseudo-aneurysms, caregivers may elect to use other classification systems.
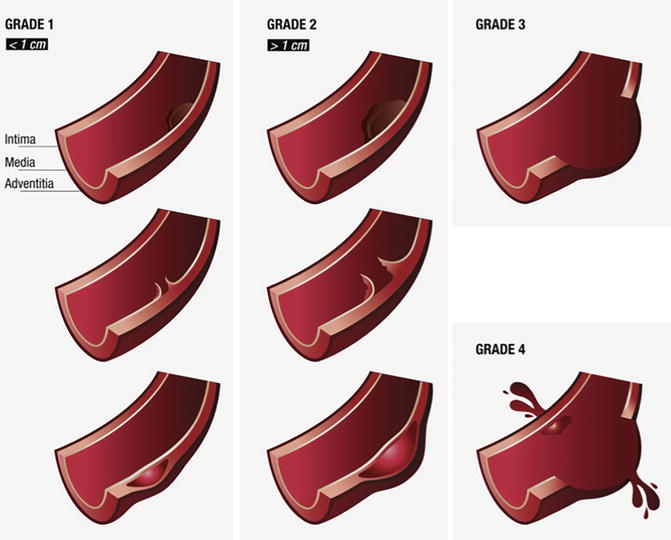

Fig. 7
Vancouver classification of blunt traumatic aortic injury. Grade 1 injuries (luminal thrombus, intimal flap or intramural haematoma) are less than 1 cm; if larger than 1 cm, injuries are grade 2; aortic pseudo-aneurysms are grade 3; and grade 4 are injuries with active contrast extravasation indicating ongoing haemorrhage (From Lamarche et al. (2012), Reply to the Editor, copyright 2012. Reprinted with permission from Elsevier)
3.2 Clinical Issues
Clinical symptoms and signs are non-specific for BTAI, nor sensitive. Patients may have chest pain, back pain or difficulty breathing, and signs include external chest wall injuries, systemic hypotension, concomitant upper limb hypertension with lower limb hypotension or a substantial difference in blood pressures between the right and left brachial arteries (present in up to 50 % of patients with BTAI). Some patients may present initially with no clinical signs but rapidly develop hemodynamic instability (Steenburg et al. 2008; Raptis et al. 2015). Factors negatively influencing prognosis are a systolic blood pressure <90 mmHg and hypothermia with T < 35 °C and injury severity score (ISS) >25 (Arthurs et al. 2009).
As stated before, blunt traumatic aortic injury invariably is the result of high-energy trauma to the chest and most often is not confined to this anatomic area. In most cases, significant concomitant injuries are present. Depending on the severity of the aortic injury, the hemodynamic stability of the patient and the severity of the other injuries, treatment priorities have to be decided. If possible, repair of BTAI should be delayed for favourable outcome, especially in concomitant head injuries (Fox et al. 2015; Rabin et al. 2014b). This will also increase the time to carefully evaluate the extent of other injuries. If aortic injury and traumatic brain injury coexist, research has shown that the most unstable injury should be prioritized to immediate care. It also suggests that intracranial haemorrhage is not a contraindication to endovascular aortic stenting or an absolute contraindication to systemic anticoagulation during this procedure (Fox et al. 2015; Kitagawa et al. 2013).
To predict which patients need urgent repair instead of delayed repair, Harris et al. proposed a new aortic injury score to predict early rupture (Harris et al. 2015). From their study, they conclude that a patient is at high risk of early rupture if any two of the following three factors are present: lactate >4 mM, posterior peri-aortic haematoma >10 mm or lesion/normal aortic ratio >1.4 (meaning an aortic diameter more than 40 % increased compared to the nearest normal aortic diameter). Whether these criteria are better predictors of the need of urgent repair of grade 3 injuries than the secondary signs of injury (SSI) defined by Rabin et al. (2014a, b) (see before) remains to be seen. Likely, the criteria of both study groups overlap, but these findings will help select patients that need urgent instead of delayed treatment.
4 Imaging
Post-intravenous contrast CT angiography (CTA) is the standard diagnostic test for detection of thoracic aortic and branch vessel injury (Fox et al. 2015). The sensitivity is 96 %, specificity is 100 % and negative predictive value 99.9 %. These percentages are by far superior when compared to plain chest radiography, transoesophageal echography (TEE) or conventional arch aortography (Steenburg and Ravenel 2008). Furthermore, CTA is very accurate in detection of most other chest injuries, which exist with or without the presence of BTAI. However, other imaging modalities can be useful, so we will discuss strengths, weaknesses and findings of those as well.
4.1 Radiography
Although CT is increasingly being used as the primary imaging modality in trauma resuscitation, chest radiography (CXR) in many centres is still used as the first adjunct to the primary survey. Signs seen on plain films are either sensitive or specific, but never both; therefore, utility of CXR in the setting of suspected BTAI is low (Nagy et al. 2000; Cook et al. 2001; Gutierrez et al. 2016). Especially in the case of MAI, modern CTA can demonstrate BTAI without changes to the outer wall of the aorta and even without bordering haematoma, further underscoring the potential to miss these injuries on CXR. However, signs can still be present on a conventional chest X-ray and, if so, prompt for further evaluation with CTA (Fig. 8).
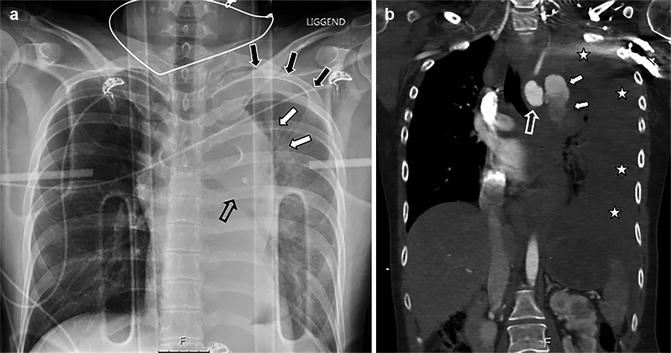

Fig. 8
A 27-year-old woman struck by a car while on moped. Supine AP chest X-ray taken during resuscitation with unsuspected findings consistent with aortic injury (a): indistinct aortic contour (white solid arrows), depressed left main bronchus (open black arrow) and apical cap consistent with haemorrhage extending to the pleural space (solid black arrows). Injury was proven with CT (b): aortic arch (open white arrow) demonstrates frank extravasation (solid white arrows) into the pleural cavity (white asterisks), consistent with grade 4 injury. Note the homogenous low density of the haemothorax, indicating hyperacute exsanguination with lack of time to form cloth. Patient did not survive to treatment
Signs in decreasing order of sensitivity according to Cook et al. are as follows: mediastinal width >8 cm (sens 90 %, spec 30 %), mediastinum-to-chest width ratio >0.25 (sens 90 %, spec 6 %), opacified AP window (sens 90 %, spec 51 %), irregular aortic knob (sens 80 %, spec 68 %), blurred aortic contour (sens 70 %, spec 53 %), nasogastric tube deviation (sens 50 %, spec 91 %) and trachea shifted to patient’s right (sens 40 %, spec 86 %) (Cook et al. 2001).
Signs in decreasing order of specificity are as follows: thoracic spine fracture (spec 93 %, sens 11 %), first rib fracture (spec 91 %, sens 10 %), NG tube deviation (spec 91 %, sens 50 %), depressed left main bronchus (spec 90 %, sens 10 %), wide left paraspinal line (spec 90 %, sens 29 %), clavicle fracture (spec 87 %, sens 0 %), trachea shift to patient’s right (spec 86 %, sens 40 %), left apical cap (spec 80 %, sens 20 %) and pulmonary contusion (spec 72 %, sens 30 %) (Cook et al. 2001).
Rather than spending a lot of time analysing all separate findings and measurements, a general assessment of the mediastinum and decision to ‘normal’ or ‘abnormal’ results in better sensitivity and interobserver agreement (Ho et al. 2002). If the only abnormality seen on an initial screening supine anteroposterior CXR is mediastinal width more than 8 cm, it can be worthwhile repeating the CXR standing and posteroanterior, in which case, 38 % of exams will be normal (Schwab et al. 1984). However, we fully agree with Raptis et al. and others that patients with an abnormal CXR, patients with high index of suspicion for BTAI or patients with chest pain out of proportion to their known injuries after appropriate trauma need to proceed to CTA of the chest (Raptis et al. 2015; Fox et al. 2015).
4.2 Angiography/Aortography
The publication of the Eastern Association for the Surgery of Trauma (EAST) practice management guideline for the diagnosis and management of blunt aortic injury in 2000 still defined angiography as the ‘gold standard’ for the diagnosis of BTAI. However, the workgroup already acknowledged that ‘CT scanning is taking more of a role, especially for screening’ (Nagy et al. 2000). Since their publication, rapid developments of CT techniques have rendered CTA more sensitive than conventional angiography, mostly due to the detection of extra-luminal abnormalities, either confined to the aortic wall or beyond. This has caused the same society to revise their guidelines, which in their publication in 2015 strongly recommend CT with intravenous contrast for the diagnosis of BTAI (Fox et al. 2015). Angiography is of course still used if endovascular treatment for BTAI is undertaken (Fig. 9).
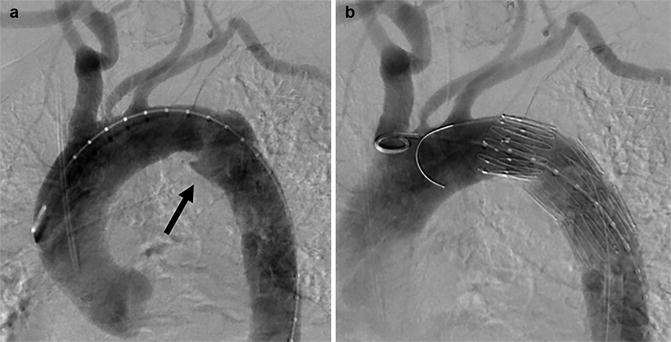

Fig. 9
Thoracic aorta angiogram in left anterior oblique view before (a) and after TEVAR (b) for aortic pseudo-aneurysm at the isthmus (arrow)
4.3 Abdominal and Transthoracic Ultrasound
During trauma resuscitation, many centres will include an ultrasound scan according to the focussed abdominal sonography in trauma (FAST) as an adjunct to the primary survey. A FAST examination essentially is a quickly performed ultrasound to demonstrate the presence or absence of free intraperitoneal fluid by evaluating the four abdominal quadrants. In addition, one can look for presence of pericardial effusion, pleural effusion and/or pneumothorax in a reasonably quick and reliable fashion, the so-called extended FAST (e-FAST). Although e-FAST could demonstrate a secondary sign of injury in BTAI, notably large left haemothorax, or demonstrate pericardial effusion, the aorta itself usually is poorly visualized and can only be examined below the diaphragm. The same holds true for transthoracic ultrasound.
4.4 Transesophageal Echocardiography (TOE or TEE)
Already in 2008, Demetriades et al. found that between 1997 and 2007, there had been a near elimination of transoesophageal echocardiography for the diagnosis of BTAI, dropping from nearly 12 % of BTAI patients receiving TOE in 1997 to 1 % in 2007 (Demetriades et al. 2008). The primary use currently is to evaluate cardiac dysfunction and injury or hemodynamic state and response to treatment. TOE can be used to evaluate the descending aorta, being reasonably sensitive for traumatic aortic injury in that location but overall only moderately sensitive (Patel et al. 2003; Rippey and Royse 2009). In hemodynamically unstable patients, transoesophageal echocardiography can be used as bedside test, and the probe can remain in place even in the operating theatre.
4.5 Intravascular Ultrasound (IVUS)
In two papers by Williams et al. in the early 1990s, the use of IVUS is discussed as a tool to be used in equivocal angiographic results for BTAI (Williams et al. 1992; Williams et al. 1993). The limitation of this technique as an initial diagnostic test due to practical restrains was already discussed in their second paper and has not been altered since. Malhotra et al. as well as Patel et al. demonstrated better sensitivity compared to angiography especially for picking up MAI, before the era of CTA as a gold standard (Patel et al. 2003; Malhotra et al. 2001). A more recent paper again proved IVUS to be better than angiography in patients where CTA findings were equivocal. Therefore, the authors advocated the use of IVUS in potential TAI patients in whom angiography is being considered (Azizzadeh et al. 2011). However, the nature of the procedure requires vascular access, with another option to rule out aortic injury being a repeat CTA, with or without ECG triggering. If a patient is managed with endovascular stent-graft placement, IVUS has been shown to be useful for selection of stent-graft size (Wallace et al. 2015; Shi et al. 2015). Compared to the acute CTA, IVUS during the procedure of stent placement demonstrates a larger aortic diameter, possibly reflecting intravascular hypovolemia during resuscitation at initial imaging, necessitating preoperative reassessment of aortic lumen diameter (Wallace et al. 2015).
4.6 Magnetic Resonance Imaging (MRI)
Length of examination time, limited accessibility, non-compliant materials and relatively small bore sizes render MRI unfeasible in the initial assessment of patients with multiple injuries. In patients with equivocal CTA findings for BTAI, an MRI study can be considered if they are hemodynamically stable and also otherwise able to undergo the examination. Fattori et al. published two papers in the mid- to late 1990s on the use of MRI for the diagnosis of acute BTAI and for follow-up in the delay to treatment, demonstrating its feasibility (Fattori et al. 1996; Fattori et al. 1998). The paucity of results when performing literature searches to find the use of MRI in BTAI indicates the limited role for this modality especially in the acute phase, despite the well-documented use in other aortic and major vessel diseases.
4.7 Computed Tomography Angiography (CTA)
As stated before, CTA now is the gold standard for the evaluation of BTAI, given its high accuracy and near-perfect negative predictive value. In these usually severely injured polytrauma patients, the chest will oftentimes be imaged in conjunction with the abdomen and pelvis.
4.8 CT Technique
Different protocols exist to administer iodinated intravenous (IV) contrast material, with either single-bolus, split-bolus or triple-bolus administration being used. To avoid streak artefacts from high concentrations of iodine in the left brachiocephalic vein, IV contrast material should preferably be administered via the right arm whenever possible. Depending on scanner manufacturer and type, an empirically fixed delay or a bolus-triggering mode can be used. The administration of a separate timing bolus before the definitive scan is not commonly chosen in current practice, mostly due to time constrains. Furthermore, with current technical advances in multi-detector computed tomography (MDCT) resulting in high spatial resolution, vascular injuries will be detected even if acquisition phase is past proper arterial phase, which still is the preferred phase of acquisition (Raptis et al. 2015). In our opinion, the radiologist should be present at the acquisition to decide if image quality is satisfactory and to instantaneously order repeat scan or extra phases, especially for the abdomen and pelvis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree