Micropuncture introducer.
Angiocatheter.
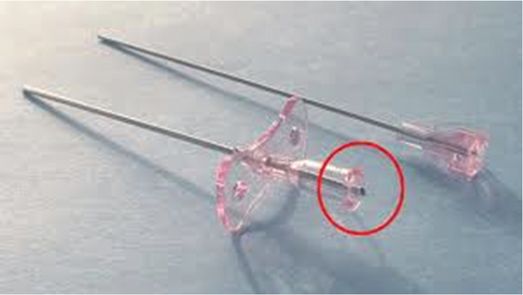
Potts–Cournand needle.
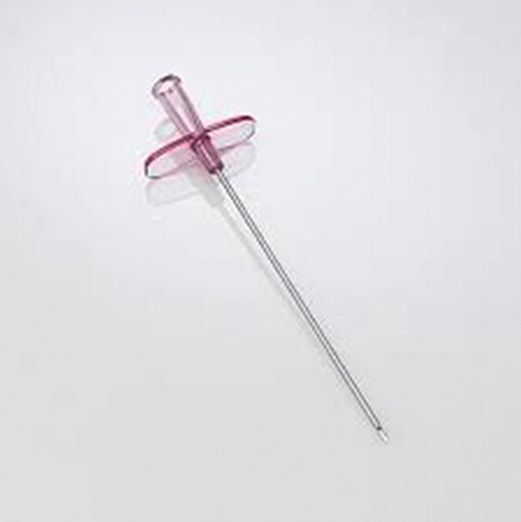
Single-wall access needle.
Angiographic catheters should be available in sizes ranging from 3 French (usually 40 to 65 cm), to 5 or 6 French (usually 90 cm or longer). It is our preference to use 3 French catheters for children less than 10 kg, 4 French catheters for children between 10 and 30 kg, and 5 French catheters for larger children. The length and diameter of the primary and secondary curves must be proportional to the child’s aorta. Therefore, a standard 4 French pre-shaped catheter may not be well suited for infants and young children in some instances. Dilute (140 to 240 mg iodine/ml) non-ionic low osmolarity contrast medium may be used with digital subtraction angiography and provides diagnostic quality images and conserves contrast, although higher concentrations may be necessary in infants and small children to compensate for the small volume of their vessels. Commonly used drugs include heparin, glucagon, dexamethasone, nitroglycerine, and papaverine (see Chapter 1). The equipment utilized to monitor children varies somewhat between interventional laboratories but should include at minimum continuous pulse oximetry and electrocardiography (EKG) and automated blood pressure cuff recorders, and preferably also intravascular pressure recording equipment and a sidestream (capnography) CO2 monitor. Standard wall suction and medical gas outlets or appropriate substitutes are mandatory.
Patient preparation
Each child must be prepared for either deep sedation or general anesthesia. The patient is kept NPO according to relevant institutional policies regarding sedation and anesthesia. Appropriate IV fluids may be given to maintain hydration, especially in small infants. Routine blood work is usually not necessary. However, preprocedural testing is tailored to the individual patient and situation. For example, if prolonged thrombolysis is planned, baseline studies including a coagulation profile and fibrin split products are obtained. Measurements of renal and hepatic function should be obtained where appropriate. Relevant diagnostic studies should be available for guidance in tailoring the diagnostic angiogram.
For patients less than 10 kg, appropriate warming devices should be prepared before the patient is placed on the angiographic table. Physiological monitoring (pulse oximetry, blood pressure, EKG) is instituted in all cases. Side-arm (for sedation) or in-line (for general anesthesia) capnography is recommended. Intravenous access is obtained in all children prior to beginning the procedure. Restraints are applied to patients not undergoing general anesthesia. Light restraints are generally used even in children having general anesthesia. Depending on the case, other preparation may include a Foley or small rubber urinary catheter for vascular interventions and other long procedures, and placement of lead shields over the gonads and other radiation sensitive areas whenever possible. For long cases, and for any case performed under deep sedation or anesthesia, padding of pressure points (e.g., egg crate foam, gel pads, diapers, or other cloth) to avoid neuropathy or other injury is essential, as described in Chapter 1.
Peripheral pulses (e.g., dorsalis pedis and posterior tibial) are marked on the skin so that they can be easily evaluated during or after completion of the procedure. We use a pulse Doppler or pulse oximeter to monitor lower extremity pulses during the procedure and while hemostasis is achieved after catheter and sheath removal (Figure 8.2). Standard groin preparation including shaving of pubic hair in teenagers is appropriate. In the case of femoral access both groins should be sterilely prepared and draped.
Standard technique
Arterial access
We find it helpful to place a small bolster (rolled-up towel or equivalent) under the hips to straighten the pelvic vessels somewhat by extending the pelvis, and to prevent the pelvis from moving posteriorly when the needle is advanced. In the past, initial arterial access was accomplished with a modified Seldinger technique using visible landmarks and palpation of arterial pulses. Although this technique is still widely practiced by interventional cardiologists, it is no longer recommended for routine access, especially in infants and small children, due to the added procedure time and the relatively high prevalence of arterial injuries such as intimal dissection, pseudoaneurysm, and arteriovenous fistula associated with this technique. Real-time access guidance with US is now the preferred standard of care. The general principles of angiography in children are summarized in Table 8.3.
Sedation or general anesthesia
Puncture artery and insert a sheath (infused at 3 ml/h or greater rate, adjusted to patient size)
Heparinize PRN (50 to 100 U/kg), may repeat every two hours during the procedure
Selective/superselective catheterization of target vessel(s)
Perform vascular intervention
Remove catheter and achieve hemostasis
Treat vasospasm or occlusion PRN
Follow-up vascular complications with US
The double-wall puncture technique can be safely performed, advancing the needle tip through both walls, withdrawing the sharp stylet, and pulling back the blunt cannula until pulsatile flow is obtained, then inserting a soft-tipped guide wire into the artery. However, real-time guidance with US permits a single-wall technique to be performed with great accuracy and a high rate of success, advancing a single-wall needle through the superficial wall of the artery until the bevel and needle tip are securely within the arterial lumen and pulsatile flow is obtained, then advancing a soft-tipped guide wire into the artery. This may be preferable especially when lytic therapy is planned to decrease the risk of bleeding or other arterial complication. Ultrasound guidance is usually performed with a transverse view of the artery, although a long view can sometimes be useful. The ability to visualize the entire length of the needle as well as the target vessel in the long view is balanced by the difficulty overcoming partial volume artifact to keep the needle centered on the vessel in real space, not just seeing both needle and vessel in the same image plane.
If access is gained by palpation, it may be difficult to use the standard Seldinger technique for arterial puncture, especially in small infants. Instead of palpating the pulse with two fingers so that dampening can be appreciated with the distal finger, the interventionalist may use a one-finger technique. The pulse Doppler (or oximeter) is substituted for the distal finger. Using downward pressure with the needle, the artery is compressed until the pulse is no longer audible. While maintaining this downward pressure and arterial compression, the needle is thrust forward into the arterial lumen.
For patients having procedures performed under sedation, the intended puncture site is slowly and carefully injected with local anesthetic (1% buffered lidocaine), with attention to avoiding the femoral artery or vein with the infiltrating needle. It is helpful to use the smallest needle available (27- or 30-gauge) since this will minimize discomfort. A small skin incision is made approximately 1 to 1.5 cm below the site where the common femoral artery crosses anterior to the femoral head, located either by US or fluoroscopy. This assures a stable platform beneath the puncture site at the time of hemostasis at the conclusion of the procedure.
In general, it is best to puncture at or below the inguinal ligament and above the origin of the profunda femoris artery. Puncture above this level risks retroperitoneal hemorrhage as well as penetration of the hip joint and may make hemostasis more difficult. Puncture below this level may risk end-arterial injury, necrosis, and compartment syndrome. When blood return is obtained and provided no resistance is encountered the guide wire is advanced into the abdominal aorta. The operator must be careful not to “crush” the femoral artery against the guide wire or catheter while performing exchanges. Catheters or sheaths should be advanced gently over the guide wire, with as little manipulation at the groin as possible. Any vascular procedures requiring more than one catheter exchange or associated with an interventional procedure should be performed through an arterial sheath to minimize vascular trauma and to ensure vascular access in the event that a catheter becomes occluded. Some operators prefer to use a sheath in all cases. These precautions will reduce the possibility of arterial injury, arterial spasm, or occlusion.
Once the sheath is in place, Steri-Strips® or Tegaderm® are applied to secure the sheath to the drape to prevent excessive movement or inadvertent removal. The sheath may be infused with a heparinized flush solution by a pump or pressure bag. The infusion rate and volume should be limited by the use of a check flow valve (3 ml per hour for small infants) or an infusion pump, in order to avoid fluid overload. In small children, flush aortograms can be performed through a straight or pigtail catheter with multiple side holes. Pigtail catheters may cause problems as the loop may be too wide to reform in the narrow aorta. Standard injection volumes and rates for different vessels are listed for children of various ages in Table 8.4. In general, the smallest catheter that will provide the necessary contrast injection rate is selected. Shorter catheters are used when possible since they are able to accept higher flow rates.
| Rate, per second (total volume ml) | |||
|---|---|---|---|
| Vessel | <10 kg | 10 to 30 kg | >30 kg |
| CCA | 4–5 (6–7) | 6–7 (8–9) | 8–9 (10–11) |
| ICA | 2–3 (4–5) | 4–5 (6–7) | 6–7 (8–9) |
| ECA | 2–3 (3–5) | 3–5 (5–7) | 5–7 (7–9) |
| vertebral | 1–2 (2–3) | 3–4 (4–5) | 5–6 (6–7) |
| aortic arch | 5–10 (10–20) | 10–15 (20–30) | 15–20 (30–40) |
| descending aorta | 5–10 (10–20) | 10–15 (20–30) | 15–20 (30–40) |
| visceral | 2–3 (4–6) | 3–4 (6–8) | 5–6 (10–12) |
| splenoportography | 2–3 (10–15) | 3–4 (15–20) | 5–6 (25–30) |
| renals | 2–3 (4–6) | 3–4 (6–8) | 5–6 (10–12) |
| prox extrem | 2–3 (4–6) | 4–5 (8–10) | 6–7 (12–14) |
| distal extrem | 1–2 (2–4) | 2–3 (4–6) | 4–5 (8–10) |
The routine use of heparin for diagnostic and interventional vascular procedures is controversial. In some laboratories, children less than 10 to 15 kg are routinely heparinized immediately after insertion of a catheter or arterial sheath (50 to 100 units/kg). Heparin boluses are repeated every two hours if the procedure is still under way.
In general, contrast volume is limited to 5 ml per kg (300 mg iodine per ml) for diagnostic angiography. Complex vascular interventions may require larger volumes. In our experience, larger volumes of non-ionic contrast (10 ml or occasionally greater) are well tolerated over long procedures, if the patient is kept well hydrated and urine output remains normal. However, fluid input and output need to be carefully monitored. At the end of the procedure, pedal pulses are checked manually or with a pulse Doppler or oximeter. Hemostasis is achieved with ten minutes of manual compression. If a pulse cannot be palpated, contrast injection is made through the catheter or sheath into the access artery. If the artery is completely occluded, 2 to 3 µg/kg of nitroglycerine, diluted in 3 to 5 ml of saline, can be injected through the catheter while it is being withdrawn. Alternatively, injection of a local anesthetic around the artery may help relieve vasospasm. On occasion the response is dramatic.
At the completion of the procedure, the pulse Doppler or oximeter may again be used to monitor hemostasis at the time arterial access is withdrawn. Our approach to hemostasis is to always use manual compression, and we do not use or recommend the use of arterial occlusion devices or of sandbags or other pressure devices in the pediatric population. Initially the vessel is compressed so that there is no antegrade blood flow (no audible pulse with Doppler over the dorsalis pedis or posterior tibial artery) for one minute. Then the compressive force is reduced so that blood flow is audible but there is no bleeding from the puncture site. This lighter compression is maintained for nine minutes. After ten minutes, manual arterial compression is discontinued unless there is bleeding. This approach is intended to minimize the chance for formation of an occlusive thrombus. If rebleeding or persistent bleeding occurs, arterial compression is re-established and progress checked at five-minute intervals. In children in whom hemostasis is difficult to achieve, a hemostatic agent (such as a Surgicel® pad or StatSeal® powder) is placed over the puncture site while the vessel is compressed. The hemostatic agent is then left in place and covered by a sterile 2 × 2 gauze and a transparent dressing.
During the recovery period, the lower extremities must be kept warm. Pedal color, temperature, and pulses should be monitored frequently. If the ipsilateral foot is cool to touch and if the pedal pulses cannot be manually palpated, then we recommend that the patient receive another bolus of heparin intravenously. If pulses do not return within three to four hours, a heparin infusion should be started and the patient kept in the hospital for treatment and observation. Color Doppler US is useful to document the status of the femoral artery. If the femoral artery remains occluded 24 hours after the procedure, systemic thrombolysis with an intravenous infusion of tPA should be considered.
Technical variations
Alternative access
Although most transarterial procedures are performed via the femoral artery, alternate vascular access should be considered in specific situations. Since the risk of femoral artery thrombosis is relatively high in neonates, the umbilical artery should be used for vascular access whenever possible (Figure 8.3). The main advantages of the umbilical artery approach include decreased significance of vascular occlusion since this is an artery that can be lost without concern, and the ability to use larger catheters or sheaths (4 to 5 French catheters or sheaths can be regularly utilized). The primary disadvantage of this route is difficult catheter maneuverability due to the tortuous vessel course. However, this problem can be overcome with the use of a sheath, directional guide wire, and coaxial catheter systems.
A newborn female presented with hypoxemia, congestive heart failure, and pulmonary hypertension. Initial US showed a large high-flow intrahepatic vascular abnormality.
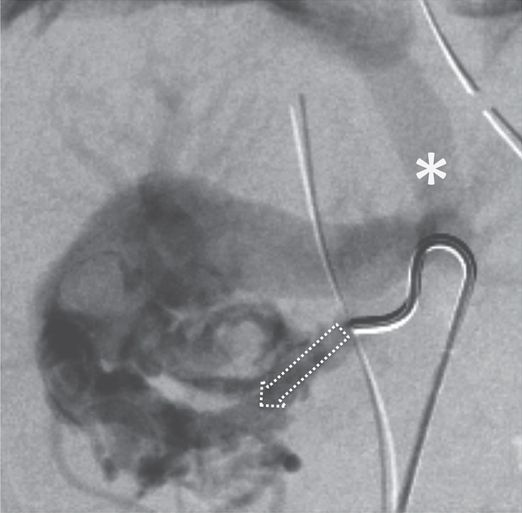
A 4 French “shepherd’s hook” angiographic catheter was inserted using preexisting umbilical arterial access, was formed in the aortic arch then pulled down into the replaced right hepatic artery through the SMA. Contrast injection showed hepatoportal fistulous connection (arrow) to a grossly enlarged right portal vein. The left portal vein drained through a patent ductus venosus (asterisk) to the vena cava.
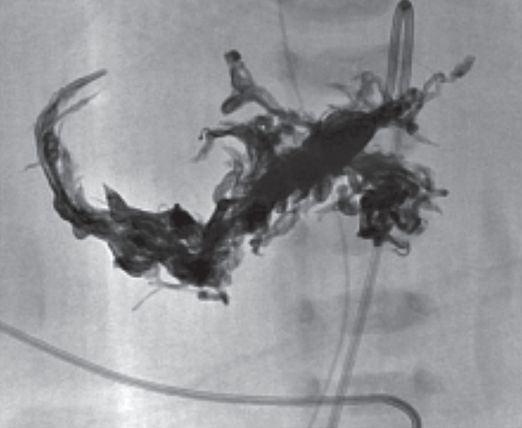
The numerous fistulae (more than 100) could not be embolized with coils or particles. Onyx® flowable liquid was used for preliminary embolization, and resulted in dramatic, albeit temporary, clinical improvement.
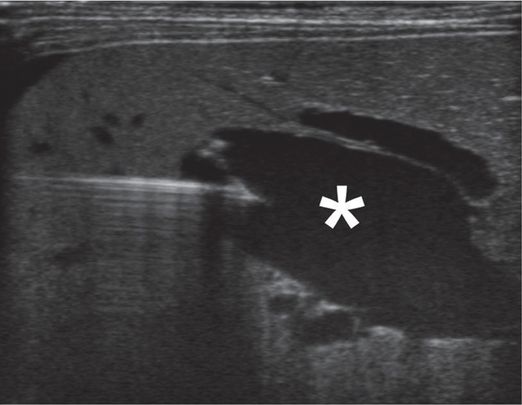
To enable embolization of the patent ductus venosus, percutaneous transhepatic access to the portal vein (*) was obtained at the bedside in the NICU under US guidance.
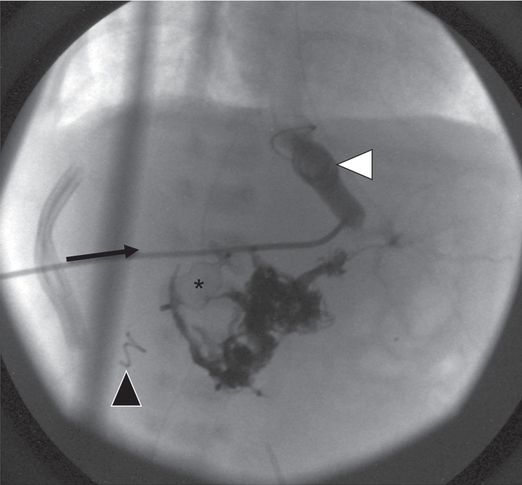
The transhepatic access was used to deliver a 5 mm × 5 cm coil that successfully occluded the ductus (white arrowhead). An intrahepatic tract coil (black arrowhead) is visible from prior transhepatic delivery of an Amplatzer® plug to control a large residual hepatoportal fistula. Despite successful control of the abnormal vascular shunts, the child eventually succumbed to intractable pulmonary hypertension.
If the neonate already has an umbilical artery line in place, it can usually be easily exchanged over a guide wire for a 3 to 5 French diagnostic or delivery catheter or a 4 French arterial sheath. At the end of the procedure, the softer umbilical artery catheter can be replaced, and thus access preserved for patient monitoring or a staged endovascular procedure.
A transvenous approach can be used in neonates, by passing a catheter across the foramen ovale into the left ventricle. However, this technique may be associated with cardiac arrhythmias and requires considerable experience with neonatal cardiac catheterization. A third alternative, most useful in infants with high-flow vascular lesions in the thorax or upper abdomen, is an axillary artery puncture. Since the distal abdominal aorta and the femoral arteries are reduced in size in children with high-flow visceral lesions due to the large run-off at the level of the lesion (Figure 8.4), the axillary approach is technically easier, facilitates selective catheterization of the abnormal vessel(s), and is generally well tolerated. The main risk of this approach is the risk of brachial plexus neuropathy. For select procedures in adolescents and young adults, for example when femoral access is contraindicated, brachial access in the antecubital fossa may be helpful. It may also be useful in certain cases, such as treatment of vascular malformations, to acquire direct percutaneous access of target arteries (Figure 8.5). Finally, a translumbar approach was at one time the primary method for aortic access for angiography, although it is very seldom used in modern practice.
A newborn male presented with congestive heart failure and a hepatic mass.
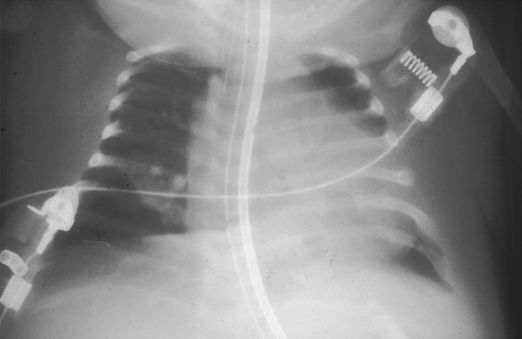
Chest radiograph demonstrating cardiomegaly and mild congestive heart failure.
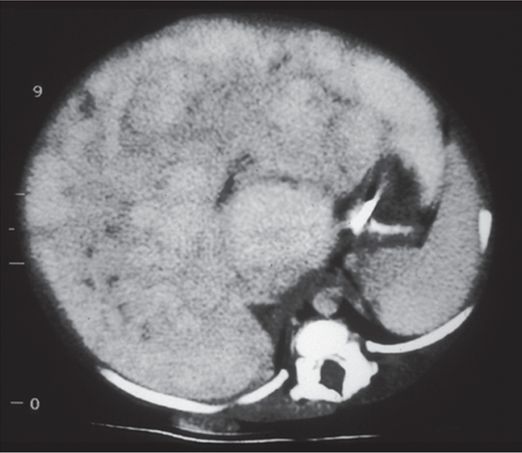
Axial contrast-enhanced abdominal CT shows hepatomegaly with marked nodular enhancement.
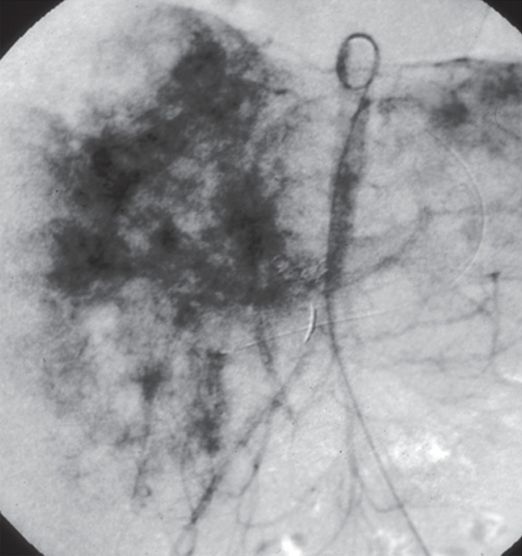
Abdominal aortogram showing marked reduction in distal aortic and common iliac diameter secondary to run-off at the celiac axis.
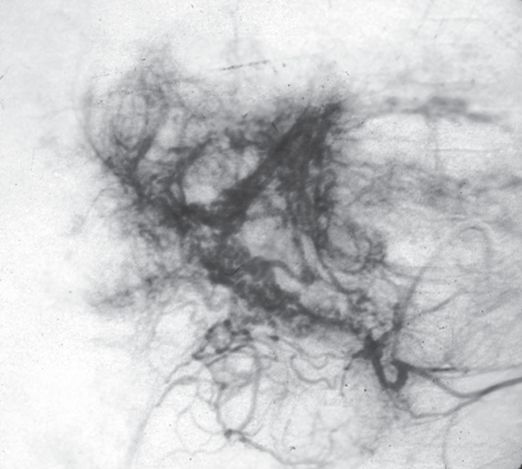
After embolization there is marked irregular contrast enhancement of the liver.
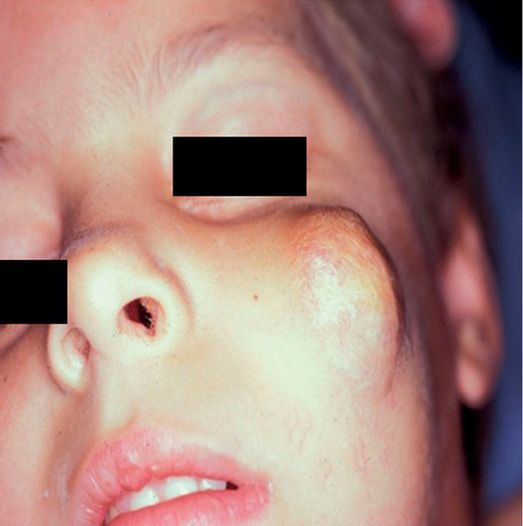
A 13-year-old male was referred by a maxillofacial surgeon for evaluation and treatment of an extensive mandibular arteriovenous malformation.
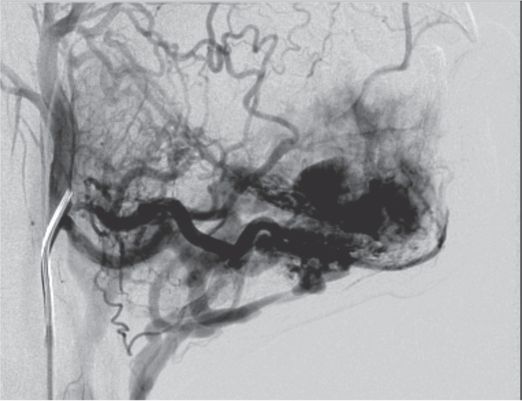
Arteriography from the right facial artery catheter demonstrates extensive intraosseous and soft tissue involvement of the AVM. However, due to surgical ligation of facial and lingual vessels in childhood, transfemoral access for more distal embolization was not possible.
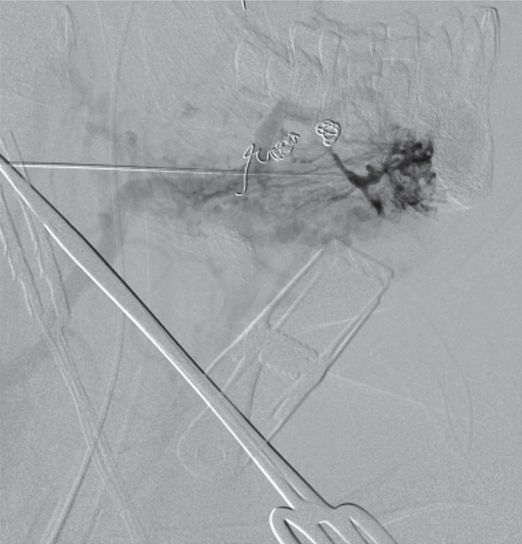
Direct percutaneous US-guided puncture of a distal facial branch was obtained. Dark staining at the tip of the 21-gauge Chiba needle (stabilized by a clamp in this image) represents partial Onyx® embolization of the nidus, with contrast seen flowing through residual arteriovenous connections.
Postprocedure and follow-up care
Postprocedure orders include standard evaluation of the vascular access site and appropriate pulses, intravenous fluids, analgesics, and functional monitoring (e.g., neurologic status following cerebral or spinal angiography). A typical approach is to evaluate vital signs and the puncture site every 15 minutes for 30 minutes, every 30 minutes for the next hour, then every hour until the child is alert or back to neurologic baseline. Standard postprocedure recovery time for outpatient angiography is four to six hours. Whenever possible diagnostic examinations and, in certain instances, vascular interventions are performed in the outpatient setting.
Technical problems and pitfalls
The arteries and veins of infants and young children are much more reactive than those of older children and adults and are prone to vasospasm. Unintentional puncture of the femoral artery during infiltration of local anesthetic, or puncture of the femoral artery with inability to pass the guide wire, may produce spasm resulting in a non-palpable pulse. In this situation, the operator should either wait until the pulse returns to normal, which may take 30 minutes or more, or attempt to puncture the opposite femoral artery. Perivascular injection of 1% lidocaine can sometimes hasten the return of antegrade blood flow. The subintimal passage of a guide wire or catheter can easily occur with little resistance in young children. Therefore, it is important to be gentle and avoid probing with a guide wire. In children less than 10 kg, excessive catheter flushing can be dangerous and result in fluid overload and anemia. To avoid this issue, we recommend substitution of smaller volume syringes (e.g., 3 to 5 ml) instead of the larger syringes (e.g., 10 ml) commonly used for flushing and contrast injection. Also, maintaining and communicating a running tabulation of all fluids used is helpful. Failure to immobilize the legs and keep them warm after an arterial procedure may result in a further decrease in blood flow to the affected extremity and a hematoma. The use of oversized catheters in small arteries contributes to intimal injury, occlusion or embolization of thrombus and should be avoided.
Complications
Femoral arterial thrombosis is the most common complication following a transfemoral arterial vascular procedure in infants and young children. Factors that increase the likelihood of arterial thrombosis include small size of the patient (less than 10 kg), a hypercoaguable state or possibly the failure to heparinize, the use of large catheters, traumatic passage causing intimal injury (e.g., unsheathed balloon catheters), excessive catheter manipulation, hypothermia, and low cardiac output. The ability to detect pedal pulses by the use of Doppler monitoring does not exclude the presence of femoral artery thrombosis, as collateral pathways open quickly between the internal iliac and superficial femoral arteries to perfuse the distal extremity. In spite of the presence of collateral supply, which will usually prevent serious tissue loss, femoral artery thrombosis in infants and young children leads to leg length discrepancies in a significant number of patients. In addition, some of these children may develop claudication later in life, requiring a surgical bypass. Several strategies may be used to minimize the risk of occlusion including the use of anticoagulation (all children <10 kg), the use of a catheter size and shape appropriate for the task (the smallest catheter that will accomplish the objective is usually preferred), the use of sheaths when possible to protect the access artery, and by avoiding excessive catheter manipulation. When femoral artery thrombosis is diagnosed, patients should be treated, in the absence of contraindications, initially with full heparinization for 24 hours and then systemic thrombolysis (tPA) if needed.
Thromboembolic complications can occur in any arterial circulation but are most significant when they involve the CNS. These problems may result from wedging oversized catheters in small or spastic arteries, or from improper catheter flushing. Vessel occlusion can also be the result of subintimal dissection with a catheter or guide wire.
Pressure sores can result from long procedures and can be prevented by the use of padding at pressure points and removal of the hip bolster after femoral artery puncture. This is especially problematic in the areas of the shoulders, coccyx, iliac crest, elbows, and occiput. Padding in the axilla and careful arm, shoulder, and neck positioning will help prevent brachial plexopathy. Special attention should be paid to the position of the elbows because of the additional risk of ulnar nerve injury, and the occiput where pressure can result in hair loss. We have found the use of egg-crate foam and gel rings or pads to be effective. It is important to avoid the use of padding that is substantially radio-dense near areas likely to be included in the imaging field, since the automated detector may compensate with a significant increase in radiation dose.
Fluid overload and anemia can result from excessive catheter flushing and may require admission to the intensive care unit for diuresis or transfusion.
Hypothermia may result in metabolic acidosis with generalized deterioration of the patient’s condition and, on rare occasion, cardiac arrest. It is important to be careful when performing procedures on neonates since they are prone to hypothermia. This complication may be prevented by carefully protecting the patient with a variety of actions including warming the contrast (to approximately 27 to 32°C), covering or wrapping the infant’s body with a blanket or plastic wrap, covering the head with a hat or plastic wrap since the head is the greatest source of heat loss, or using a water blanket, heater, or other method to minimize heat loss and keep the child dry.
Hepatic arteriography
Introduction
Before the introduction of US, CT, and MRI, hepatic angiography, along with the radionuclide liver–spleen scan, played a major role in the evaluation of liver masses and other types of hepatic pathology. Currently, the initial work-up of liver masses is performed with cross-sectional imaging that is safer, more sensitive, more accessible, and more cost effective than angiography. Magnetic resonance imaging with MR angiography (MRA) is usually selected for initial evaluation. If additional information is necessary US is often used. However, there are several circumstances in which angiography still plays a significant role. In the setting of a transplant center, the majority of hepatic angiography is performed on patients who will undergo or who have undergone transplantation. A smaller number of patients are referred from the neonatology, gastroenterology, oncology, and surgery services.
Indications
A common indication for hepatic angiography is in the preoperative evaluation of a patient with end-stage liver disease prior to liver transplantation. In order to evaluate whether or not a patient is a viable candidate for liver transplantation the size, patency, and anatomy of the hepatic artery, IVC, portal vein, and mesenteric veins are assessed. Initially, these vessels are evaluated with color Doppler US. If they are not well visualized because of overlying bowel gas, if the arterial or venous anatomy is distorted because of previous surgery, if the portal vein is less than 4 mm, or if there are internal echoes in the portal vein suggestive of thrombus, then angiography is usually performed. Biliary atresia is one of the major indications for liver transplantation in the pediatric age group, and it is associated with a higher frequency of vascular and visceral anomalies, including congenital absence of the intrahepatic IVC, aberrant origin of the hepatic artery, pre-duodenal portal vein, situs inversus, polysplenia, and malrotation. The other major indication for angiography in the pretransplantation evaluation is suspected congenital absence of the IVC or other congenital vascular abnormalities.
Hepatic angiography is also considered if a vascular intervention is contemplated, for example, evaluation of a neonate with a hypervascular liver mass and congestive heart failure. These masses are usually hemangioendotheliomas or cavernous hemangiomas. Cavernous hemangioma is the most common benign hepatic neoplasm in the pediatric age group. Most hemangiomas are solitary lesions and are discovered as an incidental finding in imaging studies being done for other reasons (see Figure 4.58). Children with symptomatic hepatic vascular lesions usually have large lesions and present in the prenatal period or infancy with congestive heart failure and a hepatic mass on physical or imaging examination.
Hemangioendotheliomas occur exclusively in neonates and very young infants and are more likely to be symptomatic than hemangiomas, usually presenting before six months of age. The most frequent symptoms at presentation are hepatomegaly and congestive heart failure. Hemangioendotheliomas tend to grow rapidly after initial presentation, and usually undergo slow, spontaneous regression over time. However, infants with these neoplasms are at significant risk of life-threatening congestive heart failure, ischemic damage to the heart and kidneys, portal hypertension, Kasabach–Merritt syndrome (thrombocytopenia with DIC), and tumor rupture. If the child is symptomatic the diagnosis is made with cross-sectional imaging and a plan is formulated. Cross-sectional imaging, especially MR and US, is best for discerning the extent and distribution of the lesion and is far less stressful on a sick neonate or young infant than is angiography. Angiography should be reserved for children who require vascular intervention. It is variable and somewhat controversial as to the best time to intervene in children with hemangioendotheliomas. It is our opinion that children without CHF or other life-threatening symptoms should be treated medically whenever possible. Today, propranalol has become the drug of choice in our practice since its time to action is short. Steroids and interferon remain other drug options. However, those children who fail medical therapy, or who are in CHF, or who have coagulopathy should be considered for prompt referral for angiography and embolization of the hepatic artery. Treatment of the hyperdynamic state should be accomplished before irreversible myocardial or renal injury occurs. We feel strongly that it is best to err on the side of early intervention since delayed treatment may result in a fatal outcome.
In general, there are no absolute contraindications to hepatic angiography, with the possible exception of previously documented anaphylactic reaction to intravenous contrast. Relative contraindications to angiography include an uncorrectable coagulopathy, severe anemia, hypertension, and acute or chronic renal failure.
Technique
Equipment and patient preparation are identical to that discussed above for general angiography.
Standard technique
The general elements of angiography are discussed above. For aortic injections a standard pigtail catheter with multiple side holes is used. For selective cannulation of the mesenteric vessels catheter selection is much more dependent on the preference of the operator. We most often use C-1, JB-1, Cobra, or RIM catheters. Be aware that a Simmons-type catheter may be difficult to use since the aortic diameter may be too small to allow the catheter to form. We do not routinely find this shape to be helpful in smaller pediatric patients. Once access to the desired artery is achieved, more distal access often requires the coaxial insertion of a microcatheter.
Standard injection rates and volumes for arteriography are given in Table 8.4. The exception to these suggested contrast volumes and flow rates is an SMA portogram where the injection is usually five to seven seconds with a volume of 25 to 35 ml after injection of intra-arterial papaverine or priscoline. The splenic artery may also be injected for portal venography. In children over 20 kg, we inject 45 mg of papaverine over two to three minutes prior to contrast injection. In patients less than 20 kg the dose is decreased to 22.5 mg or less, depending on the patient’s weight. If the injection of vasodilator needs to be repeated, a minimum of ten minutes should pass between doses. Systemic blood pressure should be monitored. If hypotension occurs, it is easily corrected with IV infusion of normal saline in most cases.
To visualize the mesenteric arteries in neonates and young infants we film at rates of five frames per second (fps) or higher, if possible, in single plane, or 3.0 fps biplane. In larger patients we usually film at 3.0 fps single- or biplane. For studies of the portal vein we use single plane and film at 3.0 fps for the entire time in small infants and in older children at 3.0 fps for three seconds and 1 fps until the portal vein is opacified. This means that in older children the filming may last 15 to 25 seconds or longer. Filming rates in selective runs depend mainly on flow rate and can vary from 1 to 5 fps.
Technical problems and pitfalls
There are a few factors that can present problems in the interpretation of an arterial (indirect) portogram (Figures 8.37, 8.43). Splenic or superior mesenteric vein occlusion or thrombus can be simulated when there is inflow of unopacified blood from the other large tributary of the portal vein, e.g., inflow from the splenic vein during superior mesenteric artery injection and vice versa. Ultrasound or other cross-sectional imaging, evaluation of the venous phase via injection of the other artery, splenoportography, or transhepatic (direct) portography may be needed to clarify the findings. When there is significant splenomegaly there can be poor or absent opacification of the splenic vein, suggesting splenic vein occlusion. In this case using a larger volume of contrast during splenic artery injection, splenoportography, or transhepatic portography will allow assessment of splenic vein patency. Poor visualization of the entire system may occur when there is hepatofugal flow or if there is significant spontaneous portal-systemic shunting. Finally there may be incomplete demonstration of gastro-esophageal varices if the splenic, superior mesenteric, and left gastric arteries are not all injected. Cases that require a higher total volume of contrast can be quite problematic in pediatric patients where total allowable contrast volumes are dictated by body weight or in those patients who have preexisting conditions requiring fluid restriction, such as congestive heart failure or renal disease.
Arterial embolization
Introduction
Catheter-directed occlusion of one or more vessels (embolization) is indicated for the treatment of a wide range of pediatric conditions. Experience obtained has shown that embolotherapy can be performed safely and effectively in children of all ages and sizes. The successful outcome of catheter-directed therapy has been aided by the significant improvements in equipment and materials. The development of the variable stiffness microcatheter, coils in sizes from 0.018-inch to 0.038-inch, particulate materials ranging in size from 50 to 1,500 microns (e.g., PVA, Embospheres®, Gelfoam® powder), glue, Onyx® (Figure 8.3), and devices such as vascular plugs and locoregional ablation techniques has enabled a greater variety of vascular territories and vessel diameters to be safely catheterized and treated.
Indications
Embolotherapy may be considered for either the primary treatment of a vascular lesion or as an adjunct to another form of therapy, usually surgery (Table 8.5). The goal of the vascular intervention and the nature of the vascular supply will usually influence the materials selected, technical approach, and endpoint of therapy. There are five principal goals of therapy: control of hemorrhage, preservation of threatened vital structures, protection of organ function, alleviation of adverse effects, and cosmesis. Hemorrhage may be directly related to the lesion, such as in gastrointestinal bleeding, trauma, or rupture of an arteriovenous malformation. It may be iatrogenic, such as dehiscence of a vascular anastomosis or following tonsillectomy (Figure 8.6). It may be due to dysregulation, such as the consumptive coagulopathy of Kasabach–Merritt syndrome (Figure 8.7). Alternatively, bleeding may be anticipated, such as prior to resection of a highly vascular tumor, e.g., juvenile angiofibroma. Vital structures may be threatened by compression or obstruction, including airway, vessels, nerves, and the auditory canal. Organ and tissue function may be threatened by alterations in physiology, such as cardiac failure from high-flow peripheral arteriovenous malformations, as well as by compression or occlusion, such as obstruction of vision by tumor mass or compression of the globe by a retrobulbar hemangioma. Adverse events may include pain as well as bony or soft tissue overgrowth. Cosmesis for a child is in no way a trivial issue. Even small lesions in the central face can be disfiguring and result in serious psychological and social issues. Procedures undertaken for cosmesis should include a thorough understanding of the expectations of the patient and clear communication of the limitations of the operator and intended technique. Successful therapy does not necessarily require obliteration of the lesion or malformation. In many cases, this is not practically achievable, and efforts to reach such an outcome may result in severe complications. For example, if the goal of therapy is pain management, then the procedure is complete when pain is controlled, understanding that the pain may return and that further intervention may be required in the future.
Preoperative to limit blood loss
Trauma
Gastrointestinal bleeding
AVMs and AVFs
Complicated high-flow hemangiomas
Trap aneurysms when other therapies fail or are too risky
Iatrogenic injuries, e.g., postbiopsy bleeding
A ten-year-old female presented emergently following cardiopulmonary arrest related to massive pharyngeal hemorrhage three weeks after tonsillectomy. A suture was noted in the right tonsillar fossa associated with voluminous bleeding. Right external carotid angiography showed a defect in the proximal right facial artery.
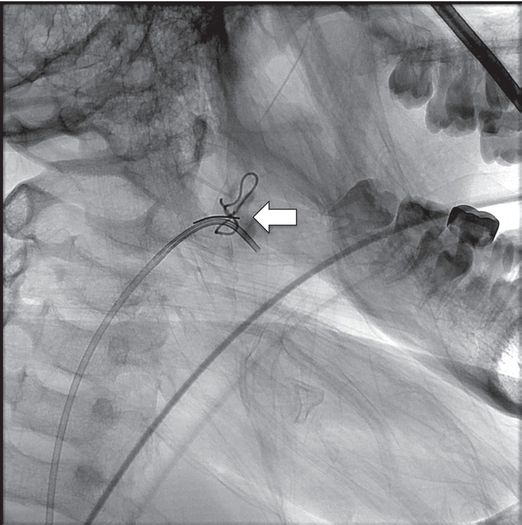
Visible (arrow) on selective injection following initial embolization with two Hilal coils, immediately resulting in stabilization of hypotension.
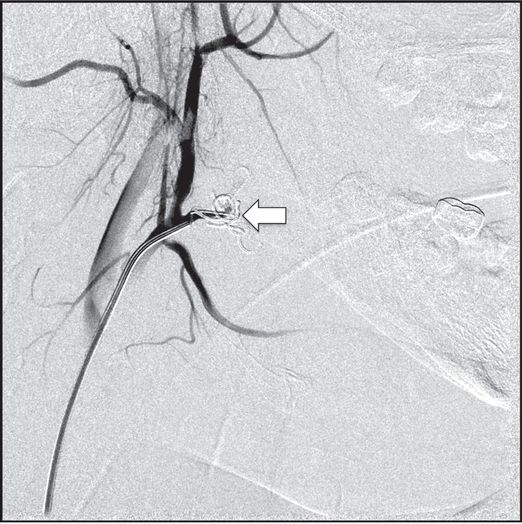
After further embolization with a 5 mm × 3 cm tornado coil and a 5 mm × 5 cm coil through a JB-1 catheter positioned in the facial artery across the defect, complete occlusion was achieved, and the child recovered with minimal neurologic sequelae.
Selective catheterization of a branch of the lateral thoracic artery in a 4.8 kg, 86-day-old female with Kaposi’s hemangioendothelioma and Kasabach–Merritt syndrome.
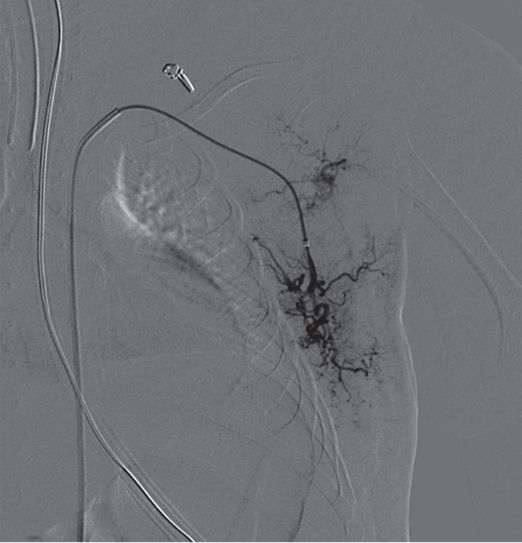
Microcatheter in lateral thoracic branch vessel supplying tumor.
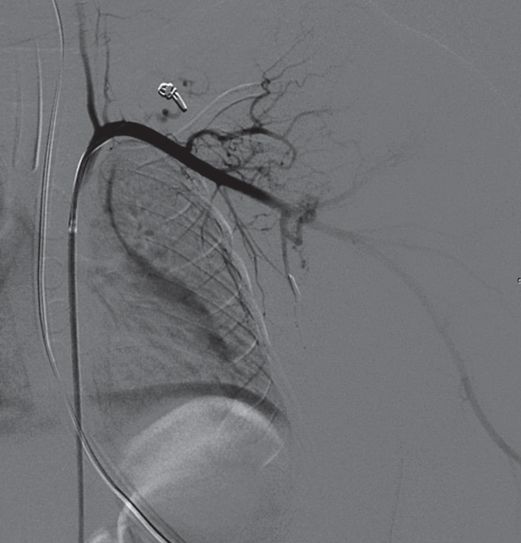
After embolization using microcoils, the angiogram shows marked reduction in blood supply and tumor vascularity.
Currently, the majority of pediatric embolization procedures are performed for the treatment of hemangiomas and high-flow vascular malformations. Today, medical therapy using propranolol has replaced surgery and embolotherapy as the initial therapy for infantile hemangiomas. In addition, there are a wide variety of other indications for embolotherapy including preoperative or primary treatment of hypervascular tumors (e.g., nasopharyngeal angiofibroma), primary or adjunctive treatment of an aneurysmal bone cyst, occlusion of a varicocele, treatment of acute or chronic bleeding from a variety of causes (e.g., hemoptysis in patients with cystic fibrosis, bleeding from an arteriovenous malformation (AVM)), and ablation of splenic or renal tissue in the treatment of hypersplenism and hypertension.
High-flow AVMs and arteriovenous fistulae (AVFs) occasionally present in the neonatal period with severe congestive heart failure. The most common of these involves the central nervous system and can take the form of galenic, pial, or dural types as well as liver involvement (hemangioendothelioma). Less commonly, diffuse AVMs of the extremity or isolated AVFs involving systemic or cardiac vessels may present with neonatal congestive heart failure. Urgent embolization, when successful, results in dramatic reversal of the cardiac failure and survival of the child. Delay in treatment often results in worsening of the congestive heart failure due to progressive myocardial ischemia and injury to other organs, e.g., kidneys and brain.
Anomalous systemic arterial supply to the lungs may be seen in isolation, in association with scimitar syndrome or pulmonary sequestration, and in patients with pulmonary atresia and ventricular septal defects. These malformations may result in severe congestive heart failure or hemoptysis. Prior to the embolization of systemic arteries, careful angiographic mapping of the pulmonary circulation should be performed.
Endangering hemangiomas (e.g., those associated with consumption coagulopathy in Kasabach–Merritt syndrome, bleeding, visual obstruction, ulceration with significant bleeding, and congestive heart failure) are generally initially treated by aggressive pharmacotherapy. Embolization may be useful in those patients failing medical management or in children who are at risk for or who present with a complication. Unfortunately, positive results are often transient and additional procedures are needed, especially in lesions with multiple arterial feeders (Figures 8.3, 8.5).
The indications for embolization of peripheral AVMs include cardiac failure, ischemic skin ulceration, bleeding, and pain. Most of these lesions are not curable by embolization alone and treatment planning should be carried out in conjunction with the appropriate surgical specialty.
Hypersplenism is an uncommon problem in childhood although a variety of etiologies may be responsible. Depending on the geographic location, the most common causes may vary. Portal hypertension, cystic fibrosis, thalassemia, and idiopathic thrombocytopenia purpura are among the more common etiologies. Total splenic embolization (TSE) is rarely indicated since it is associated with a high incidence of postprocedural abscess formation and severe postembolization syndrome. In general, TSE is only performed prior to surgical splenectomy. Instead, partial splenic embolization (PSE) is preferred to treat children with persistent severe thrombocytopenia, severe pain, or respiratory difficulty. Regardless of the etiology, the goal of PSE is to ablate 30 to 60% of the splenic parenchyma. This may be accomplished either by (1) selectively entering splenic branch arteries and systematically occluding them, or (2) positioning the catheter distal to the short gastric arteries but proximal to the bifurcation of the main splenic artery to perform particulate embolization, which we prefer. Proximal splenic artery catheter position (just distal to the short gastric arteries) allows for the injection of particulates (>300 microns) so that they will occlude the splenic parenchyma in all areas of the spleen, especially at the periphery. This has the advantage of symmetrically reducing spleen size and minimizing the time necessary to complete the procedure. Regardless of the approach selected, children will invariably develop postembolization syndrome, which consists of low grade fever (usually less than 40°C), pain, and elevated WBC count with left shift, as well as elevation of the platelet count in the first few days after treatment.
Hypertension secondary to segmental renal artery stenosis and secondary renal ischemia is a rare indication for embolotherapy. In these stenotic segmental renal artery branches not amenable to balloon angioplasty, embolization is a reasonable option and may cure or help control the child’s hypertension.
Technique
Equipment
It is of paramount importance to preserve the patency of the access artery after completion of the procedure. Therefore available equipment should include the smallest possible arterial sheath, catheter, and guide wire to accomplish the procedural objective (Table 8.6). The temptation of using larger systems for easier delivery of embolic materials should be avoided since they likely expose children, especially small infants, to a higher risk of complications and are not necessary for successful occlusion of vascular lesions.
Arterial sheath 4 to 7 French for most indications
Directional catheters (JB-1, Harwood-Nash, Cobra, C-1, etc.)
Variable stiffness microcatheters (fit through a 4 French catheter with a 0.035-inch lumen)
Guide wires (0.018- to 0.038-inch); choice depends on the vascular territory treated and operator preference
Bentson, hydophilic (angled Glidewire®), Newton, etc.
directional guide wires for microcatheters (≤0.018-inch)
Pressure bag or infusion pump for sheath perfusion
Embolic material
temporary agents
particles: Gelfoam®, autologous clot, Avitene® (may act as a permanent agent in some situations)
permanent agents
particles: PVA, Embospheres®, Gelfoam® powder
liquid: absolute alcohol, thrombin
tissue adhesives: glue and Onyx®
detachable balloons (not readily available)
Coils: Gianturco, platinum microcoils, Hilal, GDC, or other detachable coils, etc.
Embolization should be carried out by physicians with appropriate training and with access to pediatric anesthesia, nursing, and technical staff. High-resolution digital subtraction angiographic equipment, especially with road mapping capabilities, is essential. A selection of arterial sheaths (3 French and larger), thin-walled 3 to 5 French delivery catheters, variable stiffness microcatheters, and guide wires in short and long lengths are needed. Embolic materials include particulates, calibrated from 50 to 1,000 microns, Gelfoam®, absolute ethanol, tissue adhesives (glue and Onyx®), detachable balloons, devices such as vascular plugs, ablation equipment (e.g., laser, radiofrequency, microwave, and cryoablation) and platinum/fiber and stainless steel wire/fiber coils. Unfortunately, not all of these materials are readily available and approved for use in children. Coils and particles are currently the embolic materials most often used when permanent occlusion is desired.
Patient preparation
Evaluation of a child’s cardiovascular status is important in high-flow lesions. Infants in heart failure often require earlier treatment and have an increased anesthesia risk. We generally consider this an emergent indication for therapy. Assessment of the neurologic status of a patient with a facial lesion is important to exclude an existing cranial nerve abnormality. This may avoid the possibility of the finding being considered a treatment complication. Physical exam and documentation of lesion extent help to plan the likely number of treatments, especially when sclerotherapy is considered. Clinical evaluation and explanation of the treatment to the patient and parents also establishes the necessary confidence that a family must have in the treating interventionalist. Photographing lesions is important to document the baseline and subsequent improvement or need for further treatment. Routine laboratory tests are not performed unless specifically indicated. In general, children are not typed and cross-matched for either blood or platelets.
Prior to the procedure, the child should be well hydrated and have a working IV line. Corticosteroids may be given at the beginning of the procedure or 24 hours prior to the embolization, depending on the preference of the interventionalist. In most instances, it is best to prepare the child and family for a lengthy procedure. General anesthesia is usually advantageous and preferred for these procedures, since it can insure patient cooperation especially during a technically demanding, lengthy, and risky procedure. In addition respiratory and other physiologic factors may be controlled, when necessary. A urinary catheter is usually inserted because a large volume of contrast may be used. With the catheter in place, the urinary output can be monitored and bladder decompression assured. In most cases, the urinary catheter is removed before the child awakens. Systemic heparinization is suggested for infants under 15 kg, procedures involving the use of coaxial catheter systems, and procedures of the head and neck. Preoperative hematocrit, coagulation studies, and other laboratory tests are obtained on an as-needed basis.
Standard technique
In general, embolization is performed during the same session as diagnostic angiography. The angiographic approach is planned out according to the vascular territory of the lesion to be treated. Both non-invasive imaging and pre-embolization diagnostic angiography are performed to confirm the diagnosis and to finalize the interventional planning. Embolization is performed only after selective or superselective catheterization of the vessel(s) supplying the lesion is achieved. The precise technique utilized depends on the goal of the procedure, the nature of the lesion, and the embolic device/material being used (Table 8.7). When necessary, hypercarbia (via hypoventilation) may be helpful by producing vasodilation, thereby facilitating distal vessel catheterization.
Preoperative testing as needed
Hydration of the child
Puncture femoral artery and insert sheath
Infuse sheath with heparinized saline
Systemic heparinization (50 to 100 units/kg PRN), except children <10 kg who are routinely heparinized (50 to 75 units/kg)
Diagnostic angiography
Selective catheterization of pathologic vessels
Embolize vascular pathology to complete occlusion
Postembolization angiography
Before deploying an embolic agent, safe catheter tip position within the abnormal vessel must be confirmed. The material utilized for vessel occlusion depends on the interventionalist’s preference and experience, the type of lesion being treated, the goal of the procedure, and the availability of various materials. Particulates and coils are the most frequently used embolic agents. PVA, especially small particles (50 to 250 microns), are preferred since this material has the best chance of occluding the nidus of the lesion. In our practice, Embospheres® have replaced PVA as the particulates of choice. Embospheres® are available in a wide variety of particle sizes that generally meet the needs of the interventionalist. Recently, with the development of the liquid agent Onyx®, a third choice has become available, although it is quite costly. Coils are more often chosen for preoperative occlusions or when fluid volume excess is problematic (e.g., neonates with congestive failure secondary to arteriovenous shunting from a hemangioendothelioma). In general, coils are avoided in the proximal portions of vessels, since this approach often leads to the development of collaterals. When particles are used, we prefer to use a separate Mayo stand for any equipment containing particles. We mix the particles with contrast (1:1 ratio) via a three-way stopcock and two 6 to 10 ml syringes. One of the 6 to 10 ml syringes then acts as a reservoir for the particles while a 1 ml syringe is attached to the stopcock and eventually used for embolization (Figure 8.8). The particulate/contrast mixture is injected while observing with real-time digital subtraction fluoroscopy until blood flow becomes slow. The solution is pulsed in small boluses so that one can observe the speed of the forward flow and assess the progress of the embolization. One must be careful when flushing a catheter that has been contaminated with an embolic material since there may be residual material in the catheter. When coils or other devices are required, it may be useful to use flow-control techniques to assist in placing these devices in high-flow lesions. This is accomplished with proximal arterial or distal venous balloon occlusion, a tourniquet or blood pressure cuff, or glue/Onyx® control of appropriate outflow vessels.
During preparation for particulate embolization, all embolic materials and related equipment are maintained on a separate sterile tray to prevent contamination of other equipment and subsequent inadvertent injection. Embolic particles are mixed using two syringes: a larger reservoir syringe and a smaller injection syringe.
Particles (less than 100 microns, e.g., Gelfoam® powder), tissue adhesive, Onyx®, and ethanol are extremely aggressive agents and may cause tissue necrosis when delivered to a capillary bed or end arterial circulation. In addition, these materials can pass through small collateral connections resulting in downstream embolization and untoward effects. Because of their small size, these agents have several advantages including being able to flow into the nidus of a lesion, thus increasing the chance of a cure, and may be injected through microcatheters.
Preoperative embolization of tumors
The goal of a preoperative embolization is to devascularize a lesion so that it can be excised or (in the case of bone tumors) curetted out with minimal bleeding (Figure 8.9). Embolization should be performed with the goal of occluding the vessels supplying the tumor as distally as possible while sparing normal adjacent tissue. The safest and most effective technique is selective catheterization and occlusion of the vessels feeding the lesion (Figure 8.10). The embolic agent utilized is based on the lesion type and the experience and preference of the interventionalist. In preoperative cases, “perfect is the enemy of good”. Thus it is not necessary to try to completely devascularize the lesion but rather to minimize operative blood loss. It is often a good strategy to use large particles, coils, or other agents that will be the easiest and fastest to deploy. Before beginning the embolization procedure, cross-sectional imaging should be reviewed to determine the location of the mass. The vascular territory is predicted by the anatomic location of the tumor. The region of abnormality is studied angiographically to confirm and map the vascular supply, and exclude dangerous anastomotic communications with adjacent arterial territories. Once the pathologic anatomy is understood, each vascular trunk supplying the mass is selectively occluded. In general, the sequence of catheterization and embolization proceeds from distal to proximal vessels, to avoid the need to enter vessels that have already been treated and may be in spasm. For cases in which embolization is the primary therapy, it may be appropriate to utilize more aggressive embolic agents such as ethanol, small particles, or Onyx®, in an attempt to cure the patient.
Twelve-year-old boy with epistaxis.
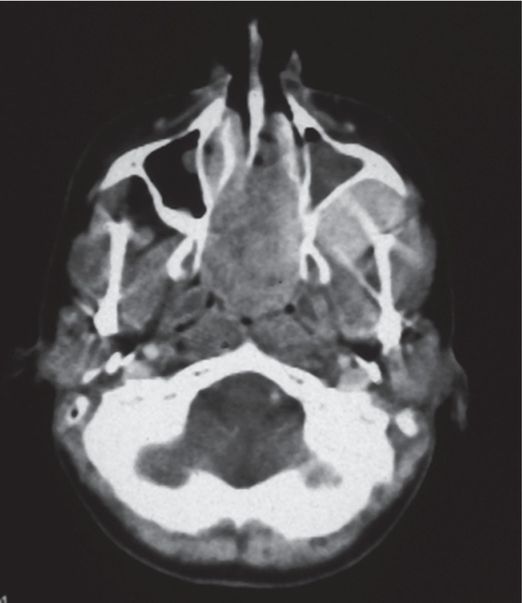
Axial CT scan demonstrates an enhancing mass originating in the left pterygo-maxillary foramen and extending into the nasal fossa and behind the maxillary sinus. Note there is widening of the pterygo-maxillary foramina with anterior bowing of the posterior sinus wall (Holman–Miller sign).
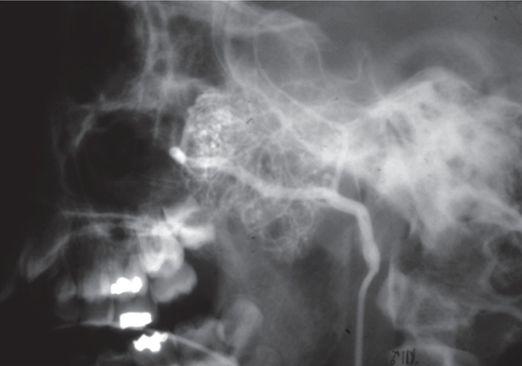
Selective internal maxillary artery injection shows a large enhancing mass.
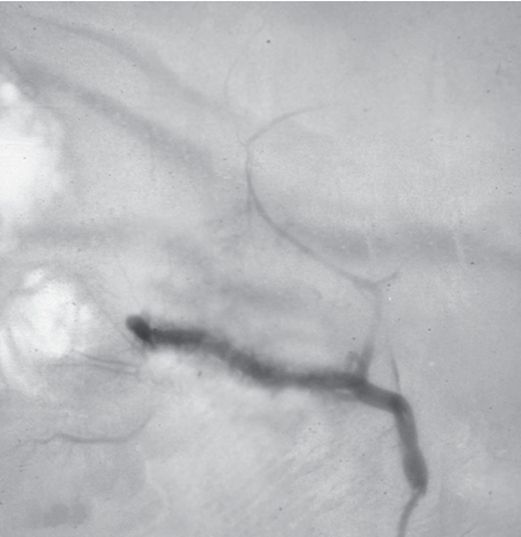
After particle embolization, angiography demonstrates reduction in the pathologic vascularity.
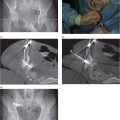
An eight-year-old female presented with back pain.
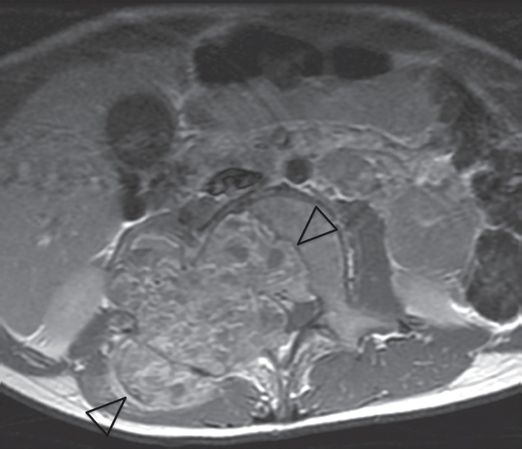
Axial T1 post-contrast fast spin echo MR image shows a lobulated solid and cystic 7 × 7 × 9 cm mass (arrowheads) centered on the L2 vertebral body that invades the spinal canal and right intervertebral foramen extending into the paraspinal tissues.
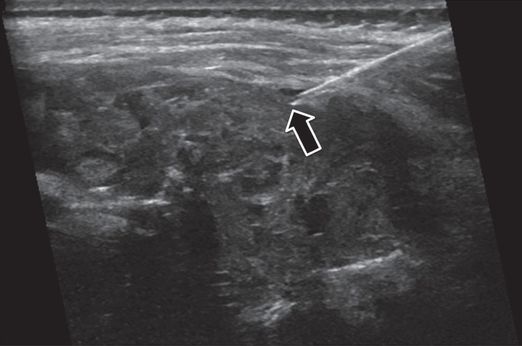
Under US guidance, multiple passes with a 16-gauge OstyCut® core biopsy needle obtained only blood. Several passes with an 18-gauge BioPince® core biopsy needle (arrow) confirmed diagnosis of aneurysmal bone cyst.
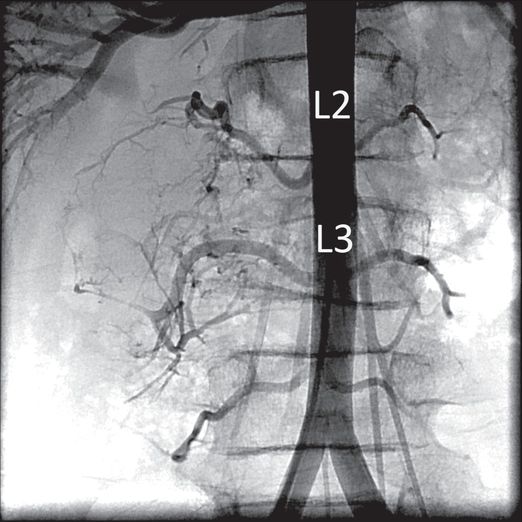
Abdominal aortography showed tumor vascular supply from the right second and third lumbar vertebral arteries.
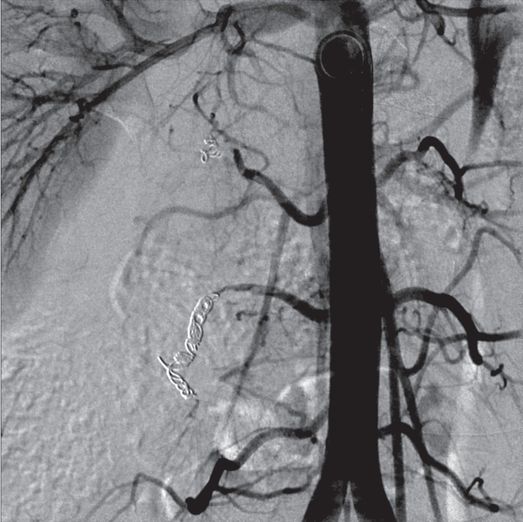
After multiple coils were used to embolize the involved arteries, there was negligible residual tumor vascularity detectable.
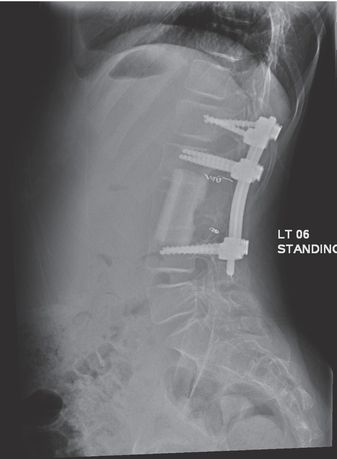
On the following day, the tumor was completely excised surgically. The surgeons reported that the field was essentially bloodless. Spinal reconstruction was successful and the patient was asymptomatic at two-year follow-up.
Embolization of hemangiomas and high-flow vascular malformations
The management of congenital vascular lesions is challenging. In the past few years, the approach to the treatment of congenital infantile hemangiomas (CIHs) has dramatically changed. Over the past decade or two, surgery and embolotherapy were the mainstays for the treatment of CIHs. Today, medical therapy with oral propranolol is the initial therapy of choice, with both surgery and interventional solutions reserved for treatment when medical therapy fails, when there are complications, or when patients are at risk for complications. Examples of at-risk lesions are those high-flow lesions causing congestive heart failure, severe or recurrent bleeding, Kasabach–Merritt syndrome, DIC, and lesions that can lead to poor vision or unilateral blindness.
The historical lack of standardized terminology for vascular lesions resulted in confusion and interfered with data analysis and the creation of standardized therapeutic approaches. In 1982, Mulliken and Glowacki suggested a classification system based on physical examination, histology, and the natural history of the lesions. Although imperfect, this classification led to an improved categorization of lesions and a more organized approach to therapy. Presently, the accepted terminology of vascular anomalies is provided by the International Society for the Study of Vascular Anomalies (ISSVA), which classifies vascular tumors and vascular malformations. Please refer to the ISSVA classification of vascular anomalies for further reading.
Hemangiomas are the most common tumors of infancy and are noted in 1 to 2% of neonates and 10 to 12% of Caucasian children by one year of age. Interestingly, hemangiomas are three to five times more common in girls. Only 30% of hemangiomas are noted at birth, while about 70% are identified in the first month of life. Multiple lesions are found in approximately 20% of infants. The sites most often involved are the head and neck (50 to 60%), trunk (25%), and extremities (15%).
Congenital infantile hemangiomas are defined as benign vascular neoplasms that develop from the dermal capillary network and consist of masses of endothelial cells and pericytes with or without vascular lumina. Typically, a hemangioma evolves through three distinct phases: proliferative, stable, and involution. Once the lesion appears, there is rapid growth for eight to eighteen months with maximum growth usually at one year. The rate and extent of the growth is unpredictable, but growth eventually ceases and the stable phase is then entered. During the proliferative phase, there is endothelial cell hyperplasia, 3H-thymidine uptake, and an increase in the number of mast cells. While in the involution phase, the lesions are less cellular, take up little or no 3H-thymidine, have normal mast cell counts, and have fibrosis and fatty infiltration. The length of the stable period is unpredictable. The stable phase is followed by the involution period, when there is slow and often complete resolution of the lesion. In most series, there is a 50% chance of involution in five years, 75% chance in seven years, and 90% chance in nine years. Unfortunately, not all lesions resolve, and in about 20% of children, there are residual skin changes of cosmetic significance. All CIHs have high endothelial immunoreactivity for erythrocyte-type glucose transport protein GLUT1 (GLUT1 positive) that may be found in cells during all phases of evolution. Interestingly, in hepatic vascular lesions, there are two types of lesions, the focal hemangioendothelioma, which is GLUT1 negative, and the multifocal hepatic infantile hemangioma, which is GLUT1 positive.
Two variants of the common juvenile hemangioma have been described: non-involuting congenital hemangioma (NICH) and rapidly involuting congenital hemangioma (RICH). The NICH is a rare, cutaneous abnormality that grows proportionately with the child but does not regress. There is a slight male predominance, and the lesion generally is solitary, round to oval, with the most common locations being the head and neck, extremities, and trunk. The lesions are usually pink to purple in color with an average diameter of 5 cm. Magnetic resonance imaging and angiographic findings are similar to that of congenital infantile hemangioma. However, these lesions are GLUT1 negative. Histologically, the lesion appears as a lobular collection of small, thin-walled vessels with a large and often stellate central vessel. The interlobular veins seem dysplastic, and the endothelial cells are “hobnailed’ in appearance.
Rapidly involuting congenital hemangioma has similar characteristics to NICH regarding its appearance, distribution, and slight male predominance. The MR and angiographic features are similar, but RICH also have areas of inhomogeneous enhancement, larger flow voids, and arterial aneurysms on angiography. Histologically, the lesions are composed of small to large lobules of capillaries with moderately plump endothelial cells and pericytes surrounded by abundant fibrous tissue. About half of the lesions have lobular loss, fibrous tissue, and draining channels that are large and abnormal. These lesions may contain hemosiderin, calcifications, thrombus, and cyst formation. Like NICH, these lesions are also GLUT1 negative.
Vascular malformations are distinct from hemangiomas, occur much less frequently than hemangiomas, and have a significantly different natural history. These lesions are generally present at birth and tend to enlarge steadily in proportion to somatic growth throughout childhood. Males and females are affected equally. Spontaneous enlargement of lesions may result from interval problems such as thrombosis, infection, ectasia, or the development of new connections especially at puberty. Unlike hemangiomas, vascular malformations do not involute and tend to become more nodular and thickened as they age. Vascular malformations are congenital errors of morphogenesis and may involve formation of one or more vessels. Thus they can be classified as arterial, venous, capillary, lymphatic, or any combination of the above. Histologically, the lesions are not tumors and consist of abnormal clusters of vessels, have normal endothelium, normal mast cell counts, and no non-vascular soft tissue component.
Both hemangiomas and vascular malformations may be defined by their flow characteristics as either low-flow or high-flow. It is important to distinguish the velocity of flow through the lesion since they have different clinical behavior, prognosis, and require a different approach to therapy. Vascular abnormalities may be imaged in a variety of ways at the preference of the referring physician. In general, there is little use for plain radiography and scintigraphy in the work-up of these children. Ultrasound with Doppler can be useful in the initial evaluation and follow-up of these patients. Hemangiomas usually appear as mixed echogenic to hyperechoic soft tissue masses with internal arterial and venous flow in low-resistance vessels. Occasionally, dilated vascular spaces are identified. Distinct feeding and draining vessels may not be seen. Vascular malformations, especially high-flow types, are often distinct structures with multiple anechoic internal structures representing vessels. Spectral waveforms of feeding arteries indicate a low peripheral resistance and dilated draining veins with pulsatile flow suggesting arteriovenous communication. The exact number of supplying vessels is usually not predictable in most cases.
Magnetic resonance imaging and MRA are currently the modalities of choice for diagnosis and treatment planning in most cases. In general, T1- and T2-weighted images are performed in at least two planes (axial and coronal) prior to and after injection of contrast. Magnetic resonance angiography or MRV is obtained in cases with high-flow abnormalities. Proliferating hemangiomas usually appear as homogeneously low or intermediate signal intensity on T1-weighted images and have high signal intensity on T2 sequences. T2-weighted imaging often demonstrates flow voids in high-flow lesions. Involuting lesions are more heterogeneous in appearance. With MRA, vessels are usually identified in high-flow lesions. The appearance of vascular malformations again depends on the lesion type and blood flow characteristics. Arteriovenous malformations will generally demonstrate no significant soft tissue component, flow voids on static imaging, and distinct vessels on MRA. Venous and lymphatic malformations are slow-flow lesions. Venous malformations can enhance homogeneously or heterogeneously if there is thrombus, or can have fluid–fluid levels if the flow is extremely slow. Lymphatic malformations consisting of macrocysts do not enhance, although there can be enhancement of tissue in microcystic lymphatic malformations. Magnetic resonance imaging is recommended to follow children after treatment. In most cases, only high-flow lesions are referred for angiography and vascular intervention. Low-flow lesions are referred for sclerotherapy without embolization.
Although angiography can accurately diagnose and define the vascular pathology, it is only recommended if it is necessary for diagnosis (e.g., a vascular neoplasm) or if vascular intervention is contemplated. Proliferating hemangiomas appear as well-circumscribed masses with intense, persistent tissue blush in a lobular pattern. Involuting hemangiomas have less intense tissue staining and lobules that are more distinctly separated by linear or patchy lucencies that represent fibrofatty septae. Hemangiomas are usually supplied by two to four slightly enlarged branches of normal feeding arteries, which often form a circumferential network around the periphery and give off numerous small feeding vessels. Arteriovenous shunting is not seen and draining veins funnel into a large vein at the base of the mass. High-flow vascular malformations are poorly circumscribed and show faint, if any, vascular blush. The AVMs are supplied by a variable number of enlarged, often ectatic, feeding arteries and have enlarged, often early-draining veins.
Hemangioma
Hemangiomas should only be embolized after failure of appropriate medical therapy (beta-blockers, usually propranolol). Exception to this approach is considered for complicated lesions, especially in young children with congestive heart failure. The technique is similar to that of treating hypervascular tumors and involves selective catheterization and occlusion of feeding arteries with small- to medium-sized particles, coils, glue, or Onyx® (Figures 8.11, 8.12). Caution should be used with coil embolization as recurrence via collaterals can occur if coils are not deployed sufficiently close to the nidus, resulting in increasing complexity of the vasculature. Because these lesions tend to recruit new vascular supplies, in most cases embolotherapy needs to be repeated several times before a stable endpoint is achieved.
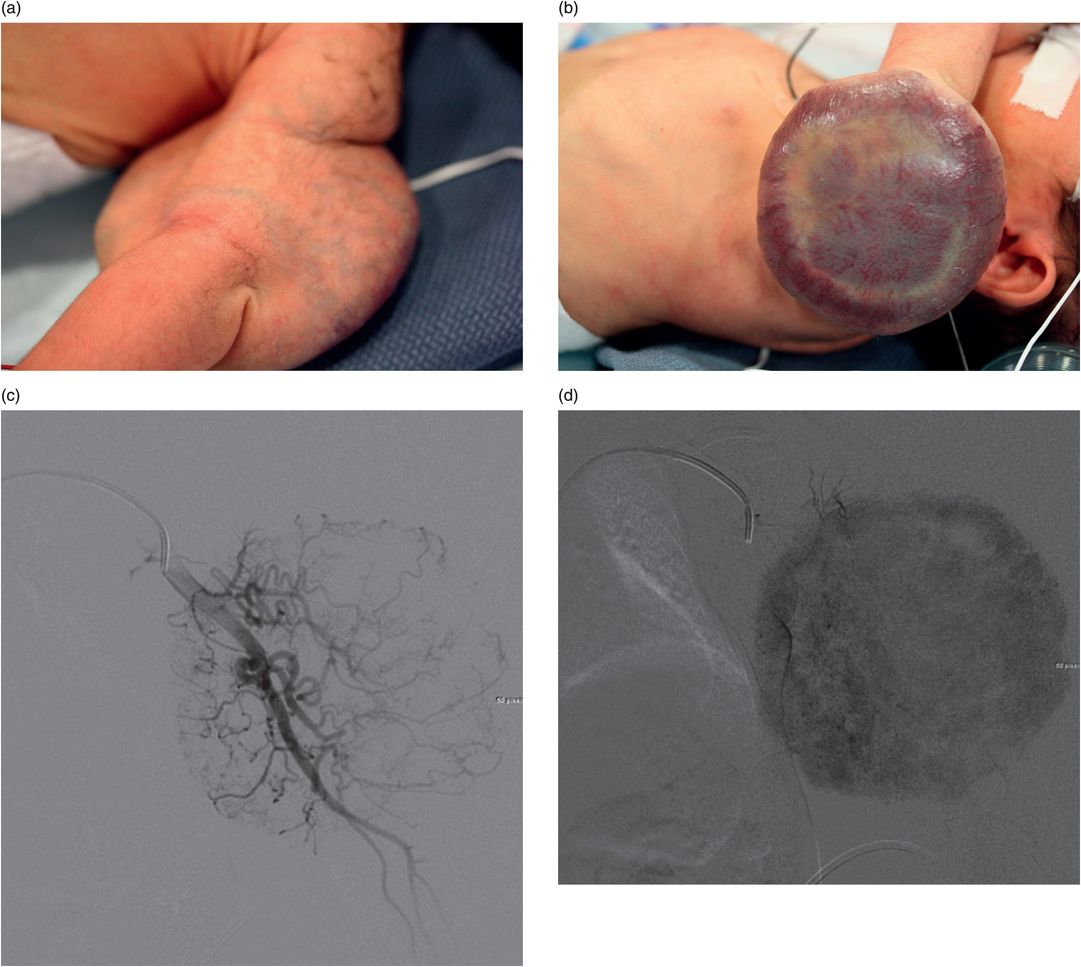
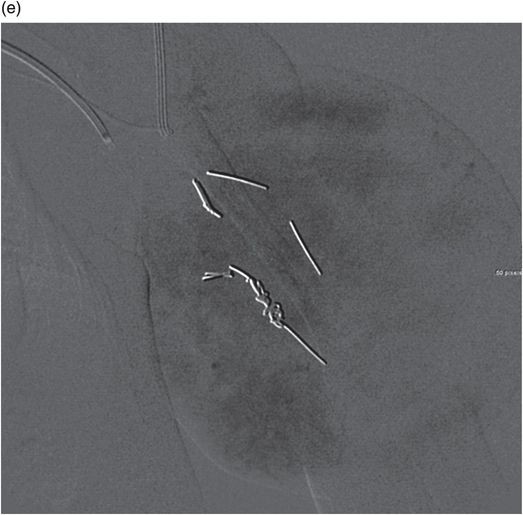
(a,b) Four-day-old female with a large congenital hemangioma involving the left arm associated with cardiomegaly, severe thrombocytopenia (platelets 7,000) and coagulopathy. (c) Transfemoral digital subtraction angiography with the catheter in the brachial artery shows a hypervascular mass. (d) A capillary phase image shows marked enhancement. (e) After coil-embolization there is decreased vascularity prior to surgical resection.
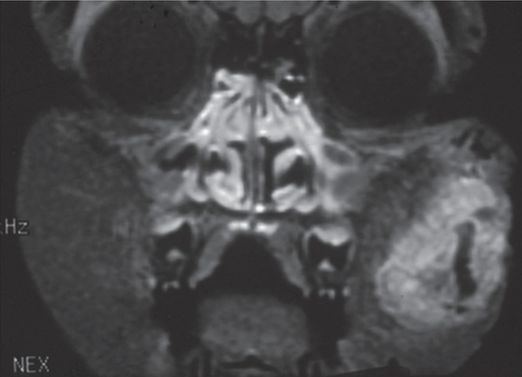
Coronal T1 post-contrast MRI shows a homogeneously enhancing mass with large flow voids.
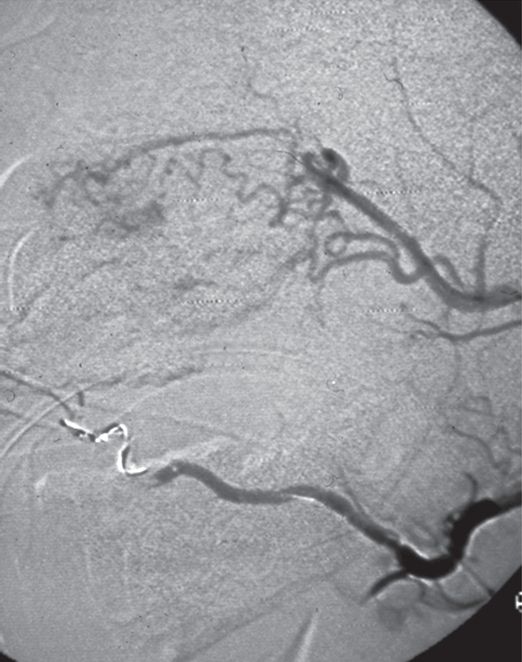
After coil and particle embolization the tumor vasculature is markedly reduced.
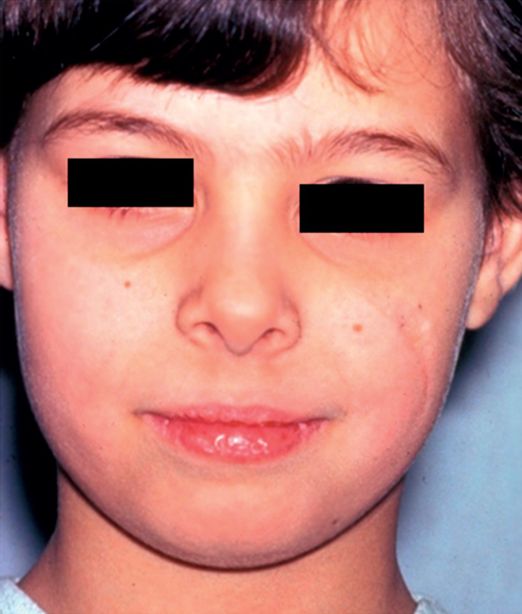
Photograph shows results after cosmetic repair at age eight.
Hepatic hemangioma (hemangioendothelioma)
Hepatic hemangiomas, associated with refractory congestive heart failure, should be considered for emergent treatment when the child requires respiratory support or when drug therapy fails to satisfactorily reduce the excessive blood flow. Treatment of this group of children is challenging. These infants are usually critically ill and in congestive heart failure, with possible renal insufficiency. Thus maintaining their body temperature, carefully monitoring the volume of fluids injected (via contrast and flush), and completing the treatment quickly is extremely important. Prior to treatment, the vascular pathology should be studied carefully, with MRI and US if possible, so that a thorough understanding of the problem and a treatment plan can be developed.
Although hepatic hemangiomas are always supplied by the hepatic arteries, in rare cases they may have extensive systemic collateral arterial and portal venous supply, factors that diminish the effectiveness of hepatic artery embolization. In the presence of extensive portal venous supply and poor hepatic portal perfusion, hepatic arterial embolization has resulted in extensive hepatic necrosis and death. When recognized, the portal vein–hepatic vein fistulae can be occluded with coils using a retrograde transfemoral or transhepatic portal venous approach. These small infants present technical problems in many cases. We have found that in order to minimize the risks for occlusion of the femoral artery in children less than 10 kg, a 4 French directional catheter, through which a microcatheter can be inserted, may be substituted in place of an endovascular sheath.
The type of embolic material used for embolization of the hepatic arteries varies throughout the literature. The most important factor in selecting the type of material to be used is the size of the vascular shunts within the hemangiomas. While particles and tissue adhesives are most effective in achieving distal hepatic artery branch occlusion and reducing revascularization by collaterals, these materials may result in fatal embolization to the pulmonary circulation. Proximal occlusion of the main hepatic artery, distal to the origin of the gastroduodenal artery or hepatic artery branches, using balloons or coils (our preference) has been effective in relieving cardiac failure, although glue or Onyx® are therapeutic alternatives. This approach has several additional advantages, including minimizing injected fluid volume, short time to vessel occlusion, and completion of the case with a high degree of safety. Hepatic artery branch occlusion using microcoils (0.018-inch, ≥5 mm length) appears to be relatively safe and effective.
It is important to keep in mind that the goal of embolization in children with symptomatic hemangioendothelioma is not to eradicate the tumor, but to control life-threatening complications, most importantly CHF, by diminishing arterial blood flow, thereby reducing the rapid AV shunting through the lesion, until the malformation involutes in the range of 12 to 18 months of age. Thus care should be taken to occlude the fewest number of vessels necessary to control the child’s CHF.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree