Case presentation
A 16-year-old male presents with left-sided chest pain for the past week. He states that the pain began during band practice. He plays the trombone and states that he “must have pulled a chest muscle” because he was “playing really loud.” He immediately felt the pain but continued playing. He reports no shortness of breath, but the pain is worse with deep inspiration and he feels like he is breathing faster than usual after marching. He denies recent illness and abdominal or back pain. He was seen by his primary care provider 2 days ago and had an electrocardiogram that was normal. He was told to take ibuprofen which has helped some.
Physical examination reveals a tall, thin, pleasant adolescent in no obvious distress. He is afebrile; his heart rate is 85 beats per minute, respiratory rate is 18 breaths per minute, and blood pressure is 118/70 mm Hg, and he has a pulse oxygenation reading of 93% on room air. He converses easily but does complain of chest pain to the left side. He has slightly diminished breath sounds to the left without crackles or rhonchi. He has no retractions. The remainder of his physical examination is unremarkable.
Imaging considerations
Patients with suspected pneumothorax may have imaging to confirm the diagnosis. In patients with concern for tension pneumothorax or decompensating clinical status, imaging should not delay immediate treatment and stabilization.
Plain radiography
This imaging modality is the initial test of choice for patients when a pneumothorax is a clinical consideration. It is readily available and inexpensive relative to other modalities. The presence of a pleural line and the absence of vascular markings beyond this line are the hallmarks of pneumothorax identification on chest radiography. The deep sulcus sign, which is an area of hyperluncency in a depressed costophrenic angle, is pathognomonic for pneumothorax and seen on nonerect radiographs. Pleural air may also be seen in the subpulmonic and anteromedial areas and laterally. , In an adult meta-analysis, the pooled sensitivity and specificity of chest radiography for pneumothorax were 52% and 100%, respectively. ,
The optimal position of the pediatric patient undergoing radiography for suspected pneumothorax has not been well studied. Traditionally upright inspiratory chest radiographs have been utilized, but this may not be feasible in the pediatric population, since cooperation may be an issue, or in the trauma patient, who may be immobilized. While upright inspiratory films are preferred, supine or lateral decubitus views may be obtained. A chest radiograph should visualize the entire chest and lungs.
Computed tomography (CT)
CT is often used as a follow-up study in patients with recurrent pneumothorax or if there is a clinical concern for pleural bullae/blebs. It is generally not a first-line imaging modality; however, in cases of penetrating thoracic trauma or concern for vascular injury, CT may be utilized.
For patients with chest radiography that reveals large pulmonary bullae or if there is concern for structural anomalies of the lung, CT may be indicated and is often utilized if operative management is being considered. , , CT for first-time spontaneous pneumothorax is not recommended by the American College of Chest Physicians guidelines. , However, there are authors that recommend obtaining CT on all patients with first time primary spontaneous pneumothorax to evaluate for pulmonary abnormalities and to assist in a management plan but this is not universally recommended. In one series of patients, Tsou et al. did not find differences in length of stay, length of operation, or rate of complications in patients who underwent CT prior to thoracoscopic surgical procedures. ,
Ultrasound (US)
The use of ultrasound to identify pneumothorax has gained interest. The advantages are the lack of ionizing radiation, lower expense relative to other advanced modalities, and general availability. There are several findings suggestive of pneumothorax on sonography. The absence of lung sliding and so-called comet tails (grayscale artifacts that are present in normal lung parenchyma) has been shown to have a sensitivity and specificity of 100% and 96.5% for pneumothorax ; the absence of comet tails was found to be present in 100% of patients with pneumothorax.
Magnetic resonance imaging (MRI)
This modality is not indicated for the evaluation of patients with suspected pneumothorax.
Imaging findings
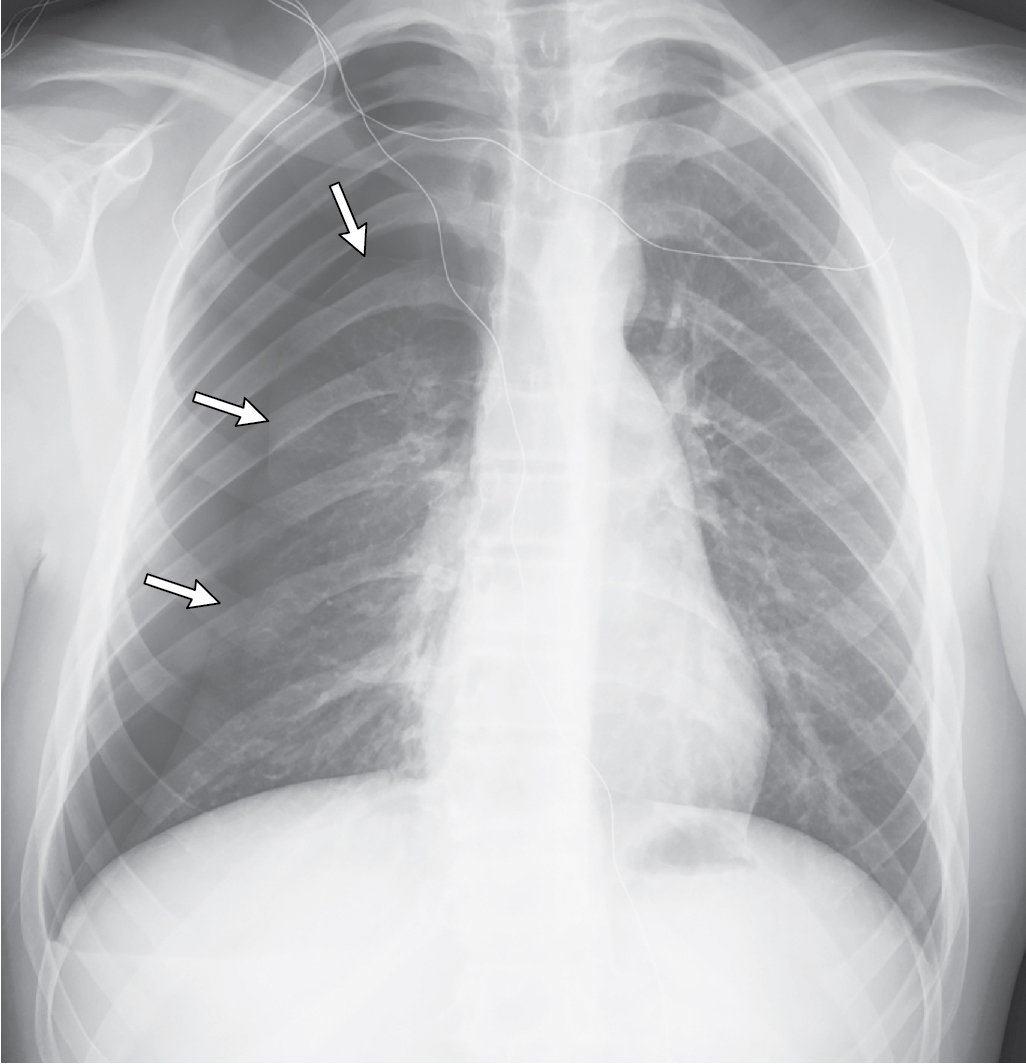
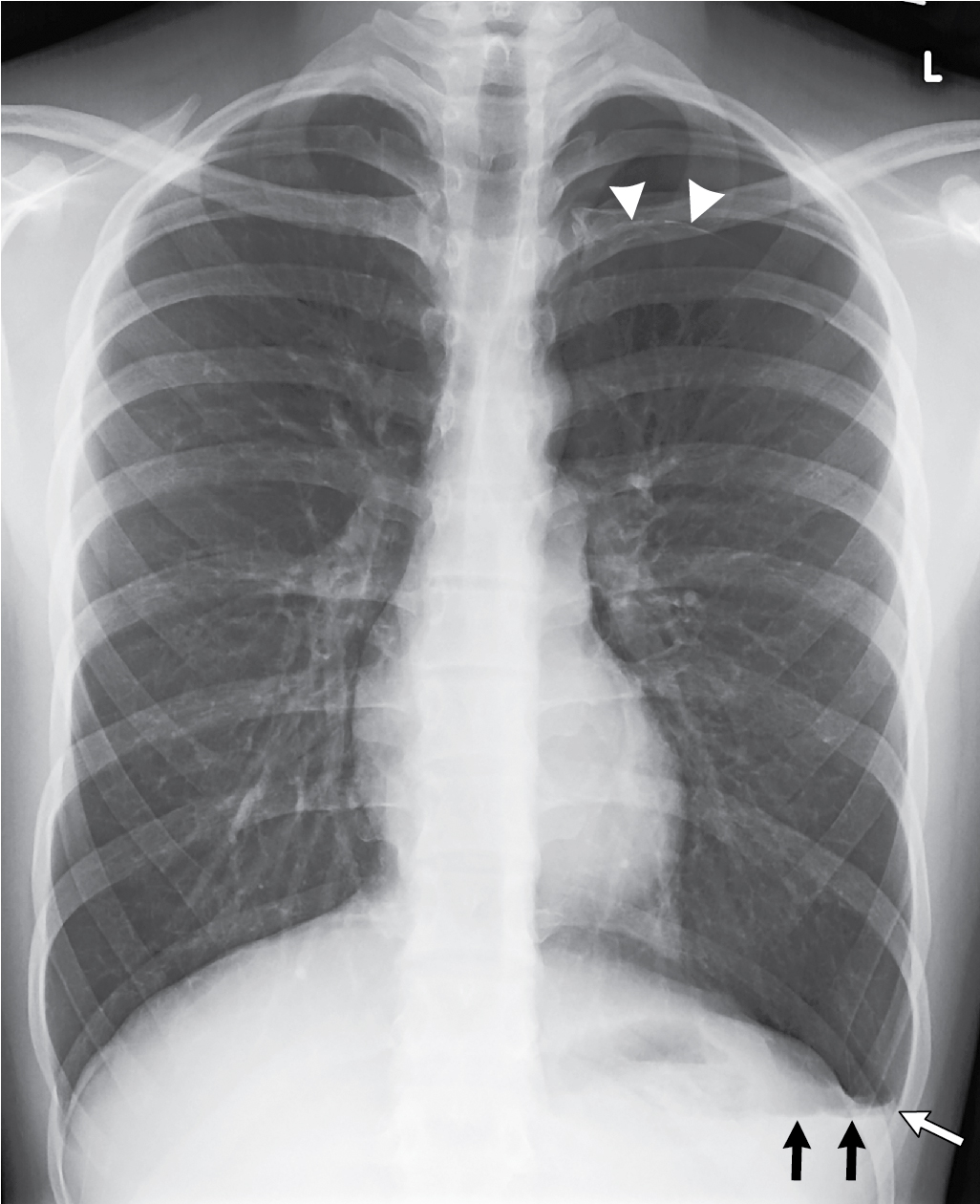
Chest radiography was obtained. This two-view study demonstrates a left-sided pneumothorax, and a very small left pleural effusion, without tension or other obvious pulmonary abnormalities ( Fig. 42.1 ).
Case conclusion
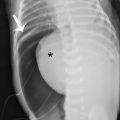
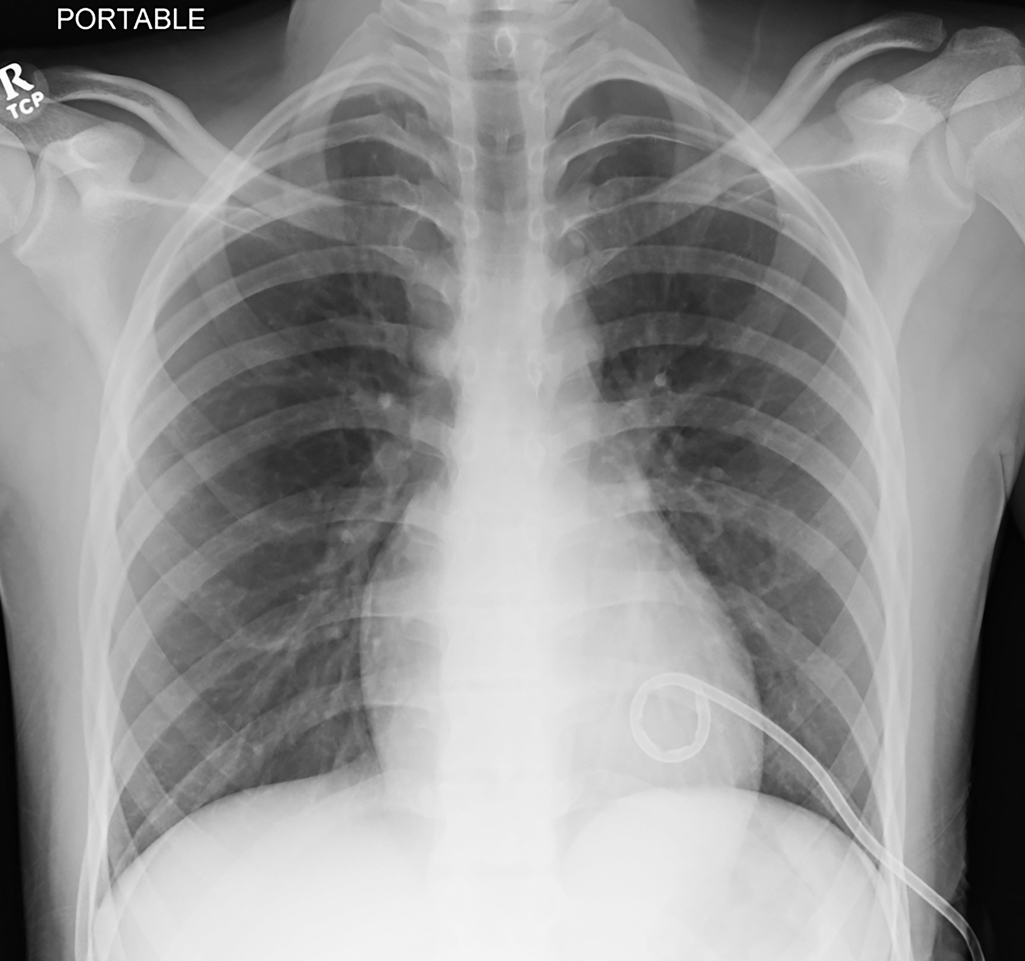
The Pediatric Surgery team evaluated the patient in the emergency department. Oxygen by face mask delivery was initiated and the decision was made to admit the patient to the hospital for monitoring and serial chest radiography. Later during the admission day, it was revealed that the patient had two prior episodes of chest pain and similar symptoms which resolved on their own after several days. This prompted concern for recurrent episodes of pneumothorax. Chest CT was obtained to evaluate for the presence of underlying blebs or bullae. CT showed the pneumothorax, but no other findings. A pigtail catheter chest tube was placed for decompression of the pneumothorax, which was successful ( Fig. 42.2 ). The patient was observed in the hospital for several days with serial radiography documenting pneumothorax resolution. The chest tube was removed and follow-up radiography demonstrated no residual pneumothorax. He was discharged with follow-up and did well.
Pneumothorax is defined as the presence of air between the visceral and parietal pleura and, due to pressure differences between the lung and the pleura space, leads to lung collapse. Pneumothorax can be spontaneous or traumatic. Typical presenting symptoms of pneumothorax include ipsilateral chest pain, which may radiate to the ipsilateral shoulder, and dyspnea. Symptom severity is related to the size of the pneumothorax.
There are several manifestations of pneumothorax. ,
Primary spontaneous pneumothorax
This form of pneumothorax is not associated with radiographically identified lung disase. , Primary spontaneous pneumothorax occurs more commonly in adolescent males who are thin and tall; most cases occur while at rest. , Smoking is a known risk factor. , Although patients with primary spontaneous pneumothorax do not have clinically apparent lung disease on chest radiography, the majority are found to have subpleural bullae when undergoing video-assisted thoracoscopic surgery (VATS).
Secondary spontaneous pneumothorax
Patients with asthma, cystic fibrosis, Marfan syndrome/Marfanoid habitus, and connective tissue disorders are prone to the development of secondary spontaneous pneumothorax. , Patients may develop a pneumothorax as a sequelae from severe coughing or pneumonia. These patients tend to have a more severe clinical presentation and have higher recurrence rates.
Traumatic pneumothorax
Traumatic pneumothorax may be due to iatrogenic causes (such as when a patient is on ventilatory support, after undergoing transthoracic needle biopsy, which is a leading cause of iatrogenic pneumothorax, and after central line placement) or due to blunt or penetrating thoracic trauma. In the latter case, there may be significant associated injury.
Open pneumothorax
This variety or pneumothorax is often associated with other injuries resulting from thoracic trauma.
Tension pneumothorax
Tension pneumothorax is due to the entry of air into the pleural space that cannot exit, leading to ipsilateral lung collapse with a mediastinal shift toward the contralateral side. This can impede venous return, reduce cardiac output, and compromise normal cardiovascular status and hemodynamic stability. Clinical symptoms suggestive of tension pneumothorax include hypotension, tracheal deviation, tachycardia, and cyanosis, in addition to chest pain and dyspnea. ,
There are no consensus guidelines for the management of pediatric pneumothorax, but in general, management depends on many factors, including the type, size, and location of pneumothorax. The patient’s history, including existing medical conditions and previous episodes, also is taken into consideration. The size of the pneumothorax is a critical factor in determining a treatment plan. , There are, however, no universally accepted normal lung volume values in pediatric patients. The American College of Chest Physicians classifies pneumothorax size by using the distance between the lung apex and the ipsilateral dome of the thoracic cavity; a small pneumothorax is less than 3 cm and a pneumothorax is considered large when this distance is greater than 3 cm. , , Other classification systems include the British Thoracic Society system, which utilizes a greater than 2-cm gap between the pleural edge and the chest wall as a large pneumothorax. ,
Management
Small primary spontaneous pneumothorax
Small pneumothoraces are generally treated with observation only and generally reabsorb at a rate of 1% to 2% per day. , Some authors recommend oxygen therapy, which, due to “nitrogen washout” (a process that increases nitrogen absorption, increasing the rate of pneumothorax reabsorption), has been shown in some studies to increase the rate of pneumothorax reabsorption to 5% per day. , , Other studies have demonstrated no acceleration in pneumothorax reabsorption with oxygen admnistration. A follow-up chest radiograph, which may be obtained based on ongoing clinical examination, should reveal a stable, if not smaller, pneumothorax; if this is the case, no further treatment is indicated; treatment is indicated if there is worsening of the pneumothorax. , Patients with a small, minimally symptomatic, clinically stable pneumothorax who have follow-up can be managed as outpatients after a period of observation with repeat imaging in the emergency department. , , If adequate outpatient observation and follow-up cannot be assured, the patient should be admitted to the hospital. , A conservative management option would also be admission for supplemental oxygen, serial radiography, and subspecialty consultation.
Large primary or small symptomatic primary pneumothorax
Patients with a large pneumothorax generally are admitted to the hospital and are treated with placement of either a chest tube or a small-bore (“pigtail”) catheter. , , There are few studies comparing the use of pigtail catheters versus traditional chest tubes in the pediatric population, but one study demonstrated similar efficacy, similar lengths of stay, and less need for analgesic medications in patients who had pigtail catheter placement. , The use of a modified Seldinger technique has been shown to be an effective and less invasive method for managing pneumothoraces. , , The British Thoracic Society guidelines suggest attempting needle aspiration of the pneumothorax followed by careful observation in the emergency department for several hours; hospital admission is recommended for pneumothorax that recurs, if there is inadequate pain control, or if outpatient follow-up cannot be assurred. , The American College of Chest Physicians does not recommend drainage of pneumothorax by needle aspiration. ,
Secondary pneumothorax
Patients with secondary pneumothorax often are managed by subspecialists due to underlying chronic medical conditions. Management of these patients should be done in consultation with the subspecialist who is caring for the patient.
Tension pneumothorax
Since tension pneumothorax is a life-threatening condition, immediate recognition and management are warranted. If a tension pneumothorax is suspected, do not delay treatment for confirmatory imaging. Needle decompression is the first intervention, followed by chest tube thoracostomy. Management should also address coexisting injuries.
Open pneumothorax
Initial management consists of the application of a “flutter valve” dressing (a dressing secured on three sides with the fourth side open) and placement of a chest tube. Patients require surgical intervention for definitive management.
There are other management options available for pediatric patients with pneumothorax, although when to utilize these techniques is a matter of debate. Surgical management involves the resection of blebs or bullae, or apical resection. VATS has been proven to be effective and well tolerated. The use of sclerosing agents may also be considered. The instillation of these agents causes irritation between the parietal and visceral pleural layers, causing scarring and closing this space, with the goal of preventing recurrence. , Indications for surgical management include second ipsilateral pneumothorax, first contralateral pneumothorax, bilateral pneumothorax, and persistent air leak. , ,
The recurrence rate for a primary spontaneous pneumothorax has been reported to be 30% to 50%, with the majority recurring within the first 2 years after diagnosis. , Risk factors for recurrence include young age, thin body habitus, evidence of pulmonary fibrosis, and smoking.
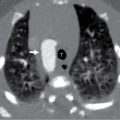
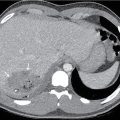
The patient in Figs. 42.3–42.5 , a 16-year-old male, developed acute onset of right-sided chest pain while taking an examination at school. He presented with mild tachypnea and a room air oxygenation of 92%. Chest radiography revealed a large right-sided pneumothorax. He had a chest tube placed with subsequent resolution of the pneumothorax. He presented several months later with a recurrence of the pneumothorax, had CT imaging revealing several blebs, and underwent a VATS with apical blebectomy, pleurodesis, and chest tube placement.