Chapter 34 Cancer of Unknown Primary
Introduction*
Unproved theories regarding the localization of the primary CUP site include (1) the primary tumor has involuted and only the metastatic disease is evident and (2) metastatic disease is favored over the primary growth based on the phenotype and genotype of the tumor.1
Epidemiology and Risk Factors
Worldwide, CUP is one of the 10 most frequent cancers constituting about 3% to 5% of all cancer cases.2 In 2011, according to the American Cancer Society, there were an estimated 30,500 cases of “Other & unspecified primary sites” in the United States or approximately 2% of all cancer cases and an estimated 44,260 deaths.2 At presentation, the median age is approximately 60 years and slightly more frequent in males.3
Anatomy and Pathology
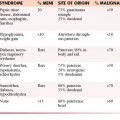
Because only carcinomas are included in the diagnosis of CUP, four main histologic types of CUP have been described: well to moderately differentiated adenocarcinomas (50%), undifferentiated or poorly differentiated carcinomas (30%), squamous cell carcinomas (15%), and undifferentiated neoplasms (5%). The latter group includes lymphomas, sarcomas, germ cell tumors, poorly differentiated carcinomas, neuroendocrine tumors, and embryonal malignancies that can be characterized by immunohistologichemistry.4 In children, CUPs represent less than 1% of solid tumors and the majority of these tumors are embryonal malignancies.2
The chromosomal and molecular abnormalities identified in CUPs are beyond the scope of this chapter.
Clinical Presentation
These patients face a grim prognosis with reported survival rates of 6 to 10 months for those enrolled in clinical studies.2
However, those CUP patients not enrolled in clinical trials have reported life expectancies of 2 to 3 months.5 Given the diverse clinical presentation of these patients, it is best to classify them into favorable and unfavorable subsets, with the former having a better prognosis.6
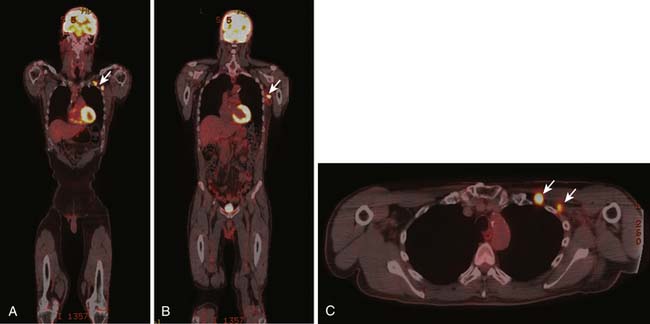
1. Squamous cell carcinoma involving cervical lymph nodes (Figure 34-1).
2. Women with adenocarcinoma involving only axillary lymph nodes.
3. Women with peritoneal cavity papillary serous adenocarcinoma.
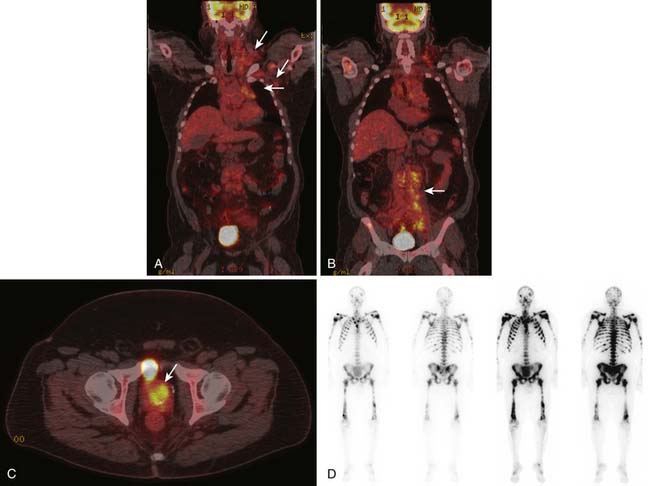
4. Men with elevated PSA and blastic osseous metastases (Figure 34-2).
5. Poorly differentiated carcinoma with midline distribution (extragonadal germ cell tumor syndrome).
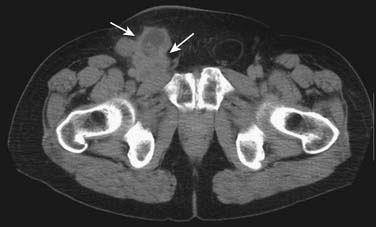
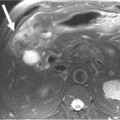
6. Isolated inguinal adenopathy with squamous cell carcinoma (Figure 34-3).
7. Poorly differentiated neuroendocrine carcinomas.
8. Patient with a single, small, potentially resectable metastasis.
1. Adenocarcinoma primarily metastatic to the liver.
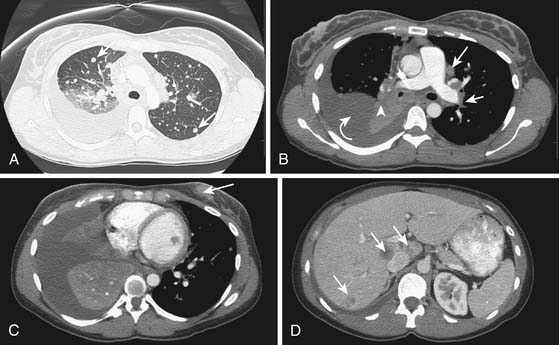
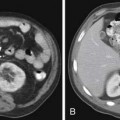
2. Adenocarcinoma with multiple metastases to the lungs (Figure 34-4).
3. Adenocarcinoma with mainly diffuse osseous metastatic disease.
4. Multiple cerebral metastases.
5. Malignant ascites with nonpapillary adenocarcinoma histology.
To complicate the clinical presentation, more than 50% of CUP patients present with multiple sites of metastatic disease and approximately 30% have three or more organs involved with metastatic disease. In comparison, in patients with known primary tumors, fewer than 15% have metastatic disease in three or more sites.5,7,8
Patterns of Tumor Spread
The most common sites for CUP metastatic disease are the liver, lungs, bones, and lymph nodes. However, when compared with tumors of known origin, CUPs typically present with unpredictable patterns of spread. For example, when pancreatic and hepatic carcinomas present as CUPs, there is a higher incidence of lung and osseous metastatic disease compared with a known primary originating in the pancreas and liver. Brain and osseous metastatic disease from a CUP originating in the gastrointestinal tract is more commonly seen than that from a known gastrointestinal primary carcinoma. Approximately 50% of pulmonary carcinomas have osseous metastatic disease, whereas CUPs of pulmonary origin have a lower incidence of osseous metastatic disease. Hepatic and pulmonary metastatic disease is seen more frequently in cases that present as a CUP from a prostatic carcinoma than from a known prostatic carcinoma.9–12