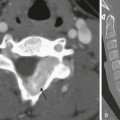
Chapter 97 Overview: First described in 1670, esophageal atresia was a universally fatal anomaly until late in the fifth decade of the twentieth century, when pediatric surgeons began successfully treating these children. Esophageal atresia with and without TEF is part of a heterogeneous spectrum of anomalies that ranges from isolated esophageal atresia to isolated TEF. Lesions are typically classified according to the association and location of the fistulous connection between the esophagus and the airway (Fig. 97-1). The incidence of esophageal atresia is approximately 1 in 4500 births in the United States and 1 in 3500 births worldwide, with a tendency to occur more commonly in boys, although the gender difference varies with the type of lesion. Maternal risk factors include white race, first pregnancy, and advanced age. There is also an increased risk in the siblings of patients with esophageal atresia.1,2 Figure 97-1 Types of esophageal atresia, with and without tracheoesophageal fistula. Etiology: The development of esophageal atresia is poorly understood and still debated. Dominant theories in the past have been based on the model of a tracheoesophageal septum, a lateral ridge of tissue believed to separate the foregut into a dorsal component (the esophagus) from a ventral component (the trachea) in a caudocephalic direction. However, further investigation has suggested that this process does not occur in humans.1,3 Kluth and Fiegel3 suggest that the formation of the esophagus and trachea is the result of “a system of folds” in the cranial and caudal ends of the tracheoesophageal space that move toward each other, thus separating the space into the ventral trachea and dorsal esophagus. Following this theory, esophageal atresia with fistula results from an abnormally ventral position of the dorsal fold; isolated esophageal atresia is attributed to a vascular accident rather than abnormal separation of the alimentary and respiratory canals.3,4 The association of the esophageal atresia complex with conotruncal anomalies and anomalies associated with DiGeorge syndrome suggests that abnormal development of pharyngeal arches may be related to this malformation.1 Further research with animal models concerns the investigation of genes that influence the temporal and spatial sequence of expression of molecular and genetic pathways, including retinoic acid receptors, and sonic hedgehog pathway effectors. Human teratogens that have been associated with this anomaly include abnormal maternal hormonal milieu (including estrogen, progesterone, and thyroid hormones), intrauterine exposure to thalidomide and diethylstilbestrol, and maternal diabetes.1 Clinical Presentation: Patients with esophageal atresia with or without fistula typically come to medical attention prenatally or soon after birth. Patients with isolated esophageal atresia are most likely to be seen with maternal polyhydramnios and to be identified at prenatal evaluation. Esophageal atresia and TEF may have been suspected during pregnancy, but neonates generally come to medical attention in the first few hours of life with excessive salivation and drooling, choking and regurgitation during feeds, and sometimes with cyanosis and/or respiratory distress.1 The incidence of associated congenital anomalies has been reported to be between 50% and 70% and is most common in infants with isolated esophageal atresia and least common in children with isolated TEF.5 The most common associated anomalies, in descending order of frequency, include cardiac, genitourinary, gastrointestinal, musculoskeletal, and neurologic anomalies. Approximately half of the children with esophageal atresia who have associated anomalies can eventually be classified as having a known syndrome, either chromosomal or named, such as VACTERL (vertebral anomaly, anorectal atresia, cardiac lesion, tracheoesophageal fistula, renal anomaly, limb defect), (Fig. 97-2) CHARGE (coloboma, heart malformation, choanal atresia, mental retardation, genitourinary and ear malformations), Fanconi anemia, Opitz and Goldenhar syndromes.6,7 In particular, patients with isolated esophageal atresia have an increased incidence of trisomy 21 (11%) and duodenal atresia (10%). Figure 97-2 VACTERL (vertebral anomaly, anorectal atresia, cardiac lesion, tracheoesophageal fistula, renal anomaly, limb defect) syndrome. Imaging: Imaging is used in both the prenatal and neonatal diagnosis of esophageal atresia. Esophageal atresia is a cause of polyhydramnios. Fetuses with esophageal atresia may also have an absent or small stomach bubble. Unfortunately, these ultrasound findings alone lack specificity, with a reported 20% to 40% positive predictive value.8 Accuracy in diagnosis is improved when a blind-ending pouch is visualized in the fetal neck, and newer data suggest that fetal magnetic resonance imaging (MRI) may play an increasingly important role in prenatal diagnosis.9,10 More commonly, the diagnosis is made in the immediate neonatal period. If the diagnosis of esophageal atresia is suspected in the first few hours of life, placement of a flexible feeding tube or enteric tube should be attempted. Introduction of contrast into the pouch is not typically necessary for confirmation of the diagnosis, although introduction of air into the pouch may improve its identification. Introduction of contrast into the pouch may reveal the rare patient who has a proximal fistula with or without atresia, although today this is typically diagnosed endoscopically. On radiographs of the abdomen, absence of bowel gas indicates esophageal atresia without a distal TEF (Fig. 97-3). In this setting, a TEF from the proximal pouch cannot be excluded with chest radiographs and requires endoscopy for diagnosis. Likewise, the presence of bowel gas on abdominal radiographs in a patient with esophageal atresia indicates the presence of a distal TEF (Fig. 97-2 and 97-4).1 In patients who are intubated, air may be forced into the stomach through the fistula, resulting in gastric overdistension, which may result in perforation. In addition, approximately 2.5% of patients will have a right-sided aortic arch that should be identified on preoperative echo or ultrasound since esophageal repair is performed on the side opposite the arch.1,11,12 Figure 97-3 Isolated esophageal atresia. Treatment/Follow-up: Surgical repair is typically undertaken within 1 to 2 days, after the clinical workup is complete, including treatment of more emergent medical issues. Open or thoracoscopic division/ligation of the fistula and primary esophageal anastomosis is the treatment of choice. For long-gap atresia, a gastrostomy tube can be placed for palliation, while mechanical efforts are made to elongate the proximal and distal pouches prior to primary repair. Alternatively, if these procedures fail, an esophageal substitute (usually colon or ileocolon) can be interposed between the proximal and distal segments. Isolated esophageal atresia is typically associated with a small proximal pouch and almost no thoracic esophagus (see Fig. 97-3), and patients are typically palliated with a gastrostomy tube in the first 24 to 48 hours of life, followed by delayed repair.1 Imaging also plays an important role in the evaluation of early and late complications of esophageal atresia repair. Complications include anastomotic leak, anastomotic stricture, and recurrent TEF; these are evaluated with esophagram (e-Fig. 97-5). Because standard esophageal contrast studies miss a reported 50% of recurrent fistulae, bronchoscopy may also be required if recurrent fistula is suspected.1 e-Figure 97-5 Recurrent fistula. Overview: Congenital, isolated TEF without atresia (type 3 in Fig. 97-1) is often termed an H-type fistula, and it differs significantly from the other types of tracheoesophageal malformations in its presenting spectrum and features. This type of fistula can be difficult to identify, both clinically and radiographically.13 Large fistulae usually present very early in life and are relatively easy to see on an esophagram (Fig. 97-6). More commonly, the fistulae are small, inconstantly patent, and may require repeated examinations to identify. A major reason for inconstant patency of a fistula is that normal esophageal mucosa can be redundant, which can transiently occlude the esophageal side of the fistula.14 Although rare, multiple fistulae can be present.15 Figure 97-6 H-type fistula. Rarely, a bronchus originates directly from the esophagus, resulting in an anomaly termed esophagotrachea or esophageal bronchus (Fig. 97-7). This anomaly leads to severe respiratory distress with feeding and may be associated with esophageal atresia, TEF, or both.16 Bronchoesophageal fistula between the esophagus and a normal bronchial tree has also been described.17 Figure 97-7 Connections between esophagus and airway. Clinical Presentation: These patients typically come to medical attention with recurrent episodes of coughing or choking and may progress to recurrent pneumonias, either in infancy or later in childhood.18,19 Patients with unexplained, intermittent respiratory distress and recurrent pneumonia should always be considered at risk for the presence of a TEF, and radiographic examination is warranted. Imaging: This is often termed an H-type fistula because of its appearance on an esophagram: it connects to the trachea posteriorly and to the esophagus anteriorly as it courses cephalad from the esophagus to the trachea (see Fig. 97-6). The examination should begin with a single contrast esophagram using digital, pulsed fluoroscopy in the right lateral to slightly right anterior oblique position. The position should be optimized during the examination to allow the best view of the anterior wall of the esophagus and posterior wall of the trachea. One of the keys of this examination is achieving full distension of the esophagus, because this is important to optimize filling and visualization of the fistula. If no abnormality is detected while the patient is swallowing, an esophageal catheter may be placed under fluoroscopic guidance, particularly in patients with normal prior examinations, in whom a fistula remains suspect, or in those in whom contrast is seen within the trachea from an unclear source.20 The examiner slowly withdraws the catheter from the distal esophagus in the cephalic direction, while carefully injecting contrast material with enough velocity and volume to distend the esophagus maximally under constant fluoroscopic monitoring. The catheter should have an end hole, without more proximal side holes, for adequate control of the contrast infusion. Even if a fistula is noted, the injection should continue until the catheter is withdrawn into the hypopharynx, but care must be taken not to allow contrast material to spill over into the trachea. Although there is a danger of spilling contrast material into the airway with injection of the high cervical esophagus, it is important to examine this area, because many fistulae occur at the level of the lower cervical or upper thoracic spine (see Fig. 97-6, C).21 Slow retraction of the catheter and careful fluoroscopic monitoring allow constant visualization and help to prevent overflow into the trachea.22
Congenital and Neonatal Abnormalities
Esophageal Atresia with and Without Tracheoesophageal Fistula
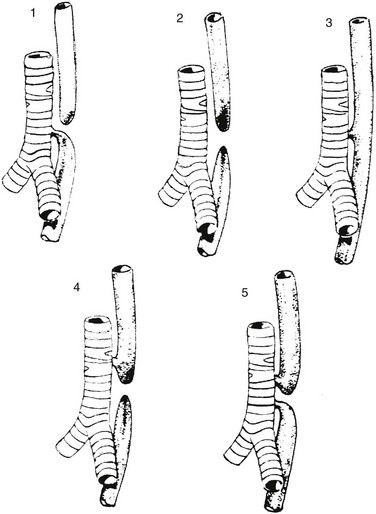
Type 1 represents esophageal atresia with a distal fistula and is the most common form, occurring in approximately 84% of patients. Type 2 illustrates esophageal atresia without a fistula, seen in about 6%. Type 3 is an isolated H-type fistula (4%), and type 4 describes esophageal atresia with the fistula arising from the proximal pouch (5%); type 5, with fistulae arising from both the proximal and distal pouch, is the least common (~1%).1
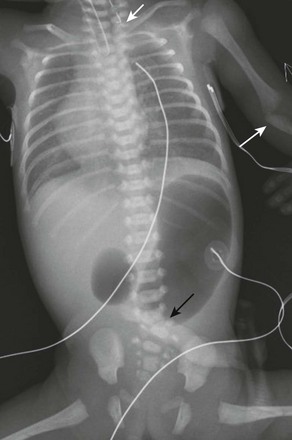
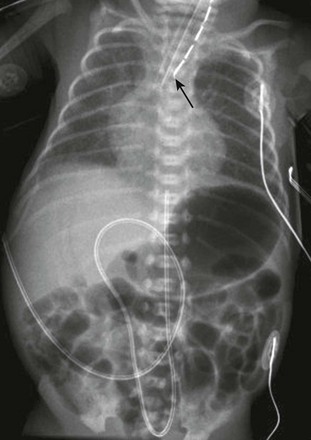
Newborn girl born at 32 weeks gestation demonstrates the abnormalities seen with the VACTERL association. The patient is intubated because of the lung disease of prematurity. The enteric tube (short white arrow) ends just above thoracic inlet in the proximal esophageal pouch, and abdominal bowel gas indicate a distal tracheoesophageal fistula, whereas a “double bubble” sign, and absence of distal bowel gas indicate duodenal atresia. There are 13 pairs of ribs, and a hemivertebra between L5 and S1 (black arrow). Note the absent radius in the left arm (white arrow points to the ulna).
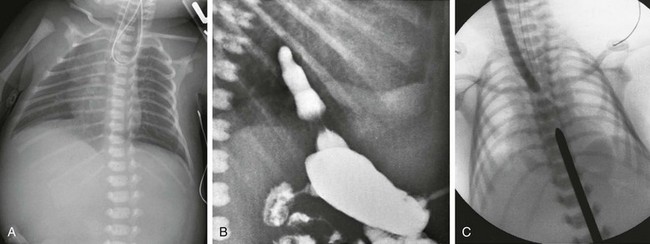
A, Newborn boy born at 37 weeks gestation with isolated esophageal atresia. The enteric tube turns cephalad in the blind-ending proximal esophageal pouch, and abdominal bowel gas is absent. B, Gastrostomy injection with reflux into the short distal esophageal segment in a patient with esophageal atresia without a distal fistula. C, Esophageal atresia with the gap between the segments confirmed at surgery. Frontal view reveals bougies in both the proximal and distal segments, showing a long gap between them.
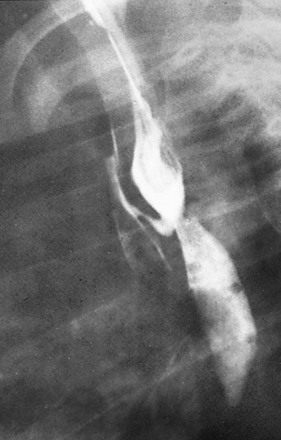
Contrast esophagram demonstrates a fistulous connection with the airway at the site of the anastomosis, a finding typical of fistula developing as a postoperative complication.
Isolated Tracheoesophageal Fistula
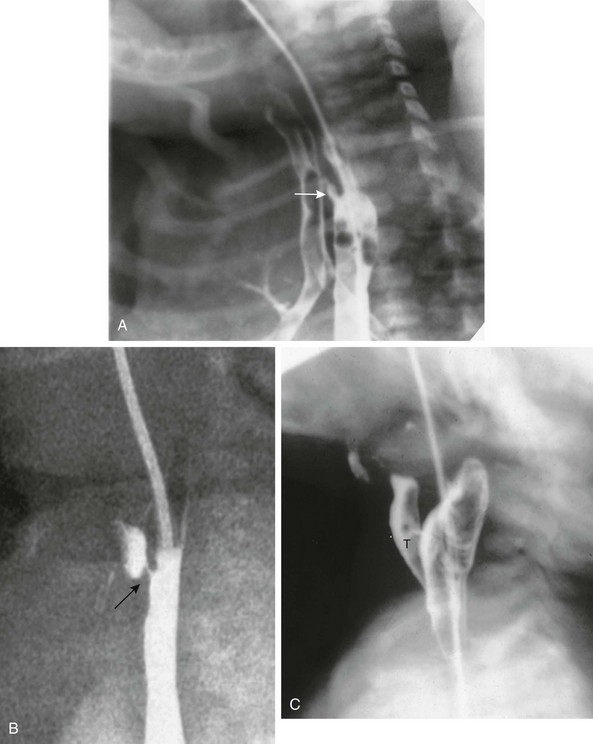
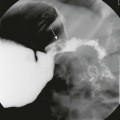
A, Tracheoesophageal fistula (arrow) in a 7-day-old boy with imperforate anus. B, Demonstration of another H-type fistula (arrow) from the upper cervical esophagus to the trachea, using the technique of contrast injection through a feeding tube with very careful volume control. C, Large H-type fistula from the upper cervical esophagus to the trachea (T).
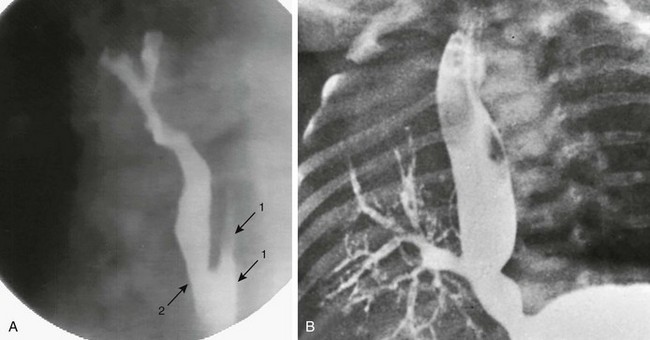
A, Congenital esophagobronchial fistula. Oblique view shows esophagus (arrows with 1) and bronchus to right upper lobe (arrow with 2). B, Esophageal bronchus. Frontal view from an esophagram demonstrates the origin of the right main bronchus from the distal esophagus.