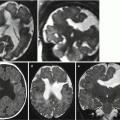
Fig. 8.1
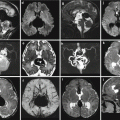
Fetal infection, unspecific signs on fetal MR. Parvovirus infection 25 GW: placental thickening and oligohydramnios (a, b). Toxoplasmosis, 22 GW: ascites (arrow c). Parvovirus infection 23 GW, fetal hydrops (arrow d)
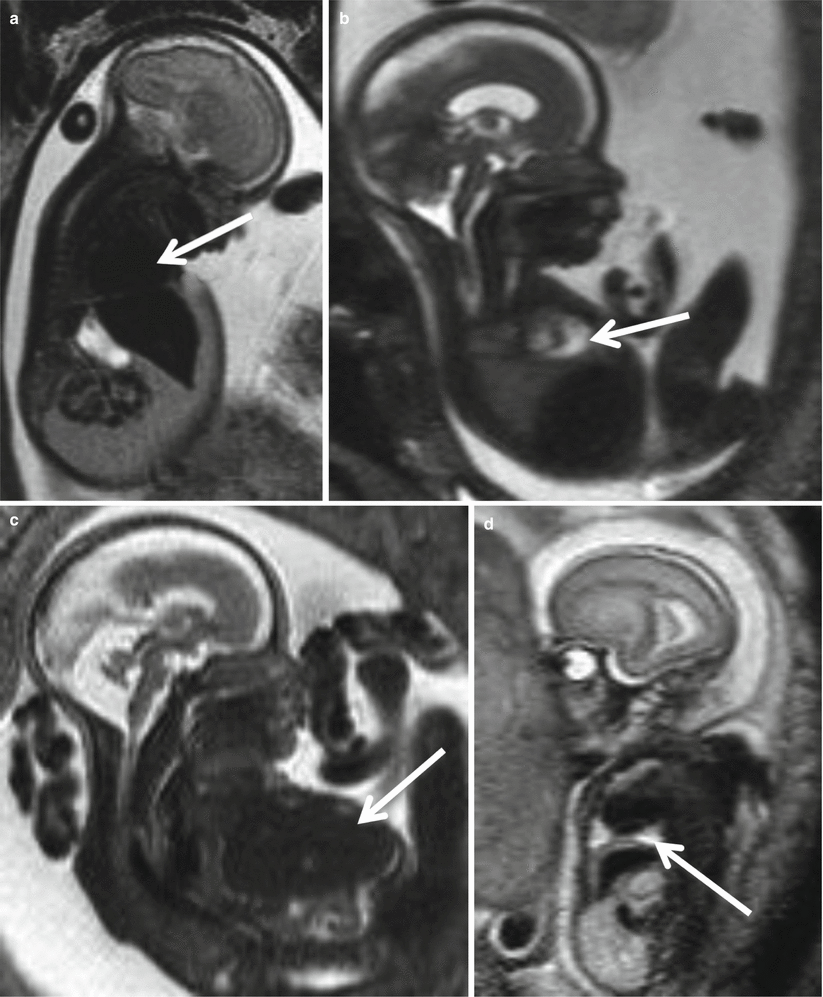
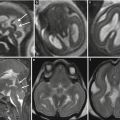
Fig. 8.2
Fetal infection, unspecific signs on fetal MR. Parvovirus infection 26 GW: cardiomegaly (arrow a) and pericardial effusion (arrow b). CMV infection 23 GW: hepatomegaly (arrow c). Parvovirus infection 23 GW: pleural effusion (arrow d)
8.1 Cytomegalovirus Infection
Cytomegalovirus is the most frequent cause of congenital infection and it occurs in approximately 1 % of newborns [3]. In the Buzzi Children’s Hospital series of 3147 fetal MR studies collected from 2001 to 2014 (unpublished data), a proven CMV infection was found in 105 cases, whereas toxoplasmosis in 12, rubella in 2, and parvovirus infection in 7.
Approximately 30–40 % of infected mothers transmit the virus to the fetus. In most cases, the infection is asymptomatic, but 10–15 % of asymptomatic cases at birth and two thirds of neonates with symptomatic infection develop neurological sequelae [3, 4] such as mental retardation, epilepsy, motor deficits, and deafness. The clinical course is generally described as static; however, disease progression has been demonstrated in some cases [5]. The infection can be diagnosed by PCR performed on amniotic fluid, fetal blood, or, in the case of the newborn, by urine analysis or PCR of cerebrospinal fluid or blood.
The infection results in different neuroradiological patterns according to the stage of gestation [1, 6]:
(a)
Before GW 16–18 weeks of gestation: microcephaly, abnormalities of cortical development, severe cerebellar hypoplasia, abnormal cerebral mantle layering, marked ventriculomegaly, and disseminated periventricular calcifications (Figs. 8.3, 8.4, and 8.5).
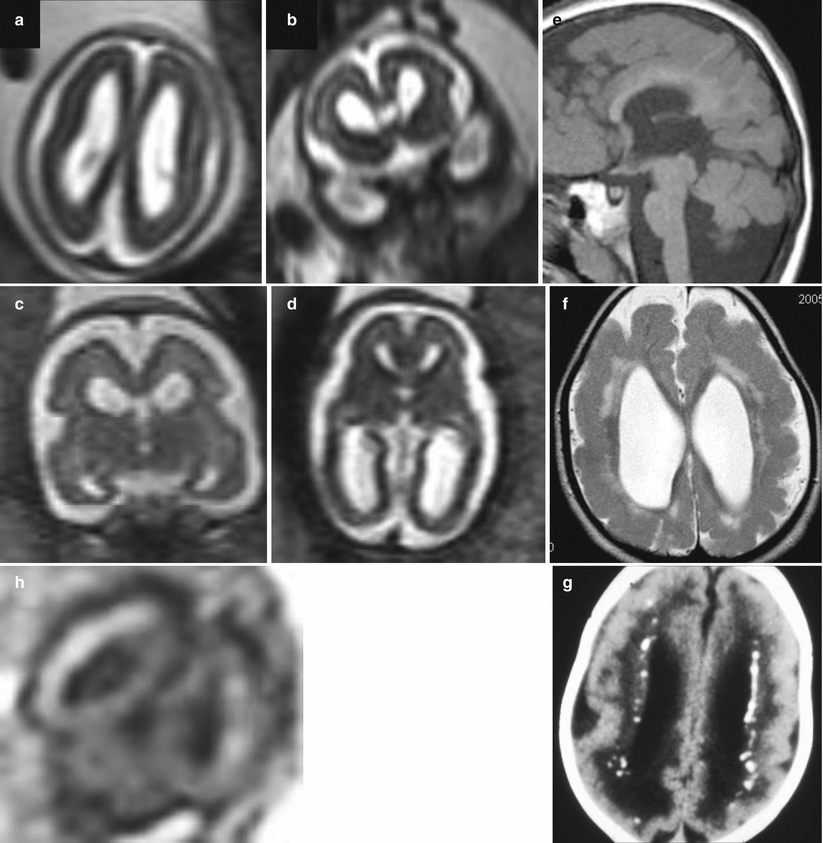
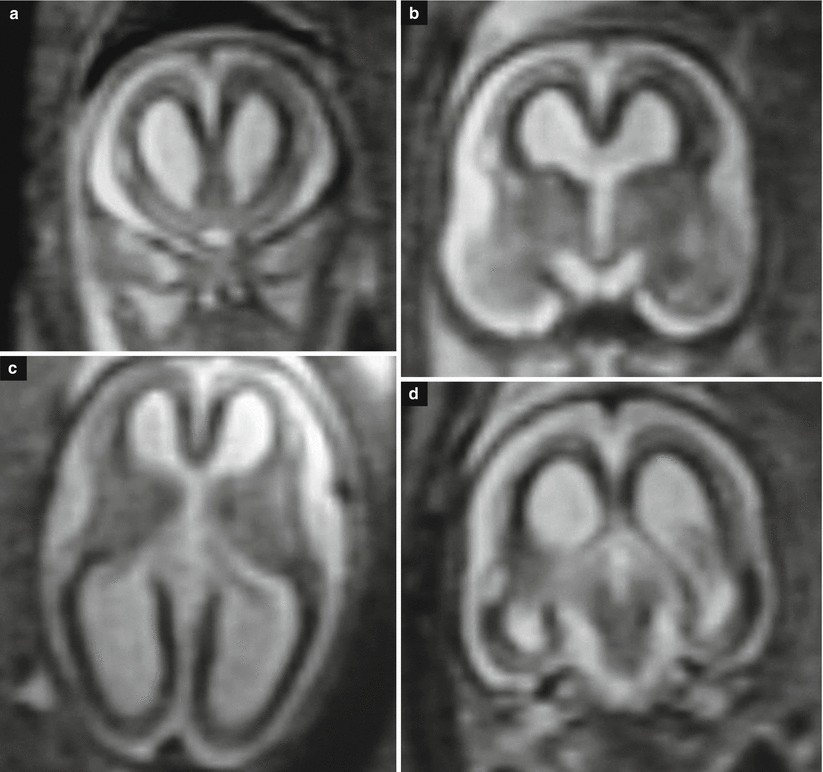
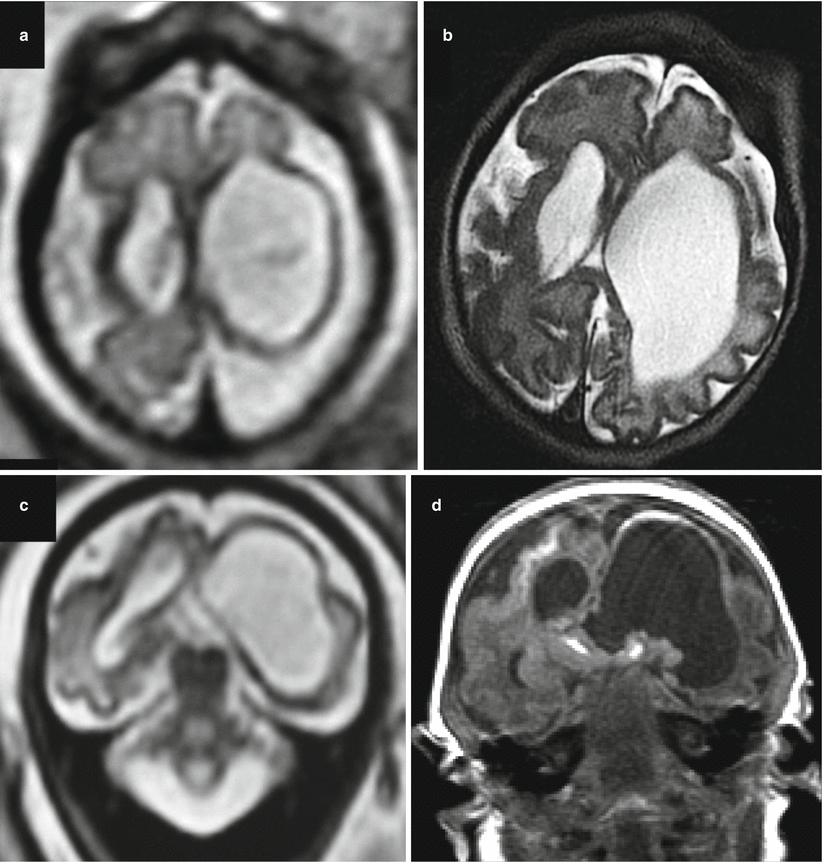

Fig. 8.3
CMV early gestation infection (before 16 GW). Fetal MR at 21 GW (a, b) and at 25 GW (c, d, h) in two cases of early fetal CMV infection shows microcephaly, abnormalities of cortical development, ventriculomegaly, and cortical subcortical T1-weighted hyperintensities (h), compatible with disseminated calcifications. Two different cases after birth show microcephaly with dysplastic cortex due to probable diffuse polymicrogyria (f, g), cerebellar hypoplasia (e), and calcifications (g)

Fig. 8.4
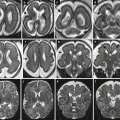
CMV early gestation infection (before 16 GW). Fetal MR study at 22 GW (a, b, d, coronal T2-weighted images; c, axial T2-weighted image) shows microcephaly, with diffuse enlargement of lateral ventricles and parenchymal thinning; subcortical regions are diffusely hypointense probably due to diffuse microcalcifications

Fig. 8.5
CMV early gestation infection (before 16 GW). Fetal MR study at 30 GW (a, c) and neonatal MR study (b, d) demonstrate severe microcephaly with an ex-vacuo enlargement of lateral ventricles, polymicrogyria is present along the right silvian region, calcifications (hyperintense areas on T1-weighted image) are detectable on the left ventricular margin, basal ganglia, and right subcortical regions
(b)
Between GW 18 and 24: microcephaly/microencephaly, abnormalities of cortical development (polymicrogyria, usually bilateral), mild-to-moderate cerebellar hypoplasia, abnormal white matter hyperintensity on T2-weighted images typically localized at the temporal pole, moderate ventriculomegaly particularly pronounced at the temporal horns, irregularities of ependymal margins, and possible periventricular calcifications (Fig. 8.6).
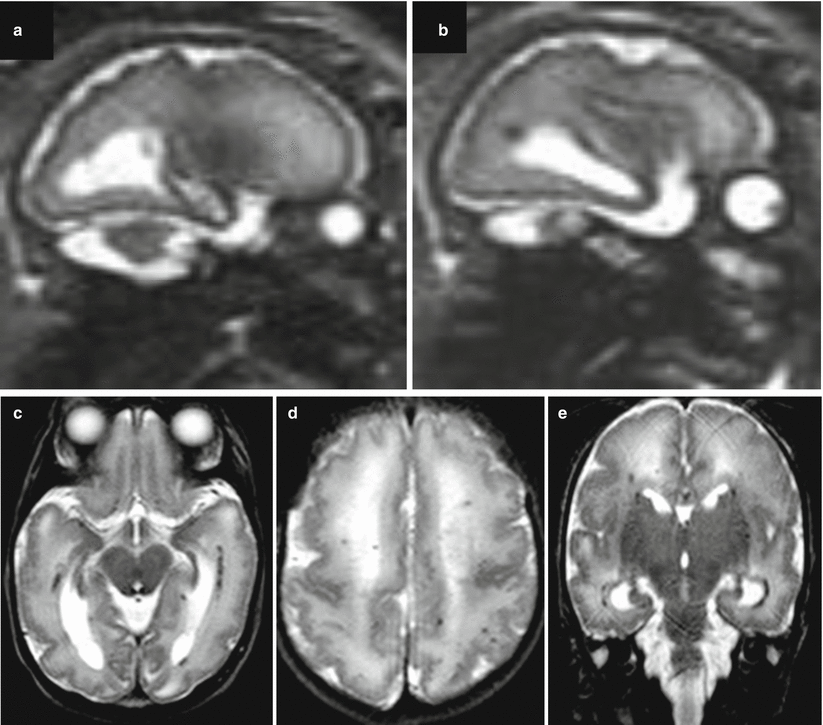

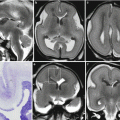
Fig. 8.6
CMV midgestation infection (between 18 and 24 GW). Same case of Fig. 4.18, Chapter 4. Diffuse polymicrogyria is evident on both prenatal (a, b) and postnatal MR studies (c, d, e). On postnatal MR study diffuse patchy white matter T2 hyperintensities are evident as well
(c)
Beyond GW 24: cortical anomalies may be usually absent, while temporal pole white matter lesions may occur, usually in association with focal enlargement of temporal horns. Mild ventriculomegaly, ependymal irregularities, and rare calcification can also be observed (Figs. 8.7 and 8.2).
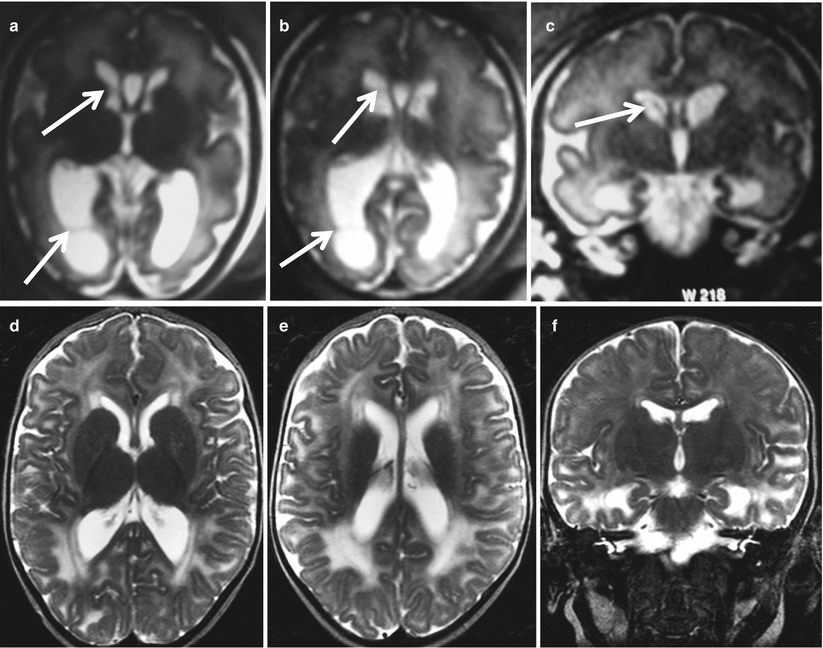

Fig. 8.7
CMV late gestation infection (beyond 24 GW). Fetal MR study at 33 GW (a–c) shows mild ventriculomegaly with intraventricular septa within the occipital and frontal horns (arrows). MR study after birth (d–f) exhibits patchy white matter hyperintensities on T2-weighted images without evidence of cortical malformations
Abnormalities of the temporal polar regions are highly suggestive of congenital CMV infection [3] (Figs. 8.8 and 8.9). The MR imaging pattern of congenital CMV after birth could partly resemble the pattern of the congenital muscular dystrophies, in which a combination of cortical abnormalities and white matter lesions can be seen. However, in congenital muscular dystrophies, hypotonia and muscle weakness are very often present and patients show other abnormalities on MRI, particularly of the brainstem and cerebellum, which usually allow the correct diagnosis. When cortical malformations are absent and white matter abnormalities are the predominant MRI features, the static course of CMV-related encephalopathy and the unrevealing metabolic screening allow to rule out classic leukodystrophies and some inherited disorders such as Aicardi– Goutières syndrome and pseudo-TORCH syndrome, which have a progressive course [3, 7].
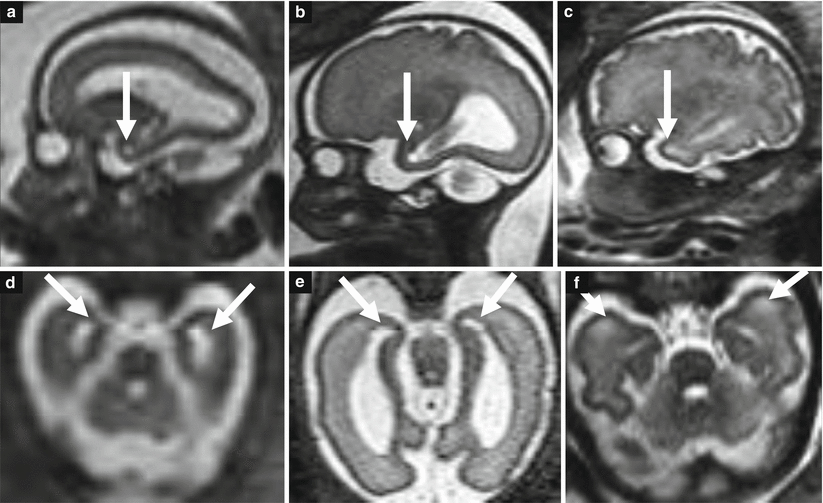
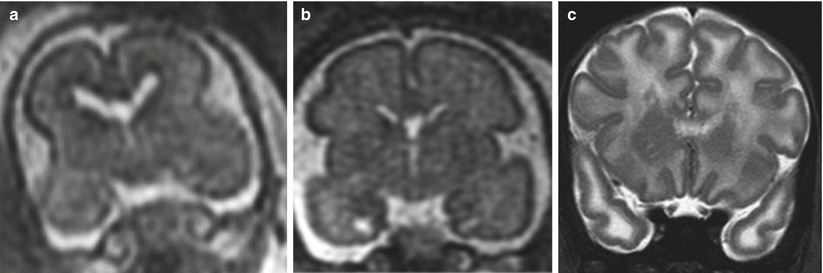

Fig. 8.8
CMV temporal pole lesions. Fetal MR studies at 25 GW (a, d), 30 GW (b, e), and 33 GW (c, f) demonstrate the evolution of temporal pole lesions (arrows) characterized by a slight diffuse hyperintensity of the subcortical white matter (c, f) usually associated with small cysts at the end of temporal horns of the lateral ventricles (a, b, d, e)

Fig. 8.9
CMV temporal pole lesions. Fetal MR study at 20 GW (a) and 27 GW (b) and postnatal MR study (c) of a case of fetal infection of CMV. No apparent lesions at the level of the temporal poles are evident at 20 GW, whereas small cysts are appreciable at 27 GW. On postnatal MR study, the subcortical temporal white matter is diffusely hyperintense
Cytomegalovirus infection-related cerebral abnormalities are well identified by MRI, with the exception of calcifications, which are better appreciated on computed tomography (Fig. 8.3) [3, 8–10]. US is able to identify areas of parenchymal hyperechogenicity, ventricular septa, periventricular necrosis, and calcification, as well as ventriculomegaly [11]. Recent efforts are directed toward the early identification of brain lesions in prenatal life. US can detect some CMV infection-associated brain anomalies, but it is substantially influenced by technical and anatomical factors. Fetal MRI is reported to provide substantial additional information, since it is able to identify almost all CMV-related lesions, even at an early gestational age, including major abnormalities such as microencephaly (sometimes missed by US if not associated with microcephaly) and cortical anomalies, with important implications in prognostic evaluation and pregnancy management [12]. Fetal MRI can also identify temporal pole lesions and their evolution throughout gestation: dilatation of temporal horns is the first anomaly followed by white matter T2-weighted hyperintensity and cystic lesions (Figs. 8.8 and 8.9).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree