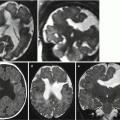
Fig. 6.1
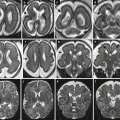
Dural sinus malformation. Fetal MR study at 26 GW (a–c) shows a mass at the level of the torcular (arrows) with a diffuse hyperintense aspect on T1-weighted image (b), compatible with subacute thrombosis within a DSM. After birth, the MR (d, f) and MRA (e) studies reveal only a minimal residual dilatation of the torcular region
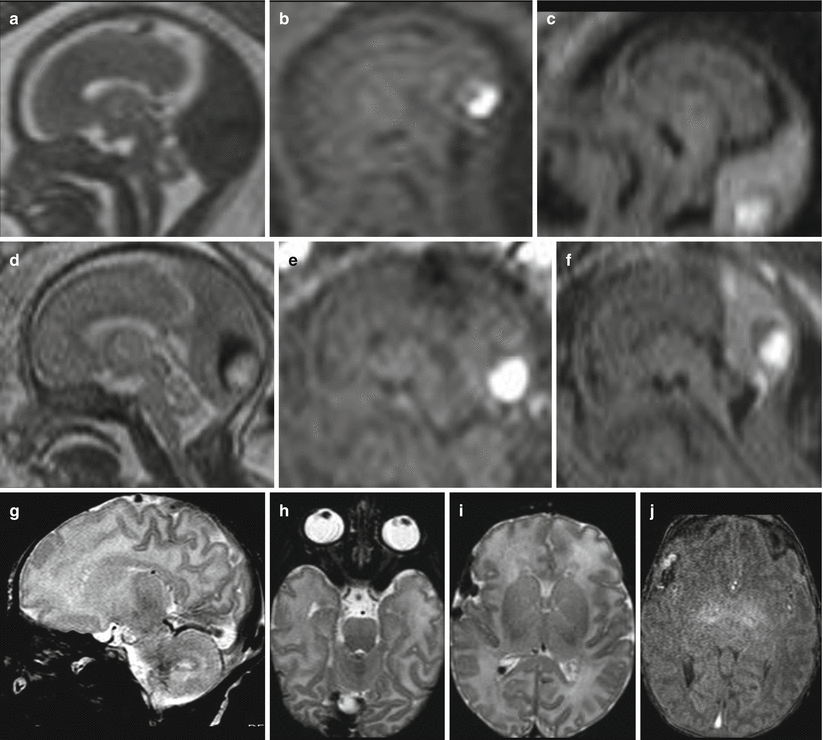
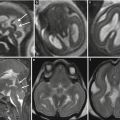
Fig. 6.2
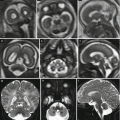
Dural sinus malformation. Fetal MR studies at 21 (a–c) and 24 GW (d–f) show a large DSM with a progressive thrombosis well detected on T1-weighted images. At birth (g–j), an almost complete resolution of the dural sinus enlargement is evident; a dilated vein (i, j) is as well evident in the right anterior silvian region
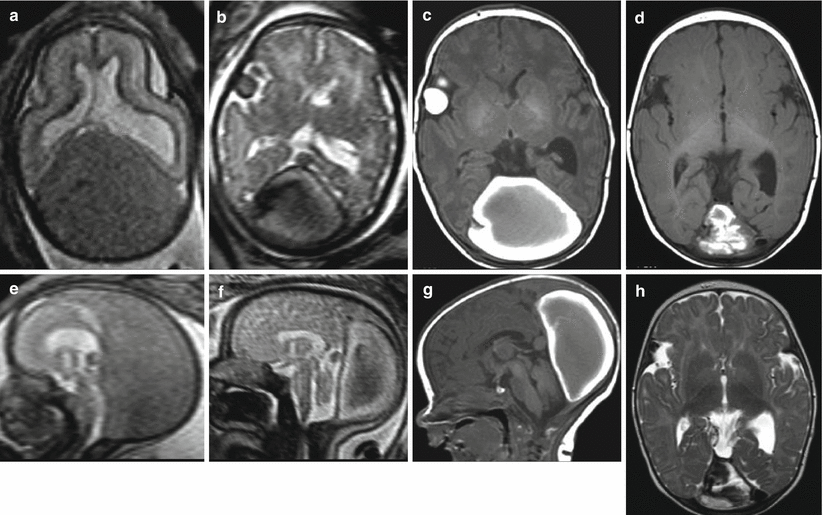
Fig. 6.3
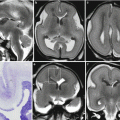
Dural sinus malformation. Fetal MR study at 23 (a, e) demonstrates a huge DSM with an important mass effect on the brain parenchyma and ventricular dilatation. Other three fetal MR follow-up studies were performed, and the last at 37 GW (b, f) shows a diffuse thrombosis of the malformation with a reduction of the mass effect. After birth, the MR study at one day of life (c, g) confirms the diffuse thrombosis that involves also a dilated vein in the right anterior silvian region (similarly to the case of Fig. 6.2). Postnatal MR follow-up at 6 months (d, h) shows a marked reduction in size of the malformation, with a residual thrombus within the torcular
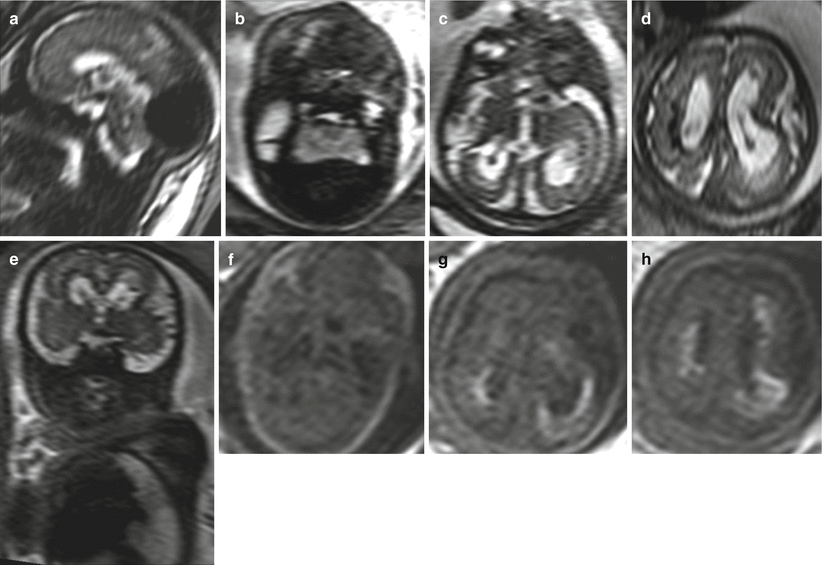
Fig. 6.4
Dural sinus malformation with diffuse brain lesions. Fetal MR study at 32 GW, after a suspected vein of Galen aneurismal malformation on US, shows a DSM (a, b) and diffuse brain parenchymal lesions (c, d) with malacic and necrotic aspect, partly hyperintense on T1-weighted images (f–h). Cardiomegaly is detected as well (e)
This giant pouch may be accompanied by multiple slow flow arteriovenous shunting within the wall of the malformed sinus, likely secondary to the hypertension caused by the presence of the dural sinus malformation [3]. Some authors suggest that AVF may not be simply a passive consequence of venous sinus enlargement, but may substantially contribute to the dural sinus malformation [5].
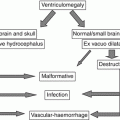
Several hypotheses have been proposed to explain the genesis of DSMs. They could correspond to the abnormal persistence of the sinus ballooning that physiologically occurs prenatally between the fourth and sixth months of gestation [4, 11]. However, this does not explain why the so-called normal ballooning is not seen antenatally in normal cases. A second hypothesis considers the possibility of an excessive and anarchic sinus development perhaps related to a local disturbing trigger. DSM is indeed associated with an uncontrolled development of posterior sinuses including transverse, sigmoid sinus, and/or confluence of sinuses. Hence, DSM is a disease of the sinus development.
Spontaneous thrombosis of the giant venous pouch can occur and is known to represent a secondary instead of causative phenomenon [7] (Figs. 6.1 and 6.2). Local mechanisms have been proposed to explain the thrombosis, such as an immaturity of the sinus, disturbances of the flow, and modification of the endothelium [6]. Early and rapid spontaneous thrombosis of the DSM lake, together with the immaturity of the jugular outlets, further compromise cerebral venous drainage and may lead to venous infarction (Fig. 6.4). These added constraints to the normal brain venous drainage will persist until cavernous sinuses maturation will provide an alternate outlet of venous cerebral flow, toward the ophthalmic veins or pterygoid venous plexuses.
The clinical course of live birth cases with thrombosed DSM is variable, ranging from death and neurological complications to normal neural development.
The prognosis of patients with DSM is still debated. Although it has been initially considered to be poor [1, 3], some recent reports, including cases studied during prenatal life, have highlighted a favorable outcome [6, 10]. The main predictor of favorable outcome is the presence of a sufficient collateral venous drainage of the brain with numerous anastomoses between the venous structures, permitting the venous flow to be redirected. Absence of brain damage, intraventricular hemorrhage, and fetal cardiac failure also are favorable prognostic criteria. In contrast, the involvement of the torcular does not appear as a negative marker as was suggested in previous series [1, 3]. The increased use of prenatal MRI has allowed a better understanding of DSMs, highlighting a good prognosis of this pathology that may spontaneously regress often without sequelae.
Prenatal ultrasound (US) diagnosis of DSM has been described as early as 20 weeks. The initial US pattern of DSM is a hypoechoic cystic heterogeneous lesion, which has to be further investigated, as color Doppler imaging may fail to detect flow in the lumen because of low velocity [9].
MRI is the best tool to diagnose a suspected DSM. Antenatal MRI identifies a T1 and T2 hypointense or isointense lesion with characteristic dural location, centered at the torcular region (Figs. 6.1, 6.2, and 6.3). The lesion can be large with a mass effect on brain parenchyma and over all on the cerebellum (Fig. 6.3). Ventricular dilatation (Fig. 6.3) and parenchyma focal alterations may be evident as consequence of venous hypertension. When the DSM is complicated by thrombosis, it may demonstrate a heterogeneous pattern on the T1-weighted images, including hyperintense areas (Figs. 6.1 and 6.2). Associated shunts are usually diagnosed postnatally, but often other ectatic veins are seen. It is mandatory to follow cases of DSM during the pregnancy, to verify the spontaneous modifications of the malformation and to rule out cerebral complications. Postnatal MRI, including magnetic susceptibility-weighted sequences and venous and arterial MR angiography, has to be performed rapidly after birth to evaluate the arteriovenous shunts and the brain parenchyma [5].
Conventional angiography should be considered in case of complications secondary to venous hypertension or cardiac failure, as a prelude to the endovascular treatment [6]. In patients with DSM, the goal of treatment is to preserve appropriate venous drainage to the brain while occluding the mural arteriovenous shunts, which can be achieved by means of staged sessions of transarterial embolization [1, 2, 8].
6.2 Vein of Galen Aneurysmal Malformation
The vein of Galen aneurysmal malformation (VGAM) is a congenital vascular malformation that comprises 30 % of the pediatric vascular and 1 % of all pediatric congenital anomalies [12–15]. VGAM is rare, but is the most common intracranial AV shunt presenting in the neonatal period. Abnormal development causes arteriovenous shunts between the primitive choroidal circulation and the median prosencephalic vein (MProsV) of Markowski, a precursor of the vein of Galen [16]. The presence of these shunts keeps the MProsV patent and promotes its enlargement, which forms the VGAM. It also prevents the normal formation of the vein of Galen. The VGAM drains only the choroidal system and does not connect with the deep venous system.
A VGAM is a specific choroidal malformation that is different from a cerebral AVM draining into a dilated but not malformed vein of Galen and is different from a dural arteriovenous fistula that develops in the wall of the vein of Galen [17].
The malformation is thought to develop between the 6th and 11th GW, after development of the circle of Willis, but a prenatal diagnosis has never been reported before the 20th week of gestation [19].
Generally, two types of angioarchitecture are described in VGAMs: choroidal and mural. The choroidal type corresponds with a primitive condition involving all of the choroidal arteries and an interposed network before opening into the large venous pouch. This condition is encountered in most neonates with severe symptoms. The mural type corresponds to direct arteriovenous fistulas within the wall of the MProsV. The fistulas may be single or, more often, multiple. The mural form is better tolerated clinically than the choroidal form [18].
The venous drainage of the choroidal shunt is always via a median prosencephalic vein and has no communication with the deep venous system. The VGAM drains into the straight sinus and uses the posterior sinuses as a route of drainage. This implies that the deep cerebral venous system is drained by alternative pathways such as the thalamic, subtemporal or latero-mesencephalic veins, and the cavernous sinuses. Only after birth do the cavernous sinuses mature, offering in patients who have a VGAM an alternative route of venous cerebral drainage through the orbits, pterygoid plexuses, or inferior petrosal sinuses. Maturation of the jugular bulbs and of the posterior sinuses may be disturbed by the presence of high-flow shunts.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree