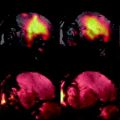
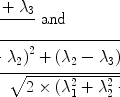([1])
where γ is the gyromagnetic ratio; ћ is Planck’s constant divided by 2π; P is the nuclear spin polarization. Therefore, it is clear that a higher spin concentration and a higher polarization of a sample give rise to a better signal. For a spin system with
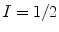 (I is the nuclear spin angular momentum), the polarization, P, is defined as
(I is the nuclear spin angular momentum), the polarization, P, is defined as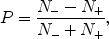
([2])
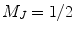 and
and 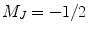 states, i.e., the lower and upper Zeeman energy sublevels, respectively.
states, i.e., the lower and upper Zeeman energy sublevels, respectively.For the conventional NMR or MRI, i.e., acquiring the nuclear magnetic resonance signal at thermal polarization, the nuclear spin polarization is quite small, and in the high-temperature approximation, it is given by (20)
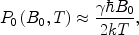
where k is Boltzmann’s constant; B 0 and T are the magnetic inductivity field strength and the temperature. For example, the proton thermal polarization is only 4 × 10−5 in a magnetic field of 11.7 T at 300 K. It requires the sample’s spin density high enough to obtain an observable magnetic resonance signal. That is the reason why the conventional NMR and MRI are only available to liquid, solid samples, or biological tissues, in which the proton spin densities are about three orders of magnitude higher than that in the gas phase samples. Traditionally, increasing the magnetic field (B 0) and lowering temperature (T) are employed to improve the thermal polarization. However, it becomes extremely costly to go to higher and higher magnetic fields with superconducting magnets and the cryogenic temperatures needed for enhanced polarization are not practical for in vivo applications. In order to obtain an observable magnetization in the gas phase, hyperpolarization is needed in order to compensate for the lack of spin density.
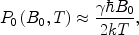
([3])
A number of methods have been developed to enhance the polarization of samples beyond thermal equilibrium including dynamic nuclear polarization (DNP) (21–26) (see Chapter 11), parahydrogen-induced polarization (27–29) (see Chapter 11), and, in the case of noble gases, spin exchange optical pumping (SEOP). Currently, the spin polarizations of 3He and 129Xe can be routinely increased up to 35–40% (30, 31) and 50–64% (32–34) for liter quantities, respectively. It should be noted that the spin polarization of 3He can also been enhanced by another technology called the metastability exchange (35–39). Since SEOP can be used for hyperpolarizing both 3He and 129Xe gases, in this chapter, we will focus on the technology of SEOP.
1.2 Hyperpolarized 3He and 129Xe
3He and 129Xe are the nuclei normally selected for hyperpolarized noble gases MRI for lungs, but in principle, any nucleus with non-zero spin (like 1H, 2H, 3He, 13C, 14N, 15N, 19F, 31P, 129Xe, 131Xe…) can be polarized above thermal equilibrium. However, the longitudinal relaxation rate of these nuclei can be rapid, causing significant decay of the spin polarization. The monatomic structure of noble gases, such as 3He, 19Ne, 21Ne, 37Ar, 83Kr, 129Xe, and 131Xe, enable long polarization half-lives compared to other nuclei. Among the noble gas isotopes, 3He and 129Xe are the only nuclei with nuclear spin 1/2, resulting in the T 1 (longitudinal relaxation time) of many hours or even days at the standard temperature and pressure. Hyperpolarized 83Kr (I = 9/2) has also been reported as a third noble gas probe for lung imaging, but quadrupolar interaction and the presence of a breathable concentration of molecular oxygen heavily affect the T 1 (40). Therefore, hyperpolarized 3He and 129Xe have generally been used for the medical lung imaging so far.
For comparison, the basic physical properties of 3He and 129Xe are listed in Table 1. For direct imaging of gas spaces, like lung MRI, 3He has an advantage over 129Xe, because the gyromagnetic ratio of 3He is 2.8 times larger than that of 129Xe. However, the diffusion coefficient of 129Xe is 30 times lower than that of 3He, and the lower diffusion constant would result in less signal attenuation during high-resolution imaging. 3He and 129Xe cover the spectrum from insoluble to highly soluble. 3He is much less soluble than 129Xe due to its smaller size. Since xenon is lipophilic, it has a higher solubility in blood-, oil-, and lipid-containing tissues. 129Xe is also extremely sensitive to its environment, which results in a chemical shift greater than 200 ppm in the body and up to 7500 ppm in different chemical compounds (41). Due to the relatively high solubility and the large chemical shift of xenon in biological tissues, such as in the lungs and the brain (42, 43), dissolved phase hyperpolarized xenon MRI and molecular imaging have shown a great potential in the study of gas exchange in the lungs, brain perfusion, and potential detection of diseases with high sensitivity (44–54). Such solubility and chemical shift properties make 129Xe a great probe for a variety of biomedical and material science applications.
1.3 The Principle of Spin-Exchange Optical Pumping
For SEOP, the first step is the transfer of circularly polarized laser light into electronic spin polarization, i.e., optical pumping. Figure 1 illustrates the principle of optically pumping Rb. In principal, any alkali metal vapor can be optically pumped. Rb is commonly used since the corresponding pumping laser diode arrays (LDA) are routinely manufactured in high power configurations. When the Rb pumping cell is placed in an oven at a temperature up to 160°C, the Rb is vaporized. Since the Rb vapor experiences an externally applied magnetic field (25 G) produced by a pair of Helmholtz coils, the ground state 52S1/2 is split into two Zeeman sublevels,  and
and 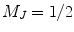 , i.e., electronic spin “down” or “up.” These two Zeeman states have a roughly equal population at room temperature. When the Rb vapor absorbs the circularly polarized laser light
, i.e., electronic spin “down” or “up.” These two Zeeman states have a roughly equal population at room temperature. When the Rb vapor absorbs the circularly polarized laser light 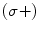 centered at 795 nm, a D1 transition of the Rb is observed, i.e.,
centered at 795 nm, a D1 transition of the Rb is observed, i.e., 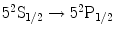 . The circularly polarized light is right handed (σ+), and therefore only the ground state with a sublevel
. The circularly polarized light is right handed (σ+), and therefore only the ground state with a sublevel 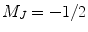 is pumped into the excited state,
is pumped into the excited state, 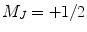 . Once the valence electron has been optically pumped to the excited state
. Once the valence electron has been optically pumped to the excited state 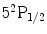 , collisional mixing equalizes the excited state populations. N2 gas molecules have many degrees of freedom in terms of rotational and vibrational levels. Thus, nitrogen gas will quench the two sublevels of the excited state back to the ground state. As the
, collisional mixing equalizes the excited state populations. N2 gas molecules have many degrees of freedom in terms of rotational and vibrational levels. Thus, nitrogen gas will quench the two sublevels of the excited state back to the ground state. As the 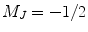 sublevel continues to absorb the circularly polarized light
sublevel continues to absorb the circularly polarized light 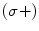 an excess of Rb atoms are optically pumped into the Zeeman sublevel
an excess of Rb atoms are optically pumped into the Zeeman sublevel 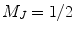 while the other sublevel
while the other sublevel 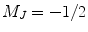 is depleted. Therefore, an electronic spin polarization of nearly 100% can be achieved for Rb.
is depleted. Therefore, an electronic spin polarization of nearly 100% can be achieved for Rb.
 and
and  , i.e., electronic spin “down” or “up.” These two Zeeman states have a roughly equal population at room temperature. When the Rb vapor absorbs the circularly polarized laser light
, i.e., electronic spin “down” or “up.” These two Zeeman states have a roughly equal population at room temperature. When the Rb vapor absorbs the circularly polarized laser light  centered at 795 nm, a D1 transition of the Rb is observed, i.e.,
centered at 795 nm, a D1 transition of the Rb is observed, i.e.,  . The circularly polarized light is right handed (σ+), and therefore only the ground state with a sublevel
. The circularly polarized light is right handed (σ+), and therefore only the ground state with a sublevel 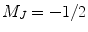 is pumped into the excited state,
is pumped into the excited state, 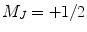 . Once the valence electron has been optically pumped to the excited state
. Once the valence electron has been optically pumped to the excited state 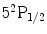 , collisional mixing equalizes the excited state populations. N2 gas molecules have many degrees of freedom in terms of rotational and vibrational levels. Thus, nitrogen gas will quench the two sublevels of the excited state back to the ground state. As the
, collisional mixing equalizes the excited state populations. N2 gas molecules have many degrees of freedom in terms of rotational and vibrational levels. Thus, nitrogen gas will quench the two sublevels of the excited state back to the ground state. As the 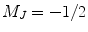 sublevel continues to absorb the circularly polarized light
sublevel continues to absorb the circularly polarized light 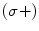 an excess of Rb atoms are optically pumped into the Zeeman sublevel
an excess of Rb atoms are optically pumped into the Zeeman sublevel 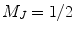 while the other sublevel
while the other sublevel 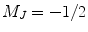 is depleted. Therefore, an electronic spin polarization of nearly 100% can be achieved for Rb.
is depleted. Therefore, an electronic spin polarization of nearly 100% can be achieved for Rb.
Fig. 1.
Schematic diagram of optical pumping of Rb.
The second step of SEOP is spin exchange between the polarized electronic spin of Rb and the xenon nucleus, as shown in Fig. 2. In the high-temperature and high-pressure optical pumping cell, the collision between polarized Rb atoms and xenon atoms results in the transfer of angular momentum from the electronic spin to the Xe nuclear spin. During this collision, the electron wave function of the Rb overlaps the nuclear wave function of xenon which causes spin exchange between the electronic spin and nuclear spin. Binary collisions dominate the spin exchange at high pressure, while three-body collision (by forming a Rb/Xe van der Waals molecule) dominate at low pressure (a few tens of torr) (5).
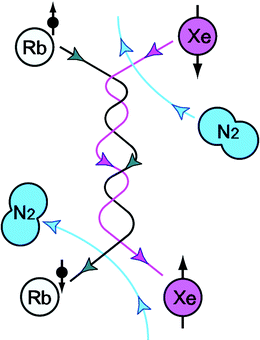

Fig. 2.
Schematic diagram of spin exchange between the Rb and Xe. (Courtesy of Brian Patton. Reprinted from Ref. (9) with permission.)
Generally, the polarization build-up time for 3He is much longer than that of 129Xe, and accordingly, the spin-exchange optical pumping for 3He is normally done in a batch mode, while hyperpolarized 129Xe can be produced in a continuous flow mode. A hybrid optical pumping technique has been developed for 3He to shorten the time needed to polarize the gas (55). In this chapter, we will focus on the building of a xenon polarizer in a continuous flow mode.
2 Materials
2.1 Non-magnetic Platform
1.
Aluminum alloy frame.
2.
Non-magnetic wheels.
3.
Stable optical platform for optical support.
2.2 Vacuum Pipeline System
1.
High performance vacuum system (Pfeiffer Vacuum HiPace 400 turbopump).
2.
Vacuum gauge.
3.
Copper and Teflon gas tubing, valves, fittings (Swagelok, Fremont, CA).
4.
Pyrex glass tubes for fabrication of optical pumping cell.
5.
Pyrex glass valves and fittings (Chemglass, Vineland, NJ).
6.
Glass coating fluid (Surfasil, Thermo scientific, Rockford, IL).
7.
Oxygen trap (with option to be regenerated (NuPure) or to be replaced (Agilent))
8.
Cold finger.
2.3 Optical Pumping System
1.
Litz wire for Helmholtz coils or 40 cm premade Helmholtz coils from Phywe Systeme GmbH & Co KG, Germany.
2.
High stability DC power supply (0–60 V).
3.
Laser safety glasses designed for a wavelength of 795 nm.
4.
60 W high power tunable laser system with a built-in air-cooled chiller; the central wavelength of laser is 795 nm (Coherent Duo FAP system, Santa Clara, CA).
5.
Beam expander, beam splitter (BS), reflector, and two quarter-wave plates with a central wavelength of 795 nm (Coherent, Santa Clara, CA).
6.
Optical spectrometer (Ocean Optics USB 2000+, Dunedin, FL).
7.
Personal computer (PC).
2.4 Flow Controlling System
1.
Mass flow controller (Aalborg DFC-26S, Orangeburg, NY).
2.
Mass flowmeter (Aalborg GFM17A Orangeburg, NY).
3.
Power supply (+15 V, −15 V) and accessories.
4.
Pressure transducer (Swagelok, Oakland, CA).
5.
Data Acquisition (DAQ) board (National Instruments PC6251, Austin, TX).
6.
DAQ software (National Instruments LabVIEW, Austin, TX).
2.5 Temperature Controlling System
1.
Insulated oven box (home built).
2.
Air heater, thermocouple, and accessories (Chromalox, Pittsburgh, PA).
3.
Flow shutoff safety (Dwyer, Michigan City, IN).
4.
Temperature controller (Chromalox, Pittsburgh, PA).
2.6 Cryogenics and Storage
1.
Ice water.
2.
Liquid nitrogen and dewar.
3.
Permanent magnet (2500 G).
4.
Cold finger.
5.
Non-magnetic lab jack.
2.7 Gases and Alkali Metal
1.
Xenon gas mixture (1% Xe, 1–10% N2, balanced He) (see Notes 1(d) and 2(a)).
2.
Rubidium ingot, ca. 1 g (Sigma Aldrich, St. Louis, MO).
3.
High pressure air (70 psi).
4.
High purified nitrogen gas (25–30 psi).
5.
Heat gun.
6.
Glass Pasteur pipettes.
2.8 Components for In Vivo Delivery
1.
Tedlar bag (Jensen Inert, Coral Springs, FL).
2.
Hot water.
3.
Air ventilator for inspiration.
4.
Pneumatic valves.
5.
High pressure helium gas (70 psi).
3 Methods
3.1 Xenon Polarizer Platform Fabrication
1.
Using aluminum alloy materials and non-magnetic wheels, fabricate a platform for the whole xenon polarizer system, which can be easily moved.
2.
The optical system should then be mounted securely on the xenon polarizer platform to minimize the displacement of the laser optics.
3.2 Pipeline System and Vacuum
1.
Assemble a pipeline system using copper and Teflon tubing, valves, and fittings according to the schematic in Fig. 3.
2.
Using Pyrex glass tubes, valves, and fittings, fabricate a cylindrical optical pumping cell, in which the laser beam can illuminate it along the cylindrical axis.
3.
The optical pumping cell needs to be washed three times using distilled water and acetone, and then baked in an oven with a temperature of 80°C for more than 24 h.
4.
Next, dilute SurfaSil siliconizing fluid in acetone to a concentration of 10% by volume.
5.
Flood the optical pumping cell in the diluted SurfaSil solution for at least 10 s.
6.
Agitate the solution to ensure a uniform coating, then rinse the object with the acetone and methanol and bake it in an oven at a temperature of 100°C for 1 h.
7.




Connect the optical pumping cell, vacuum system, vacuum gauge, and oxygen trap to the pipeline system, and then test the vacuum to make sure it is able to reach a vacuum of at least 10 millitorr (see Notes 3(e), 4(d)/(e), and 1(a)/(b)).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree