Fig. 7.1
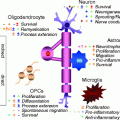
There are multiple considerations when designing the clinical translation of cell therapy. These include choosing the relevant therapeutic target(s) for the candidate disease, choosing the proper cellular platform, timing of transplantation, and route of delivery. For example, congenital dysmyelinating diseases and age-related macular degeneration require specific cell types for cell-replacement therapy (by myelin-forming cells and retinal pigment epithelium cells) and their direct delivery into the target tissue. Spinal cord injury may enjoy from the trophic effects of transplanted cells to enhance endogenous repair systems, as well as from their immunomodulatory and direct regenerative properties. Parkinson’s and Huntington’s diseases are considered as candidates mainly for cell replacement, but early therapy with neural precursors may also have protective effects against disease progression. For multiple sclerosis, all four mechanisms of transplanted cell action may be beneficial and may be achieved to certain extents by both neural and non-neural cell platforms
7.2.1 The Route of Cell Delivery
The route of cell administration represents a major constraint for stem cell transplantation and appears to be very dependent on the site of the lesion in the CNS (focal vs. multifocal). MS is indeed a multifocal disease, and it would be then very unlikely to propose lesion-targeted injection of cells (Pluchino and Martino 2008). Moreover, it is often difficult to determine which of the multiple foci observed in the brain by non-invasive magnetic resonance imaging (MRI) may underscore clinical significance and whether they would eventually be amenable to effective remyelination upon cell therapy (Chen et al. 2007). Therefore, the ability of transplanted cells to migrate into inflamed brain areas, integrate, and differentiate is a crucial requisite to test, thus further highlighting the identification of the optimal time window and route of cell delivery as two key issues for cell transplantation in MS.
Transplanted multipotent NPCs have been shown to migrate and integrate in the developing rodent CNS and adopt cellular identity according to local and temporal cues (Brustle et al. 1995, 1998; Flax et al. 1998). Furthermore, the transplantation of both somatic and embryonic stem (ES) cell-derived oligodendrocyte progenitors has led to widespread myelination in the genetic dysmyelinating shiverer (shi/shi) mouse (Windrem et al. 2004, 2008; Yandava et al. 1999) and the myelin-deficient (md) rat models (Brustle et al. 1999; Hammang et al. 1997; Zhang et al. 1998, 1999). While earlier work suggests that the normal adult brain does not allow neither long-distance migration nor does it support the survival of focally transplanted neural cells (O’Leary and Blakemore 1997), more recent data has provided evidence that in some model systems, long-stage migration and survival of oligodendrocyte progenitors can occur (Buchet et al. 2011). Consequently, cell migration might be a major limiting factor for both endogenous remyelinating cells and transplanted lineage-committed myelin-forming cells in MS (Blakemore et al. 2000; Franklin and Blakemore 1997; Gensert and Goldman 1997). These and other observations have led to two major approaches for the delivery of therapeutic cells through biological fluids accessing multiple CNS areas in experimental MS models: the intracerebroventricular (i.c.) or intrathecal (i.t.) cell transplantation and the intravenous (i.v.) cell injection.
Following i.c. or i.t. transplantation of either neurospheres or single cell-dissociated NPCs, clinico-pathological amelioration of EAE in rodents has been achieved, with transplanted cells exhibiting targeted migration almost exclusively to the inflamed periventricular and perivascular white, but not grey, matter of the forebrain (Ben-Hur et al. 2003a, b, 2007; Bulte et al. 2003; Pluchino et al. 2003; Politi et al. 2007). Using this latter approach, the majority of transplanted NPCs have differentiated in vivo into glial cells (e.g. 30 % oligodendrocytes, 25 % astrocytes) (Ben-Hur et al. 2003b; Pluchino et al. 2003). Most white matter tracts that are involved in EAE and MS are in close proximity to the ventricular and spinal subarachnoid spaces (Brok et al. 2001; Bruck 2005). Therefore, the main advantage of these specific routes of cell delivery would be that transplanted NPCs disseminate throughout the ventricular and subarachnoid spaces, enabling their inflammation-driven migration to the white matter.
Several mediators of inflammation have been implicated in regulating NPC migration. The chemokine stromal-derived-factor (SDF)-1 α/CXCL12 and its exclusive receptor CXCR4 are important regulators of the migration of dentate granule cells (Bagri et al. 2002; Lu et al. 2002), sensory neurons (Belmadani et al. 2005), and cortical interneurons (Stumm et al. 2003) during development. In agreement with the knowledge that profound recapitulation of developmental processes takes place in the adult CNS during reparative regeneration (Martino 2004), the developmental chemokines SDF-1 α and monocyte chemoattractant protein (MCP)-1/CCL2, and their respective receptors CXCR4 and CCR2, have modulated the migration of SVZ-resident endogenous NPCs following experimental cerebral ischemia (Imitola et al. 2004; Yan et al. 2007). Also, tumour necrosis factor (TNF) α (Ben-Hur et al. 2003a), hepatocyte growth factor (HGF), transforming growth factor (TGF) β, and Rantes/CCL5 have also been implicated in NPC and oligodendrocyte progenitor cell self-renewal and migration, respectively, both in vivo and ex vivo after NPC grafting into hippocampal slice cultures (Belmadani et al. 2006; Lalive et al. 2005; Nicoleau et al. 2009; Pluchino et al. 2005). Recent transplant data has shown that transplanted oligodendrocyte progenitors migrate preferentially towards focal spinal cord lesions in which TGFβ expression is increased (Wang et al. 2011).
The i.c. and i.t. cell delivery routes may therefore allow therapeutic cells to accumulate as close as needed to the multiple damaged/inflamed areas in EAE/MS, without (or with very little) separating anatomical barriers. Moreover, recent studies suggest the transition of immune pathogenesis in MS from a peripheral initiated inflammatory injury during early phase of disease to a CNS compartmentalized immune and degenerative process during the chronic phase of disease that is localized mainly in subarachnoid space/pial surface (Howell et al. 2011). Thus, i.c./i.t. delivery may target the disease process directly.
An alternative route of cell delivery that has been recently suggested is the direct injection of NPCs into the blood stream (Pluchino et al. 2003, 2005; Pluchino et al. 2005). The exact molecular mechanism sustaining the significant homing capacity of i.v.-injected NPCs has been detailed in rodents with EAE as dependent on the expression of functional cell adhesion molecules (e.g. CD44), integrins (e.g. α4, β1) (Campos et al. 2004, 2006; Leone et al. 2005; Pluchino et al. 2003, 2005), and chemokine receptors (e.g. CCR1, CCR2, CCR5, CXCR3, CXCR4) (Imitola et al. 2004; Ji et al. 2004; Pluchino et al. 2005) on NPC surface.
In addition to the ease of i.v. delivery, the main advantage of this latter protocol of NPC delivery is the mechanism of cell accumulation, initially described to target specifically the perivascular inflammatory foci in the CNS (Ben-Hur et al. 2003b; Pluchino et al. 2003, 2005). However, more recent evidence suggests that i.v.-injected NPCs may target and synergize with immune cells also outside the CNS, at the level of secondary lymphoid organs, such as the lymph nodes (Einstein et al. 2007) or the spleen (Lee et al. 2008), though this latter observation significantly supports the added therapeutic value transplantable NPCs capable of engaging multiple mechanisms of action within different CNS vs. non-CNS microenvironments in vivo (Einstein and Ben-Hur 2008; Martino and Pluchino 2006). It would also open new (still unsolved) questions about the overall safety and systemic effects of cell delivery routes allowing the dispersion of injected NPCs outside the CNS.
7.2.2 Tracking Transplanted Cells
To develop successful clinical cell-based therapies, it would be important to assess non-invasive monitoring of both cell fate and distribution. Nuclear medicine techniques, fluorescence imaging, and bioluminescence have been shown to provide valuable means for monitoring cell therapies in vivo. Positron emission tomography (PET) has been used in humans to evaluate viability and function of locally transplanted cells in the neurodegenerative disorders Parkinson’s and Huntington’s diseases (Brooks 2005; Mendez et al. 2005). Fluorescence imaging and bioluminescence have been successfully used to monitor focal stem cell transplantation in mice with cerebral infarcts (Kim et al. 2004). Unfortunately, all these techniques have shown poor spatial resolution, cannot be correlated to anatomical details, and have limited depth penetration (Jacobs and Cherry 2001). In contrast to these latter techniques, magnetic resonance imaging (MRI) is a more accurate means for cellular imaging (de Vries et al. 2005), allowing efficacious (cell) tracking over longer periods of time.
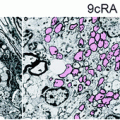
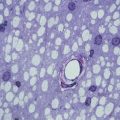
The recent development of super paramagnetic iron oxides (SPIOs) has further improved the sensitivity of cellular and molecular MRI (Modo et al. 2005). Magnetically labelled oligodendrocyte progenitors (Bulte et al. 1999; Franklin et al. 1999), neural progenitors (Ben-Hur et al. 2007; Bulte et al. 2001), and Schwann cells (Dunning et al. 2004) have been visualized with high magnetic field-strength, animal-dedicated MR scanners, when focally transplanted into preclinical rodent models of genetic, focal chemical, or inflammatory de/dysmyelination.
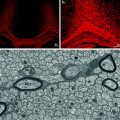
SPIO-labelled, bromodeoxyuridine (BrdU)-loaded NPCs have been transplanted i.c. into acute EAE in Lewis rats. Ex vivo MRI confirmed that while the transplant disseminated in the ventricular system of both naïve and EAE brains, widespread migration into white matter tracts occurred in EAE rats only. A good correlation was also found between the histological distribution of iron-labelled NPCs and the BrdU immunostaining, thus indicating that the magnetic label was retained by transplanted NPCs (and not transferred to neighbouring cells) in vivo (Bulte et al. 2003). Furthermore, proof of principle of the sensitive detection and quantification (e.g. down to 30 cells/region of interest) of i.v.-injected SPIO-labelled NPCs accumulating in the brain of mice with chronic EAE has also been provided by means of in vivo clinical-grade 3T MR imaging and ex vivo relaxometry (Politi et al. 2007). Also, serial MRI-based tracking of mouse adult and human ES cell-derived neural spheres showed specific (and early along the course of disease) migration of SPIO-labelled cells along white matter tracts after i.c. cell injection in chronic EAE mice (Ben-Hur et al. 2007), thus further suggesting that human ES-derived NPCs respond to inflammatory tissue signals similar to rodent cells, a prerequisite for their consideration as clinical vectors.
A recent report highlights that SPIO-labelled human adult mesenchymal stem cells (MSCs), implanted subcutaneously (s.c.) into nude mice, display preferential chondrogenic differentiation in vivo, as compared to implanted unlabeled MSCs (Farrell et al. 2008). On the other hand, SPIO labelling does not seem to change the migratory properties and lineage fate of NPCs both in vitro and in vivo (Cohen et al. 2010). Therefore, it would be necessary to evaluate whether contrast agent incorporation exerts any significant negative effects on the candidate cell population prior to use their in cell therapies.
More broadly though, these data suggest that the real-time MR monitoring of delivered cell therapeutics may become an important tool in evaluating the efficacy of transplant-based remyelination both experimentally, as well as in individual patients.
7.3 The Therapeutic Plasticity of NPCs
7.3.1 Cell Replacement
Although NPCs have been successfully transplanted in several preclinical models of neurological disorders, the remarkable functional recovery obtained upon NPC transplantation has generally showed little correlation with the absolute numbers of terminally differentiated neural cells originated by transplanted NPCs (see for review Martino and Pluchino 2006). As such, transplanted NPCs very scarcely differentiate into tyrosine hydroxylase (TH)-immunoreactive neurons, when transplanted in rodents with experimental Parkinson’s disease (PD) (Ben-Hur et al. 2004; Ourednik et al. 2002; Rafuse et al. 2005; Richardson et al. 2005) or Huntington’s disease (HD) (McBride et al. 2004; Ryu et al. 2004), despite significant behavioural improvement. Similarly, mice with spinal cord injury (SCI) (Fujiwara et al. 2004; Hofstetter et al. 2005), acute stroke (Chu et al. 2004), or intracerebral haemorrhage (Jeong et al. 2003; Lee et al. 2008) show clinical recovery regardless of transplanted NPCs acquiring preferential astroglial fate in vivo. Other evidence shows that NPCs injected into models of SCI do not differentiate at all into terminally differentiated neuronal cells (Cummings et al. 2005; Fujiwara et al. 2004; Lu et al. 2003; Teng et al. 2002). In EAE, very low differentiation of NPCs into myelin-forming oligodendrocytes is accompanied by neurophysiological evidence of axonal protection and remyelination (Ben-Hur et al. 2003b; Pluchino et al. 2003). In the very same context, much way over 20 % of transplanted cells reaching inflammatory demyelinated areas do not express differentiation markers (Pluchino et al. 2003). It is yet to be shown that transplanted cells can remyelinate efficiently in clinically relevant models of MS. This may depend both on grafting precursor cells that are already programmed to the oligodendroglial lineage (Ben-Hur et al. 1998; Hu et al. 2009) and the use of appropriate models of disease that allow remyelination to occur.
This scarce terminal differentiation and propensity to maintain an undifferentiated phenotype within the host tissue has suggested that transplanted NPCs might be therapeutic efficacious via a number of bystander mechanism(s) alternative to cell replacement.
7.3.2 (Tissue) Trophic Support
The poor repair capacity of the adult CNS results from a number of failing repair programmes, including the apparent inability of endogenous progenitors and stem cells to properly respond to disease states, to replace damaged cells and from lack of regenerative capacity of injured axons (Franklin 2002; Franklin and Ffrench-Constant 2008; Martino 2004; Pluchino et al. 2008).
NPC therapy is emerging as a mode of treatment that can enhance the host brain’s ability to repair itself in both aspects (Einstein and Ben-Hur 2008; Pluchino and Martino 2008). Recent studies have focused also on several bystander tissue-protective and -trophic properties of transplanted NPCs. NPCs seeded on a synthetic biodegradable scaffold and grafted into the cord of hemi-sectioned rats induced significant clinical recovery while reducing the necrosis of the surrounding parenchyma and preventing extensive secondary cell loss, inflammation, and glial scar formation (Teng et al. 2002). Moreover, the NPC graft induced a permissive environment for axonal regeneration (Teng et al. 2002). Substantial endogenous reconstitution of the brain structural connectivity has been found following injection of NPCs in biodegradable scaffolds into regions of extensive brain degeneration caused by hypoxia (Park et al. 2002a, b) or following i.c. transplantation of NPCs after ischemia/reperfusion injury in mice (Capone et al. 2007). Transplanted NPCs have rescued endogenous dopaminergic neurons of the mesostriatal system in a PD model in rodents (Ourednik et al. 2002), while prevention of death of motor neurons has been observed when NPCs were transplanted in models of amyotrophic lateral sclerosis (ALS) (Ferrer-Alcon et al. 2007; Kerr et al. 2003; Suzuki et al. 2007). NPC-driven bystander tissue protection has generally led to a reduction of glial scar formation and increase of survival and/or functions of endogenous glial and neuronal progenitors surviving to the pathological insult.
NPC trophic effects relate in part to increased in vivo bioavailability of major neurotrophins (e.g. nerve growth factor [NGF], brain-derived neurotrophic factor [BDNF], ciliary neurotrophic factor [CNTF], glial-derived neurotrophic factor [GDNF]) (Chu et al. 2004; Einstein et al. 2006; Lu et al. 2003; Pluchino et al. 2003; Teng et al. 2002 and others). The multiple roles of neurotrophins as mediators in cell cycle regulation, cell survival, and differentiation during development and adulthood make them potential candidates for the regulation of endogenous NPC proliferation and differentiation following brain injury and for modulation of host environment into more permissive for regeneration. For example, neurotrophins secreted by transplanted NPCs help promote corticospinal axon growth after transplantation onto an organotypic co-culture system, containing dissected brain cortex and spinal cord (Kamei et al. 2007). Moreover, several neurotrophins that may be released by NPCs were shown to inhibit EAE. Insulin-like growth factor (IGF)-1 and glial growth factor (GGF)-2 are neurotrophic factors that promote survival and proliferation in the oligodendrocyte lineage (Barres et al. 1992; Canoll et al. 1996, 1999; Mason et al. 2000). Treatment with these factors was beneficial clinically and pathologically in animals with EAE (Akassoglou et al. 1998; Cannella et al. 1998; Yao et al. 1996). Interestingly, their effect was mediated not only by enhancing oligodendrocyte survival (Butzkueven et al. 2002; Linker et al. 2002) but also by decreasing neuroinflammation (Cannella et al. 1998; Flugel et al. 2001; Ruffini et al. 2001; Villoslada et al. 2000).
The broad (tissue) trophic support by transplanted stem cells has been studied extensively in models of spinal cord injury and attributed to multiple mechanisms. These include production of neurotrophic growth factors that enhance axonal regeneration (Lu et al. 2003); induction of matrix metalloproteinases that degrade the extracellular matrix and cell surface molecules that impede axonal regeneration, thus enabling axons to extend through the glial scar (Zhang et al. 2007); induction of angiogenesis in the lesioned tissue, which provides trophic support and enables tissue repair (Rauch et al. 2009); providing proper realignment and guidance to enable axonal regeneration along long fibre tracts (Pfeifer et al. 2004); and increasing remyelination in the lesion by both endogenous and graft-derived myelin-forming cells to enhance action potential conduction and limit secondary axonal degeneration (Keirstead et al. 2005). In addition, NPC transplantation has been shown to facilitate endogenous myelin repair following chronic cuprizone-induced demyelination, by induction of resident OPC proliferation and differentiation (Einstein et al. 2009).
Recent work has also indicated that transplanted stem cells, including NPCs, can enhance endogenous neurogenesis in certain physiological and pathological conditions (Hattiangady et al. 2007; Munoz et al. 2005). Mice exposed prenatally to opioids display impaired learning associated with reduced neurogenesis, and transplantation of NPCs improves learning functions, as well as host brain-derived neurogenesis in the DG of the hippocampus (Ben-Shaanan et al. 2008). A similar neurotrophic effect was also reported in physiological ageing. While neurogenesis in the DG declines severely by middle age, transplantation of NPCs stimulates the endogenous NPCs in the SGZ to produce new dentate granule cells (Hattiangady et al. 2007).
Thus, transplanted NPCs may enhance the adult CNS capacity to repair itself by restoring the ability of endogenous progenitors and stem cells to both respond properly to disease state and replace damaged CNS cells and the ability of severed axons to regenerate. Yet, a current and comprehensive illustration of whether these and other (tissue)trophic properties of NPCs are also relevant to EAE and MS is partially lacking.
7.3.3 Immune Modulation
Important progress towards the application of stem cell therapies in MS has been made recently with the observation that NPC transplantation attenuates the clinical course of acute (Einstein et al. 2003), chronic (Einstein et al. 2006; Pluchino et al. 2003) and relapsing (Pluchino et al. 2005; Pluchino et al. 2009) EAE in rodents and primates (Pluchino et al. 2009a). While dealing with the expected regenerative potential of stem cell transplantation as direct cell-replacement therapy, recent work has highlighted peculiar additional immune modulatory mechanisms by which transplanted NPCs exhibit therapeutic effects in preclinical models of MS. The first indication of novel (anti-inflammatory?) effect of NPCs was obtained when neurospheres were transplanted i.c. in acute spinal cord homogenate (SCH)-induced EAE Lewis rats (Einstein et al. 2003). These EAE rats show acute, reversible paralytic disease that is the result of disseminated CNS inflammation without demyelination or axonal injury. NPC transplantation in SCH-induced EAE Lewis rats attenuated the inflammatory brain process and clinical severity of disease (Einstein et al. 2003).
Subsequent studies examined the effect of NPC transplantation, upon either i.c. or i.v. cell injection, in the myelin oligodendrocyte glycoprotein (MOG)35-55-induced EAE in C57BL/6 mice. In this model, there is an acute paralytic disease due to a T cell-mediated autoimmune process that causes severe axonal injury and demyelination. Subsequently, the mice remain with fixed neurologic sequel, the severity of which is correlated with the extent of axonal loss (Wujek et al. 2002). NPC transplantation in MOG35-55-induced EAE mice attenuated the inflammatory process, rescued the endogenous pool of oligodendrocyte progenitor cells, reduced acute and chronic axonal injury and demyelination, and improved the overall clinical and neurophysiological performance of the mice (Einstein et al. 2006; Pluchino et al. 2003). Recent evidence has now suggested that this latter phenomenon may be dependent on the capacity of transplanted NPCs to engage multiple mechanisms of action within specific microenvironments in vivo (Martino and Pluchino 2006). Among a wide range of potential therapeutic actions, and in addition to the cell replacement and (tissue) trophic capacities (Pluchino et al. 2003), remarkable immune modulatory capacities are described for transplanted NPCs within specific CNS (Einstein et al. 2003, 2006; Pluchino et al. 2003, 2005; Swanborg 2001) and non-CNS areas (Einstein et al. 2007). As such, considerable proof that NPC-mediated bystander immune regulation may take place both in the CNS, at the level of the atypical perivascular niches (Pluchino et al. 2005), as well as in secondary lymphoid organs, such as the lymph nodes (Einstein et al. 2007) or the spleen (Lee et al. 2008), has recently been provided. However, the exact mechanisms by which transplanted NPCs attenuate CNS vs. peripheral inflammation are not yet clear.
One school of thought has first suggested an immune suppressive effect, by which NPCs induce apoptosis of Th1—but not Th2—cells selectively, via the inflammation-driven up-regulation of membrane expression of functional death receptor ligands (e.g. FasL, TRAIL, Apo3L) on NPCs (Pluchino et al. 2005). Alternatively, it has been suggested that NPCs inhibit T cell activation and proliferation by a non-specific, bystander immune suppressive action (Einstein et al. 2007). This notion emerged from co-culture experiments that showed a striking inhibition of the activation and proliferation of EAE-derived, as well as naive T cells by NPCs, following stimulation by various stimuli (Einstein et al. 2003, 2007). The suppressive effect of NPCs on T cells was accompanied by a significant suppression of pro-inflammatory cytokines, such as interleukin (IL)-2, TNFα, and IFNγ (Einstein et al. 2007). Moreover, NPCs inhibited multiple inflammatory signals, as exemplified by attenuation of T cell receptor-IL2- and IL6-mediated immune cell activation and/or proliferation (Fainstein et al. 2008). The relevance of such NPC/T cell interaction was first suggested when NPCs i.v. injected prior to EAE disease onset (e.g. at 8 days after the immunization) were transiently found in peripheral lymphoid organs, where they interacted with T cells to reduce their encephalitogenicity (Einstein et al. 2007). In this protocol i.v. NPC injection at an early time point, transplanted cells did not cross the blood–brain barrier, and their entire effect was mediated by peripheral immune suppression, resulting in reduced immune cell infiltration into the CNS and consequently milder CNS damage.
Recent studies have started addressing the role of individual molecular candidates in regulating this novel immune modulatory (or regulatory) capacity of transplanted NPCs in EAE. As such, when injected s.c. in a setup of passive cell vaccination in EAE mice, NPCs have shown remarkable capacity to target and synergize with immune cells in secondary lymphoid organs (but not in the CNS), where they stably (e.g. for more than 2 months after cell injection) modify the perivascular microenvironment. Within this context, surviving NPCs have hindered the activation of myeloid dendritic cells (DC) via a bone morphogenetic protein (BMP)-4-dependent mechanism, which was completely reverted by the BMP antagonist Noggin (Pluchino et al. 2009b; Pluchino et al. 2009).
Indeed, such a broad immune regulatory capacity has more recently been shown also for human somatic (Pluchino et al. 2009a) as well as human ES-cell-derived NPCs (Aharonowiz et al. 2008) after transplantation in non-human primates and mice with EAE, respectively. Again, the therapeutic effects of both these latter NPC sources was not related to graft or host-driven remyelination but was rather mediated by an immune regulatory mechanism that protected the CNS from immune-mediated injury.
Taken together, the results of experimental studies that we describe here have started to reveal the mechanisms by which neural stem cells act as immune regulators and in so doing create environments within the CNS that favour protection and repair. Such mechanistic insights are essential if the full therapeutic potential of CNS stem cells is to be achieved.
7.4 Conclusions
Since the first transplant of stem cells into the spinal cord of rodents in which an acute demyelinating lesion was induced (Liu et al. 2000), we have witnessed a spur of experimental cell-based transplantation approaches aimed at fostering biological and molecular mechanisms underlying CNS repair. Theories assuming that no (or very little) renewing potential is identified within the adult CNS have been contravened, new promising sources of myelinogenic cells for transplantation purposes (i.e. olfactory bulb ensheathing cells, adult and embryonic stem cells) have been characterized, and new cell-replacement strategies have been proposed and established. A better understanding of the dynamics of endogenous remyelination has been achieved, and insights concerning the process of remyelination driven by site-specific myelin-forming cell transplantation have been discovered. This has led to the first clinical trial—performed in MS patients—based on autologous Schwann cell transplantation into brain areas of autoimmune demyelination. This Phase I trial was carried out in three patients only, and biopsies of the site of transplantation apparently failed to identify transplanted Schwann. There were no repeats of adverse events associated with transplantation, yet the lack of publication of the results of the trial leaves unanswered questions. Other limitations of cell transplantation, which have not yet been overcome, include (a) the limited number of highly myelinating cells that can be grown in vitro and (b) the limited migratory capacity of myelinating cells once transplanted. Somatic stem cells might represent therefore an alternative and promising area of investigation with some potential in its essence.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree