Median tumor size
BCLCa stage C
Previous treatment
Dose/fraction
Prescription
Local control rate
Overall survival rate
Toxicity ≧Grade 3
Author/year
n
(mm)
(%)
(%)
(Gy/fr)
(%)
Bujold, 2013, Canada [15]
102
72
65.7
52
24–54 Gy/6 fr
N.A.
87.0 % (1y)
34.0 % (2y)
30c
Andolino, 2011, USA [16]
60
31
N.A.b
N.A.
24–48 Gy/3–5 fr
80 % isodose
90 % (2y)
67 % (2y)
35c
Kwon, 2010, Korea [17]
42
N.A.
0
81
30–39 Gy/3 fr
70–85 % isodose
67.5% (3y)
58.6% (3y)
2.4d
Kang, 2012, Korea [18]
47
29
17
100
42–60 Gy/3 fr
70–80 % isodose
94.6 % (2y)
68.7 % (2y)
10.7e
Sanuki, 2013, Japan [19]
185
26
N.A.
68.1
40 or 35 Gy/5 fr
70–80 % isodose
91.0 % (3y)
70.0 % (3y)
13c
Kimura, unpublished, Japan
65
16
0
92.3
48 Gy/4 fr
Isocenter
100 % (2y)
76.0 % (2y)
23.1c
In these studies, some patients underwent TACE before SBRT. Kang et al. reported an excellent 2-year local control rate 94.6 % with SBRT for inoperable HCC as a local salvage treatment after incomplete TACE and concluded that SBRT plus TACE is promising [18]. They suggested several theoretical advantages of combined SBRT and TACE, such as tumor shrinkage, the remaining lipiodol as a target for image guided radiotherapy, and enhance sensitivity to irradiation. We retrospectively compared the treatment results of SBRT plus TACE with TACE alone in patients with small, solitary HCC [21]. No significant difference was observed in overall survival and toxicity between the groups. However, local tumor control was significantly superior in the SBRT plus TACE group than in the TACE alone group (p < 0.001), and disease-free survival of 12 patients without previous HCC treatment in the SBRT group was significantly superior to that of the TACE alone group (15.7 months versus 4.2 months; p = 0.029). In these studies of combined SBRT and TACE, local control is promising; however whether this combination can improve overall survival is unclear because of the lack of a phase III study.
14.3 Adverse Effects of SBRT and Dose Constraints for Liver
Radiation-induced liver disease (RILD) is an adverse effect that is pathologically characterized as a veno-occlusive disease (VOD) [22], and is categorized into “classic” and “nonclassic” RILD [23]. “Classic” RILD involves anicteric hepatomegaly and ascites, typically occurring within 4 months, and often exhibits severe or fatal complications following conventional radiotherapy for large hepatic volumes [23, 24]. In contrast, “nonclassic” RILD, typically occurring between 1 week and 3 months after therapy, involves liver transaminases elevated more than 5 times the normal upper limit or Common Terminology Criteria for Adverse Events (CTCAE) grade 4 levels in patients with baseline values more than 5 times the normal upper limit within 3 months after completion of radiotherapy, or a decline in liver function [measured by a worsening of Child-Pugh (CP) score by 2 or more], in the absence of classic RILD [23]. Recent advances in imaging and radiation techniques provide high radiation doses to conform to focal HCC, such as SBRT, and several studies listed in Table 14.1 have described good treatment results for SBRT without severe clinical signs of “classic” RILD. However, toxicities of more than grade 3 CTCAE criteria (so called “nonclassic” RILD) is frequently observed, shown in Table 14.1. Particularly, patients with worse baseline liver function are at higher risk for developing “nonclassic” RILD even in SBRT. Lee et al. reported that in their experience of 131 patients with HCC who received 3-dimensional conformal radiotherapy, the incidence of liver complications was significantly increased in patients with CP class B (p = 0.044). They concluded that indicators of liver function status such as CP class may be important and useful parameters for predicting radiation-related liver disease [25]. In addition, in our unpublished data of 65 patients (56 patients with CP class A and 9 patients with CP class B) who underwent SBRT (median 48 Gy/4 fr), 15 patients (23.1 %) developed grade 3 toxicity including 5 patients with decreased platelet counts before SBRT. CP class B was the only significant factor associated with more than grade 3 (CTCAE ver 4.0.) hepatic toxicity. Irradiated liver dose is also risk factor for severe hepatic toxicity. Bujold et al. reported a significantly higher median liver mean dose was observed in patients who developed grade 5 toxicity (CTCAE ver 4.0.) compared with those who did not (18.1 Gy versus 15.4 Gy; p = 0.02) in their phase I/II study [15]. Son et al. reported in their experience of 47 patients with HCC who received SBRT using CyberKnife, 4 (11 %) showed progression of CP class, and multivariate analysis showed that the only significant parameter associated with the progression of CP class was liver volume after receiving a dose of less than 18 Gy [26]. To avoid hepatic toxicity, the dose- volume limits guidelines recommended by the Quantitative Analyses of Normal Tissue Effects in the Clinic (QUANTEC) for normal liver dose constraints of 3–6 fractions of SBRT, have been offered. For example, the MLD (liver minus GTV) should receive <13–18 Gy, or that an MLD of ≥700 mL of the normal liver should receive ≤15 Gy [23].
Gastrointestinal toxicity is another severe problem associated with SBRT for HCC. Kang et al. reported that 5 (10.5 %) of 47 patients experienced more than grade 3 gastrointestinal toxicity including grade 4 gastric ulcer perforation in 2 patients (4.3 %) [18]. In patients with liver cirrhosis, portal hypertension probably affects the gastrointestinal mucosal defensive and healing mechanisms, whereas liver cirrhosis increases gastrointestinal toxicity [27]. It is recommended that target proximity to luminal gastrointestinal tract should be far from tumor more than, i. e., 2 cm [20].
14.4 Assessment of HCC Response and Normal Liver Reaction After SBRT
14.4.1 Assessment of HCC Response
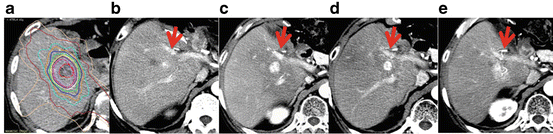
The modified Response Evaluation Criteria in Solid Tumors (mRECIST) was proposed to assess HCC response [28]. HCC is usually diagnosed by its characteristic appearance of early arterial-phase enhancement and portal venous phase hypodensity, which is revealed in most patients with either dynamic computed tomography (CT) or combined angiography CT. These guidelines consider any necrotizing effects or tumor blood flow, and complete response was defined as the disappearance of any intratumoral arterial enhancement in all target lesions. Thus observation of residual early arterial enhancement is important. In our previous report, we mentioned the dynamic CT appearance of tumor responses after SBRT for HCC [29], whereas residual early arterial enhancement was observed >3 months after SBRT in 28.4 % lesions (19/67 lesions; Fig. 14.1). We concluded that early assessments within 3 months could result in misleading response evaluations. In this study, we also observed shrinkage or disappearance of residual early arterial enhancement for more than 6 months after SBRT in 2 patients at 10 and 11 months. Therefore a follow-up of at least 12 months would be required for accurate assessments. Other modalities should also be considered in these cases, such as gadoxetate disodium enhanced-magnetic resonance imaging (EOB-MRI) or enhanced ultrasound (US).