Fig. 3.1
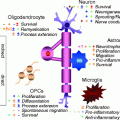
At rest, microglia display a ramified morphology with a thin rod-like cell body. Upon activation, microglia transition to an intermediate hyper-ramified morphology, followed by an activated morphology with a hypertrophied cell body and shortened/thickened cell process. Microglia can also assume an amoeboid (macrophage-like) morphology when engaged in phagocytosis
An alternative view has emerged from the examination of very early MS lesions, in some cases where death occurred within 24 h of symptom onset. Barnett and Prineas (2004) described that these very early lesions exhibited extensive oligodendroglial apoptosis together with microglial activation in the absence of lymphocytic infiltration (Barnett and Prineas 2004). These observations challenged the findings of Lassmann et al., arguing that the pathological variability described in these prior studies reflected different stages of lesion development rather than distinct pathologies, a controversy that remains essentially unresolved. These two studies also proposed differing roles for microglia in MS, with Lassmann et al. emphasising the commonality/central role of the adaptive immune response, whereas Barnett and Prineas emphasised that microglia might play a fundamental role at the earliest stage of disease activity, prior to widespread T cell infiltration. Another important feature that Barnett and Prineas identified was of oligodendroglial apoptosis, suggesting that activation of the innate immune system could be reactive, although it is unclear as to what role such activated cells might have in subsequent disease pathogenesis and, in particular, the activation of a subsequent adaptive immune response.
Further analysis from the Lassmann group has provided a clarification of their classification, which serves to accommodate some of the findings of Prineas and Barnett in relation to microglial activity, at least for so-called pattern III. The Lassmann group has chosen to interpret that early microglial activation in this pattern reflects a critical role in lesion pathogenesis, which could serve as the impetus for subsequent activation of the adaptive immune response and frank demyelination. In contrast, it has been posited by others that, in some patients, microglial activation occurs relatively late in the disease course (Weiner 2008). This activation was hypothesised to correlate with conversion from relapsing–remitting disease, driven by a peripherally invoked adaptive immune response, and which is responsive to immunomodulation, to a phase of progressive disability driven by innate immunity behind an intact blood–brain barrier, which is refractory to systemic immunotherapy.
3.2.2 Distribution of Microglia within MS Lesions and Putative Functional Roles
Detailed analysis of the distribution of microglia within extant lesions has been undertaken and provides some basis for speculation concerning their potential roles in disease pathogenesis and in the remyelinative process. Macrophages/microglia are found in large numbers in acute and chronic active lesions where, on average, they outnumber lymphocytes by 10–20 times. Microglia are also found in lower numbers in inactive, demyelinated and remyelinated lesions (Barnett et al. 2006; Brück et al. 1995).
Analysis of human tissue can only provide a snapshot of histopathology and therefore provides no direct insights into pathogenesis. Nevertheless, some intimate anatomical associations between microglia and other neural cells have been identified, suggesting that microglia might be involved in both the disruption of axons and of myelin lamellae. Such macrophages have been shown to exhibit IgG capping at the site of attachment of myelin (Prineas and Graham 1981), suggesting that immune-ligand-mediated phagocytosis could be occurring. In addition, microglial processes positive for MHC class II within active MS lesions have been shown to interact with myelin internodes, suggesting that microglia are actively involved in demyelination (Bö et al. 1994; Trapp et al. 1999). Furthermore, microglia have also been shown to interact with oligodendrocyte cell bodies, where, in one particular study, 17 % of all microglia within actively demyelinating lesion boundaries were apposed to or surrounded by oligodendroglial somata (Peterson et al. 2002). Human MS lesions and experimentally generated demyelinated plaques also contain phagocytically active microglia/macrophages that can be identified by staining for myelin degradation products or by ultrastructural examination of cell contents (Barnett and Prineas 2004; Bö et al. 1994; Brück et al. 1995; Prineas and Graham 1981).
It has been harder to obtain compelling evidence from the analysis of human tissue that microglia are actively involved in the myelin repair process. Nevertheless, histological analysis has demonstrated that myelin regeneration can occur within 2 days of the onset of MS symptoms and this correlates with the presence of oligodendrocyte progenitor cells within lesions that also contain phagocytically active microglia (Barnett and Prineas 2004). A close spatial relationship between OPCs and microglia has also been demonstrated in MS lesions (Nishiyama et al. 1997), and a recent post-mortem study of tissue from a patient with long disease course showed a high density of HLA-DR+ macrophages/microglia was positively correlated with remyelination (Patani et al. 2007). Collectively, these studies provide good circumstantial evidence that microglia have a role to play not only in pathogenesis but also in repair.
However, it remains uncertain as to whether individual microglial cells can be induced to transform from a pathogenic to a reparative phenotype or whether such transformation depends on the stimulation/activation of a separate cohort of cells.
3.3 Microglial Heterogeneity: Disparate Functions and Targets?
The question of pathological heterogeneity is pertinent not only to whether there are variable patterns between individuals but also as to whether microglial phenotype varies according to region, disease duration and/or age.
3.3.1 Developmental Origins
It is generally believed that CNS microglia are monocytic cells of mesodermal origin deriving from the yolk sac (Alliot et al. 1999; Hirasawa et al. 2005). An elegant study by Hirasawa et al. employed transgenic mice expressing a fluorescent marker enhanced green fluorescent protein (eGFP) under the control of the macrophage/microglial promoter for ionised calcium-binding adaptor molecule 1 (Iba1) to track microglial origin and development. Invasion of cells of monocytic origin into the CNS was first observed at embryonic day 10.5 (E10.5) in Iba1-eGFP mice. Microglia were shown to enter the brain parenchyma via the meninges, having been carried through the circulation from the yolk sac. A strong eGFP signal in the forebrain, spinal cord and eye was present at E11.5 and persisted into adulthood. A second, smaller microglial invasion occurred at postnatal day 6 (P6) in the supraventricular corpus callosum and cingulum. The signal from these cells diminished over the course of the second postnatal week, presumably due to migration of these cells to other brain regions. Amoeboid microglia migrate throughout the parenchyma and differentiate into a resting, ramified morphology once they have reached their final position (Cuadros and Navascués 1998; Hirasawa et al. 2005; Marín-Teva et al. 1998).
3.3.2 Regional Variation
It is also important to note that in addition to parenchymal microglia, there is a population of cells that is also resident adjacent to the CNS vasculature, known as perivascular macrophages. These cells express many of the same cell surface antigens as parenchymal microglia and the two cell types are therefore difficult to distinguish in pathological conditions. Like microglia, perivascular macrophages are bone marrow-derived and provide an interface between the relatively immune-privileged CNS environment and the circulation. These cells can interact with CNS-derived antigens and present them to circulating T-lymphocytes (Streit et al. 1999). It is interesting to note that it has been suggested that microglia and macrophages differ in their intrinsic capacity to phagocytose myelin and in the extent to which this is influenced by opsonisation (Mildner et al. 2007; Mosley and Cuzner 1996). The situation is further complicated in circumstances where monocytes invade the parenchyma. These observations raise the question as to whether these various cells have intrinsic differences in their capacity to either assist or impede the remyelinative process.
3.3.3 Sources in Disease
Initially, it had been suggested that Mac-1+/CD45hi peripheral macrophages might contribute significantly to the repertoire of inflammatory cells identified within zones of demyelination. This had been assessed by McMahon et al. by lethally irradiating mice, followed by rescue with transplanted bone marrow from GFP+ transgenic mice and then exposing the animals to cuprizone. In these animals, GFP+ cells were identified within the CNS after 2 weeks of cuprizone challenge, although they were significantly outnumbered by parenchymal microglia, such that at 3 weeks they accounted for only some 8 % of the inflammatory cells within the corpus callosum. It was reported, on the basis of HRP staining, that the blood–brain barrier was maintained in these irradiated animals: subtle deficits may not, however, have been identified using this technique. In fact, recent work using chimeric mice failed to identify any evidence of microglial progenitor recruitment from the circulation in CNS neurodegenerative disease, at least when the blood–brain barrier is intact, suggesting that microgliosis is induced by local expansion in these models (Ajami et al. 2007; Mildner et al. 2007).
3.4 Interrogating Functional Roles for Microglia in Health and Disease
3.4.1 Surveillance
A transgenic mouse strain expressing eGFP behind the promoter region for the microglia/macrophage-specific chemokine receptor CX3CR1 (fractalkine receptor) has further enriched our understanding of microglial phenotype in both a normal physiological context and in response to acute injury (Davalos et al. 2005; Nimmerjahn et al. 2005). In particular, transcranial two-photon microscopy has allowed the visualisation of superficial microglia within the first 200 μm of the cortical surface. It was revealed that microglial cell bodies are evenly distributed throughout the cortex, with cell somata lying 50–60 μm apart. Microglial cell bodies were small and generally rod shaped with numerous branched processes extending from the soma (Nimmerjahn et al. 2005). Imaging for up to 10 h revealed that there was very little somatic translocation in the quiescent state. In contrast, microglial processes were shown to be highly motile with extensive process extensions and retractions cycling in an apparently random manner with speeds of up to 4.1 μm/min. Interestingly, when neighbouring microglial cell processes came into contact, they would repel each other, thereby indicating that each cell has a defined region of surveillance. The average protrusion lifespan was about 4 min. Bulbous microglial processes were observed actively engulfing tissue detritus during routine surveillance. These observations lead the investigators to conclude that, at rest, the extracellular microenvironment is completely surveyed once every few hours (Nimmerjahn et al. 2005).
Work by Nimmerjahn et al. has identified that microglia responded immediately to a laser-induced disruption of the blood–brain barrier (BBB). Cells within 90 μm of the injury site were shown to extend processes towards the site of damage and to retract processes oriented in the opposite direction. Whilst process morphology did not change, the cell soma was shown to hypertrophy. Interestingly, the average speed of process extension was the same in the response to injury as it was in the quiescent state at rest (Nimmerjahn et al. 2005).
Using these same transgenic mice, Davalos et al. (2005) independently confirmed the findings of the Nimmerjahn study. In addition, it was found that a single microglial cell could respond to two acute injuries within very short distances of each other by extending hypertrophied branches to both sites without somatic translocation. In the Davalos study, it was shown that processes which extended to an injury site could fuse together to encapsulate the damaged tissue. It was further shown that injury-mediated branch motility was regulated by extracellular adenosine triphosphate (ATP) released by astrocytes in damaged tissue, acting via microglial purinergic P2 receptor.
The above two studies have revolutionised our understanding of microglial function in both normal physiological and acute pathological states, revealing that these cells are very quick to respond to injury, and indicating that, even at rest, microglia are highly active cells responsible for maintenance of parenchymal homeostasis.
3.4.2 Experimental Models of Demyelination
Independent of the interpretations concerning microglial function that can be gleaned from cross-sectional studies of human pathological samples, it is generally agreed that direct analysis of their function in the context of demyelination requires the use of either animal models or detailed in vitro study.
There are several experimental tools that are used in a laboratory context to study demyelination and remyelination. These can be broadly divided into two groups, immune-mediated models of demyelination and toxin-mediated models. The most common immune-mediated model of demyelination, rodent experimental autoimmune encephalomyelitis (EAE), is induced by the active sensitisation of CNS tissue to myelin antigens or by the passive transfer of auto-reactive T-lymphocytes. This leads to a stereotyped, ascending paralysis correlated with microglial activation, mononuclear cell infiltration into the CNS parenchyma and the destruction of myelin, oligodendrocytes and axons. This model can be used to study factors that influence disease pathogenesis, but not disease aetiology. As in MS, the animal models of inflammatory demyelination are complex, and demyelination and remyelination are often contemporaneous, making it difficult, in isolation, to formally assess a role for microglia in the repair process.
In addition to EAE, three toxin-mediated models are routinely used to study demyelination and remyelination. On the one hand, the copper chelator cuprizone (bis(cyclohexanone)oxaldihydrazone), when delivered to mice in their feed, induces demyelination of the midline corpus callosum and, to a lesser extent, the superior cerebellar peduncles (Blakemore 1973). On the other hand, the toxins lysolecithin and ethidium bromide can be injected focally into the white matter of the central nervous system (CNS) of experimental animals, producing demyelination, oligodendrocyte death and a variable astrocyte response, but with preservation of axons. Demyelination in these models is followed by stereotyped and extensive remyelination of over 90 % of affected axons in the lesions. Toxin-induced models are particularly useful models for investigation of the role of microglia/macrophages in repair since their presence in the lesion is in response to injury without them being involved in the primary injury, although they could still be involved in secondary pathogenic events.
3.5 Microglial Activities
The spectrum of microglial activity in response to injury has been previously posited to comprise two broad components: inflammation and immunomodulation. The inflammatory response is engaged to destroy damaged, infected or invading cells, which may incur bystander damage. Immunomodulation, on the other hand, promotes cell survival and tissue repair. In general, an inflammatory response resolves once tissue is repaired (Muzio et al. 2007). It is also important to note that other important functions for microglia have also been identified, in particular phagocytic activity. Inhibition of microglial activity can, therefore, be either beneficial or deleterious, depending on the circumstance.
A variety of molecular determinants of microglial activation have been identified. A key issue will be to identify how the relevant signalling pathways act, either in isolation or in a coordinated way, to effect the upregulation of a set of cell surface antigens leading to a specific phenotypic response, whether it be inflammatory, immunomodulatory or phagocytic or a combination thereof.
A variety of receptors expressed by microglia have been identified that, when activated, can induce the migration of these cells in response to ligands within the extracellular milieu. These include the chemokine receptors CCR5 (Carbonell et al. 2005) and CXCR3 (Rappert et al. 2004), the purinergic receptors P2Y12/13 which act in a beta-1 integrin-dependent manner (Nasu-Tada et al. 2005), the B-1 bradykinin receptor (Huisman et al. 2008), the neurotensin receptor-3 (Martin et al. 2003), the vascular endothelial growth factor receptor (Forstreuter et al. 2002), the pattern recognition receptor CD36 (Stuart et al. 2007), the neurotrophin receptor TrkA (De Simone et al. 2007) and the IL-6 superfamily receptor complexes (Sugiura et al. 2000).
3.5.1 Phagocytic Activity
Microglia have long been recognised to be phagocytic: teleologically, this activity has evolved in order to remove pathogens as part of a pro-inflammatory response. This is known to be mediated via activation of the toll-like receptors (TLRs). However, as indicated previously, in the context of autoimmune disease, microglial activity can be directed to the phagocytosis of a variety of endogenous structures including myelin lamellae, axons, apoptotic cells and myelin debris, resulting in a range of influences upon either disease pathogenesis or repair.
Several studies have used toxin-induced models of demyelination in the rat to study the role of microglial phagocytosis. Phagocytically active macrophages can be recognised in tissue sections by the presence of myelin degradation products and lysosomal lipids within their cytoplasm. It is important that such studies dissect out the influence that phagocytosis is having upon disease severity in that particular circumstance and to identify, if possible, the physical structures that are being targeted. As a corollary, it is necessary to understand the molecular mechanisms by which phagocytosis of the various targets is expedited if focused strategies to modify phagocytosis in a targeted way are to be developed for the treatment of human disease.
3.5.1.1 Targets of Phagocytic Activity
3.5.1.1.1 Myelin
In experimental models of MS, myelin lamellae have been observed to be located in coated pits on the macrophage surface. Coated pits that express the protein clathrin are known to be the hallmark of receptor-mediated phagocytosis. Myelin opsonised with anti-myelin antibodies is also more readily phagocytosed (Trotter et al. 1986).
3.5.1.1.2 Axons
Both experimental models of MS and in vitro data suggest that microglia can be directly responsible for axonal transection (Fordyce et al. 2005). Additionally, microglia in vitro have been shown to lethally injure neurons in culture via a process that requires cell proximity and which is potentially mediated via nitric oxide (Gibbons and Dragunow 2006) and also which could be potentiated by stimulating Kv1.3 channel activity (Fordyce et al. 2005).
3.5.1.1.3 Apoptotic Cells
Until recently, only phosphatidylserine (PS) had been identified as a recognition ligand on apoptotic cells. The PS molecule is known to be relocated from the internal to the external surface of the plasma membrane of apoptotic cells and has now been identified to be recognised by either Tim4 (Miyanishi et al. 2007) or by the secreted protein, growth arrest-specific gene 6 (Gas6). In turn, Gas6 has the capacity to bind to the TAM (Tyro 3, Axl and Mer) family of receptor tyrosine kinase receptors on microglia and through a modified type II phagocytic response, acting via Rac and Vav in a microtubule-dependent way, can induce phagocytosis (Grommes et al. 2008). It is also important to note that Gas6 not only induces phagocytosis but can also induce an anti-inflammatory response by microglia (Binder et al. 2008; Grommes et al. 2008) (see below).
Recently, other important molecular mediators of apoptotic phagocytosis and of tissue homeostasis have been identified. First, it has been demonstrated that calreticulin (an obligate endoplasmic reticulum protein that has been shown to be expressed on the cell surface during apoptosis) serves as a second general recognition ligand by binding and acting upon the LDL-receptor-related protein on the engulfing cell. Second, the triggering receptor expressed on microglial cells-2 (TREM2) has been identified as an innate immunoreceptor which acts to induce phagocytosis via phosphorylation of DNAX-activating protein of 12bDa (DAP12) and by inducing cytoskeletal reorganisation. Interestingly, knockdown of TREM2 in microglia has been shown not only to inhibit the phagocytosis of neural cells but also to potentiate a pro-inflammatory response, with upregulated expression of the genes encoding for tumour necrosis factor-alpha (TNFα) and nitric oxide (NO) synthase. A beneficial effect of TREM2 has recently been identified in EAE, given that mice given myeloid cells which overexpressed TREM2 exhibited less severe clinical disease as well as reduced axonal damage and demyelination but with increased lysosomal and phagocytic activity (Takahashi et al. 2007). On the other hand, antibody-mediated blockade of TREM2 during the effector phase of the disease resulted in exacerbated disease (Piccio et al. 2007). The TREM2/DAP12 complex has also been shown to be important in man such that either TREM2 or DAP12 deficiency results in an inflammatory neurodegenerative disease in the 4th/5th decade of life (Bianchin et al. 2006). The ligand responsible for activating this system is yet to be identified (Takahashi et al. 2005).
3.5.1.1.4 Myelin Debris
Macrophage depletion in young rats has been shown to result in impairment of remyelination, in part due to delayed phagocytosis of myelin debris (Kotter et al. 2005). To examine this issue further, either myelin debris, liver cell membranes or PBS were added to ethidium bromide-induced demyelinating lesions in the rat, and the capacity to remyelinate was assessed. The authors found that whilst non-CNS debris inhibited efficient remyelination relative to PBS-treated controls, myelin debris had an even greater inhibitory effect on myelin regeneration, and this effect was found to be independent of OPC and macrophage recruitment. The authors demonstrated that the remyelination block was at the level of oligodendrocyte maturation: oligodendrocytes were able to make contact with axons and to ensheath them but were unable to compact the myelin (Kotter et al. 2006).
In summary, microglia appear to play a critical phagocytic role that leads to the promotion of oligodendrocyte maturation and remyelination, possibly by removing a negative regulator. A candidate negative regulator would be the leucine-rich repeat and Ig domain-containing Nogo receptor interacting protein (LINGO), which has been shown to be expressed by oligodendrocytes (Mi et al. 2005). It is also possible, although untested, that impaired axonal regeneration could be contributed to by uncleared myelin debris (Vargas and Barres 2007).
The molecular mechanisms that regulate the capacity of microglia to remove myelin debris are beginning to be determined. Firstly, leukaemia inhibitory factor (LIF), a neuropoietic cytokine already known to be induced in animal models of central demyelination, has been shown to stimulate myelin uptake by macrophages to enhance myelin clearance. This effect is accompanied by activation of the JAK/STAT signalling pathway (Hendriks et al. 2008). Both the complement component receptor CR3 (Mac1) (Rzepecka et al. 2009) and the scavenger receptor—AI/II interact with calreticulin and have been implicated in mediating the phagocytosis of myelin by microglia and macrophages (Reichert and Rotshenker 2003), a process that appears dependent upon co-expression of MAC-2 (Rotshenker et al. 2008). Recently, it has also been identified that AI/II signals via the TAM receptor family member, Mer, during apoptotic cell uptake by murine macrophages (Todt et al. 2008). In addition, Axl, another TAM family member, appears to play a role in myelin debris clearance. In Axl null mice subjected to cuprizone-induced demyelination, there is not only a reduction in numbers of microglia but also a reduction in their phagocytic activity. This is associated with both a subsequent delay in the development of mature oligodendrocytes and increased axonal damage in the remyelinative phase (Hoehn et al. 2008).
3.5.1.2 Remodelling
An additional activity ascribed to microglia is that of synaptic stripping. Trapp et al. have demonstrated that microglia activated in response to an inflammatory stimulus can remove up to 45 % of axosomatic connections within the cortex, a process that occurs independent of phagocytosis and which has been deemed to be neuroprotective. Many of the cortical neurons that were stripped of their synapses were of GABAergic phenotype. It has been proposed that loss of inhibitory GABAergic inputs leads to an increased capacity for NMDA receptor activation which stimulates anti-apoptotic neuroprotective signalling pathways regulated by activation of cAMP-response-element-binding (CREB) protein (Trapp et al. 2007).
It has been identified that the C3R on microglia could be an important component of this response, given that both the complement components C1q and C3 are expressed by neural cells, including neuronal synapses and given that mice lacking C1q and C3 are deficient in synaptic pruning (Stevens et al. 2007). Interestingly, TNFα has been reported to inhibit phagocytosis mediated by C3 (CR3) by downregulating CR3 levels (Bruck et al. 1992). The major histocompatibility antigen class I antigen could also have a role, given that it has been shown to be implicated in mediating synapse removal after motor neuron injury (Cullheim and Thams 2007).
3.5.2 Inflammatory Activity
3.5.2.1 Molecular Determinants of Microglial Activation
Factors such as interferon-gamma (IFNγ) produced by key participants in the adaptive immune response (including NK cells, CD4+Th1 cells and CD8+T cells) appear to be key determinants of microglial activation. In fact, it has been hypothesised that a positive feedback loop might be operative, leading to enhanced activation of both microglia and dendritic cells with time, potentially explaining why systemically applied immunomodulatory therapy that works predominantly in the periphery eventually becomes ineffective. Microglial cells have been shown to exhibit increased secretion of proteolytic enzymes, potentiated oxidative stress and enhanced phagocytic activity in response to IFNγ. Microarray analyses have also revealed increases in expression of genes encoding chemokines and MHC proteins by activated microglia (Dheen et al. 2007).
A variety of other environmental factors have been implicated in potentiating a pathogenic microglial response. These include members of the TLR family which are activated via exposure to damage-associated molecular patterns (DAMPs), although they can also have roles in inducing tolerance (Fischer and Ehlers 2008). Macrophage colony stimulating factor also appears to be a significant positive regulator of microglial activity, given that its overexpression induces microglial proliferation and pro-inflammatory cytokine expression (Mitrasinovic et al. 2001).
3.5.2.2 Consequences of Pro-inflammatory Activity in Central Demyelination
A seminal study in clarifying the role of microglia in demyelination made use of a transgenic mouse strain where the thymidine kinase of herpes simplex virus (HSVTK) was inserted under the regulatory control of the alpha chain of CR3, namely, CD11b (Heppner et al. 2005). In this context, inducible suppression of microglia by activation of HSVTK ameliorated EAE disease severity. Furthermore, microglial paralysis in ex vivo slice cultures led to the suppression of reactive oxygen species, pro-inflammatory cytokine and chemokine release, implicating these cells in disease pathogenesis and again advocating a pathogenic role for microglia in demyelination, at least in the effector phase of the disease (Heppner et al. 2005).
3.5.2.3 Molecular Basis for the Pathogenic Effect
Microglia can potentiate inflammation and contribute to the pathogenic process in a variety of ways. First, they serve as antigen-presenting cells and do so via MHC class II molecules which present processed antigens to CD4-positive T-lymphocytes. The MHC class II molecule is upregulated by activated microglia in pathogenic states (Kim and de Vellis 2005), and this serves to amplify the immune response via the process of epitope spreading.
Microglia also have the capacity to secrete a wide variety of signalling molecules including inflammatory cytokines such as TNFα, interleukin-1 (IL-1) as well as leukotrienes, complement components, reactive oxygen intermediates and proteolytic and lipolytic enzymes. These molecules can exert harmful effects by acting either directly or indirectly to induce neural cell damage (Kim and de Vellis 2005; Neumann et al. 2009), although some can also have beneficial effects, depending upon the context in which they are expressed (see below).
The free radical nitric oxide (NO) has been directly implicated in inducing oligodendrocyte death (Gibbons and Dragunow 2006; Merrill et al. 1993) and in promoting MS pathogenesis (Parkinson et al. 1997). Microglia that produce nitric oxide and inducible nitric oxide synthase (iNOS, the enzyme responsible for NO synthesis) have been found in active lesions but not in either chronic MS lesions or normal tissue, supporting the notion that microglia-derived NO is, in part, responsible for disease pathogenesis (Bagasra et al. 1995; De Groot et al. 1997; Liu et al. 2001; Oleszak et al. 1998).
3.5.3 Immunomodulation
Microglia can secrete anti-inflammatory cytokines that could either support damaged cells during a demyelinative insult or serve to enhance repair. Recent work has validated that microglia have an essential role to play in the immunomodulatory effects exerted by β-interferon, a cytokine used for therapeutic benefit in MS. This was assessed by studying the influence of either generalised or cell lineage-specific deletion of the interferon type 1 receptor (IFNAR), which is responsible for mediating β-interferon-induced signalling, in mice subjected to EAE (Prinz et al. 2008). Mice with a generalised deletion of IFNAR performed significantly worse than their wild-type counterparts, with increased myelin destruction, corresponding to increased macrophage invasion of the CNS parenchyma. Interestingly, conditional deletion of IFNAR from myeloid cells (in particular from CD11b-positive microglia, monocytes and granulocytes, but not from either T- or B-lymphocytes) aggravated disease severity. These experiments thus established that CD11b-positve macrophages are the target of endogenously produced interferon: by extrapolation, it is likely that β-interferon as a therapeutic agent exerts a significant component of its effects via these cells (Prinz et al. 2008).
Recent data also indicate that the conversion of macrophages to an immunomodulatory phenotype involves not only IFNAR in isolation but also other receptors, in particular the TAM subfamily of receptor tyrosine kinases. This is potentially of direct relevance to the pathogenesis of MS, as the TAMs and their cognate ligand Gas6 have recently been identified as having a protective effect during the demyelinative stage of cuprizone-induced demyelination (Binder et al. 2008). Over the course of a 6-week insult, expression of Gas6, Axl and Mer mirrored microglial activation and infiltration within the corpus callosum. Gas6 knockout mice displayed a significantly greater degree of demyelination, which was not only commensurate with loss of mature oligodendroglia but also an increase of microglia/macrophage numbers (Binder et al. 2008). Complementary in vitro studies established that exogenous Gas6 both potentiated oligodendrocyte survival and modulated the expression of pro-inflammatory cytokines by lipopolysaccharide (LPS)-stimulated microglia.
The IL-6 family of cytokines can also regulate microglial activity. All members of this family, including LIF, CNTF and IL-6, signal through a common gp130 receptor subunit, with specificity of ligand binding driven by an array of alternative receptor subunits which associate with gp130 to form heterologous receptor complexes. Hendriks et al. (2008) have reported that LIF can inhibit the production of oxygen radicals and TNFα by macrophages (Hendriks et al. 2008). Kerr and Patterson have also reported that LIF can induce microglia to produce IGF-1, another trophic factor that is capable of inducing oligodendrocyte survival (Gveric et al. 1999; Hinks and Franklin 1999; Kerr and Patterson 2005) (see Sect. 3.5.4). Interestingly, it has been shown that partial phosphorylation of the transcription factor, STAT-3, by CNTF in microglia results in enhanced survival of neurons in mixed cultures, whereas application of IL-6, which leads to more robust phosphorylation of STAT-3, results in potentiated neuronal loss. This suggests that gp130-mediated signalling can contribute to either pathogenesis or repair, depending on circumstance, predicated on the availability of particular ligands within the family and upon the expression of key co-receptors such as the non-signal transducing GPI-linked CNTF receptor-alpha, which is required for CNTF binding.
Other cytokines could also exert anti-inflammatory roles: for example, IL-10, which can also activate STAT-3, inhibits the production of TNFα and nitric oxide by macrophages.
It has also been reported that retinoic acid (RA), a vitamin A metabolite, suppresses the activation of microglial cells and inhibits the expression of TNFα and NO (Diab et al. 2004; Xu and Drew 2006). These effects are mediated through two subfamilies of nuclear receptors, the retinoic acid receptors (RARs) and the retinoid X receptors (RXRs). It has also been shown that RA enhances the expression of the gene encoding transforming growth factor-β1 (TGFβ1), a factor which, by increasing Iκβ expression, can inhibit nuclear translocation of NFκβ, a key transcription factor implicated in the pathogenic effect of microglia (Dheen et al. 2005).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree