15 Non-Ultrasound Imaging of the Urogenital Tract Ultrasound has an important role in the investigation of the urinary tract. However, other imaging modalities are needed when ultrasound is not conclusive. The aim of the present chapter is to summarize and discuss these other imaging modalities in the context of diagnostic pathways for certain diseases, particularly in a complimentary role to ultrasound. Prior to the introduction of ultrasound, the plain abdominal radiograph followed by an intravenous or excretory urogram (IVU) was the mainstay of urinary tract imaging, staples which still maintain an important role in current clinical practice. The plain radiograph will demonstrate the presence or absence of calcification or calculi related to the urogenital tract. It is important to ensure the entire urogenital tract is imaged adequately from the upper poles of the kidneys to the bladder base. Additional views may be necessary; oblique views or tomography may improve visualization of the upper tracts if bowel gas or feces obscure the relevant areas. Radiographs in both inspiration and expiration will determine whether calcification is intrarenal or extra-renal. On plain radiographs, it is often possible to determine the renal contour and the outline of the normal psoas muscle due to the differential density of kidney and adjacent perinephric fat. Despite this, renal outlines are better demonstrated following administration of intravenous contrast at IVU or, ideally, with ultrasound. Absence of the psoas muscle outline may suggest retroperitoneal disease, but the psoas muscle outline is not present in up to 25% of “normal” radiographic examinations. The lower ribs, lumbar spine, and pelvis should be scrutinized for congenital vertebral anomalies, such as spina bifida in children and destructive lesions in adults that might indicate metastatic disease. Excretory urography was first introduced into radiological practice in the mid-1900s and the technique has changed little over the intervening years. Although an IVU remains an important source of information regarding the upper tract anatomy including the ureters and gives some indication of renal function, an IVU rarely stands alone. Increasingly, bowel preparation (administration of laxatives) is not required, although the patient should be well-hydrated, particularly if there is a history of renal impairment, diabetes mellitus, or multiple myeloma. An IVU is contraindicated in the presence of iodine allergy, previous allergic reaction, and care needs to be taken in those with severe asthma. Following a control radiograph, a nonionic, low-os-molar contrast medium is injected as an intravenous bolus. A standard radiographic sequence is detailed in Table 15.1. However, this will be subject to local guidelines and the clinical indication for the study (Table 15.2). Oblique, prone, and delayed images may all be helpful in maximizing the information obtained from the study. The early images will allow for assessment of renal size, shape, contour, and position, as well as perfusion.
Introduction
Plain Abdominal Radiograph
Intravenous Urogram
Immediate coned renal area ± tomography |
Coned renal area 5 mins after contrast injection |
Abdominal compression (if no obstruction/recent surgery/aortic aneurysm) |
Coned renal area 10 mins after contrast injection |
Full-length release at 15 mins to include kidneys/ureters/bladder |
Full-length or coned after micturition |
Calculus disease |
Obstruction |
Mucosal lesions in the investigation of hematuria |
Developmental anomalies |
Papillary necrosis |
Medullary sponge kidney |
Tuberculosis |
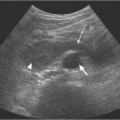
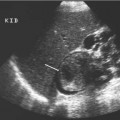
Fig. 15.1 An intravenous urography image of the right kidney, demonstrating the fine linear tracks (arrows) of early papillary necrosis in an otherwise normal pelvicalyceal system. These appearances would not be seen with ultrasound
Later radiographs will demonstrate the morphology of the papillae and calyces and also indicate the degree of renal excretion. These images should be interpreted in the context of the pattern of normal variants, which include complex and unusually large or small calyces, papillary blushes, and vascular impressions. Developmental anomalies, such as a horseshoe, duplex, or pelvic kidney should be readily demonstrated. A dilated pelvicalyceal system as a result of obstruction secondary to calculi, tumor, or bladder outflow obstruction is well-documented. However, one of the most important indications for an IVU is the investigation of hematuria and medical renal disease; an IVU is the only noninvasive imaging modality that can demonstrate calyceal and ureteric abnormalities with high resolution. Abnormalities including papillary necrosis (Fig. 15.1), medullary sponge kidney, renal tuberculosis, and small transitional cell carcinomas may only be seen at IVU.
Micturating Cystourethrography and Other Contrast Studies
The micturating cystourethrogram (MCU) is an invasive examination requiring direct catheterization of the uri-nary bladder, primarily utilized for the investigation of pediatric urinary tract infections (UTIs) and reflux nephropathy. Using an aseptic technique and following catheterization, dilute water-soluble iodinated contrast medium is instilled into the bladder to full capacity. Once the patient is in the erect position, the catheter is removed and the patient empties their bladder whilst concurrent fluoroscopic imaging documents any vesicoureteric reflux (VUR). Oblique films are necessary to show the posterior urethra, which is important in boys to demonstrate the presence of posterior urethral valves.
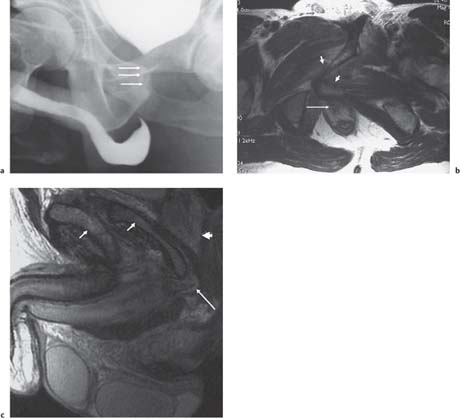
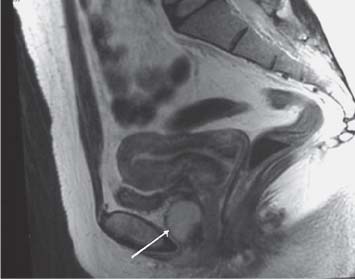
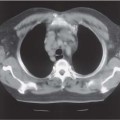
The principal method for imaging of the male urethra is contrast urethrography, either descending, as above, or ascending. Once fully distended with contrast medium, oblique images will show the full length of urethra from fossa naviculare to sphincter. The urethra can then be assessed for strictures and mucosal lesions, the commonest abnormalities. Following pelvic trauma, contrast urethrography may be combined with a cystogram and descending examination to demonstrate the integrity of the entire urethra (Fig. 15.2). There is no ideal method for imaging the female urethra, and ultra-sound (either transvaginal or a transrectal approach), MCU, and magnetic resonance (MR) imaging may be useful (Fig. 15.3).
Retrograde contrast examination of the ureters after direct catherization, usually under a general anaesthetic, may be the only method of demonstrating small mucosal abnormalities if the IVU is inconclusive. Similarly, with ileal conduits it is often difficult to demonstrate the site of ureteric anastomosis at IVU, and direct introduction of contrast medium into the ileal loop, a “loopogram,” with reflux into ureters may delineate this region.
Computed Tomography
The combination of ultrasound and an IVU may resolve many of the clinical conundrums arising in renal and urological medicine. However, cross-sectional imaging in the form of computed tomography (CT) and MR imaging are vital complementary diagnostic tools. Newer and constantly evolving technology has led to an increase in the use of CT and MR imaging. For example, unenhanced helical CT has largely replaced an IVU for evaluation of presumed calculi-induced renal colic. Table 15.3 lists important applications for the use of CT imaging.
Fig. 15.2 a Cystography combined with an ascending urethro-gram following a “closed-book” fracture, demonstrating an obstructed proximal urethra (arrows). b Axial T2-weighted MR image, demonstrating compression of proximal urethra (long arrow)by overriding pubic rami (short arrows). c Sagittal T2-weighted MR image, demonstrating compression of proximal urethra (long arrow) by overriding pubic rami (short arrows). The prostate is not involved (arrowhead)
Characterization of renal cysts/masses when ultrasound is equivocal |
Staging of malignant disease (renal, bladder, prostate, testicular) |
Assessment of renal calculi (particularly with acute colic) |
Renal trauma |
Evaluation of the retroperitoneumCT angiography |
Fig. 15.3 Urethral diverticulum on sagittal T2-weighted MR image (arrow)
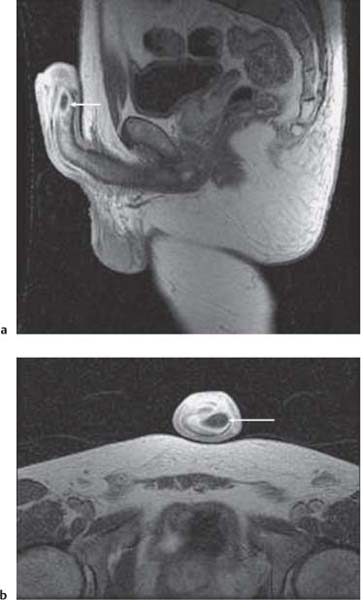
Fig. 15.4 a Squamous cell carcinoma of distal urethra complicated by abscess formation (arrow) in the corpus cavernosum on postgadolinium contrast T1-weighted MR image in the sagittal plane. b Postgadolinium contrast T1-weighted MR image in the axial plane, demonstrating the abscess (arrow) complicating a squamous cell carcinoma
The CT machine comprises multiple x-ray tubes aligned with opposing x-ray detectors, which are arranged in a ring within a gantry. As the patient passes through the scanner, x-ray photons pass through the patient with varying degrees of ease, are then detected and reconstructed to build up an image of differing tissue densities. “Slip-ring” technology and the subsequent arrival of helical then multislice CT scanners in the late 1990s has meant that the abdomen and pelvis can now be rapidly imaged in a single breath-hold, reducing artifact and allowing for fine collimation. In conjunction with advances in software, the resulting images can be reconstructed in multiple planes or as three-dimensional reconstructions. Enthusiasm for the new possibilities that these changes have brought to CT imaging must be tempered by the knowledge that there is an increase in radiation burden and large contrast load to the kidneys in enhanced studies; this is not to be dismissed lightly.
Magnetic Resonance Imaging
MR imaging relies on the spinning hydrogen ions (or protons) present in body water to obtain cross-sectional images. These ions have a positive charge and when placed in a very strong magnetic field align themselves along the axis of the field. By applying radio waves, the protons alter the way they spin and gain energy. When the radio waves are stopped, energy is released as the protons “relax” back to their original state. The energy can then be converted to produce a representative image as different tissues relax at different rates. MR imaging is not suitable for everyone, particularly the claustrophobic patient, although new “open” magnets may partially solve this problem. Contraindications include cardiac pacemakers, intracranial aneurysm clips, and intraorbital metal fragments.
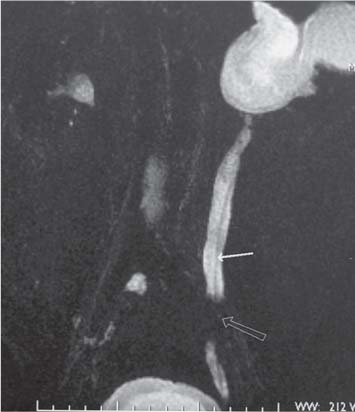
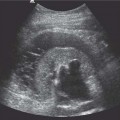
Previously relatively long imaging times proved problematic in MR examinations. However, rapid changes in MR imaging technology allows for faster imaging with less movement artifact, improved image quality, and image resolution (Fig. 15.4). Breath-hold imaging techniques allow for the performance of gadolinium-enhanced MR angiography and venography; this is of particular use in the investigation of renal artery stenosis (RAS) and for evaluation of venous involvement of renal tumors. Although CT is able to show renal calculi more accurately, MR urography using rapidly acquired heavily T2-weighted coronal images can demonstrate dilated pelvicalyceal systems and ureters down to the level of obstruction (Fig. 15.5). Advantages of MR imaging include both the lack of ionizing radiation and use of non-nephrotoxic contrast media; this is of particular advantage in pregnancy and renal failure. Clinical indications for MR imaging have been increasing over the past decade and MR imaging is now the standard imaging modality in staging pelvic cancers, including prostate carcinoma (Table 15.4). Whilst functional MR imaging has been accepted for routine use in neurological disorders, it has not yet been fully explored in nephrourological disease, but has the capacity to challenge nuclear medicine techniques.
Radionuclide Imaging
While the previously described imaging methods contribute primarily anatomical and structural information, radionuclide imaging provides complementary functional and quantitative data. The imaging agents are radiopharmaceuticals, which comprise a radionuclide combined with a biological molecule. These radio-pharmaceuticals are administered, usually intravenously, to target a specific organ or system. The radio-nuclide component emits gamma rays, which are detected using a gamma camera. Anatomical resolution is poor, but unique physiological data is obtained. Table 15.5 summarizes some of the commonly used radio-nuclides and the indications for their use.
Glomerular Filtration Rate
Glomerular filtration rate (GFR) is ideally measured using an agent that is not protein bound, is completely filtered by the glomerulus, undergoes no resorption or secretion, and is only excreted by the kidneys; in clinical practice 51Cr-ethylene diamine tetraacetic acid (51Cr-EDTA) is the most convenient radiopharmaceutical to fit these properties. To calculate the GFR, a plasma clearance method is used, which is defined as the notional volume of plasma cleared of51Cr-EDTA in a given time. A single injection method is used with varying timing of subsequent blood samples, from one to several taken three to five hours after injection but up to 24 hours if the GFR is expected to be < 15 mL/min.
Static Renal Imaging
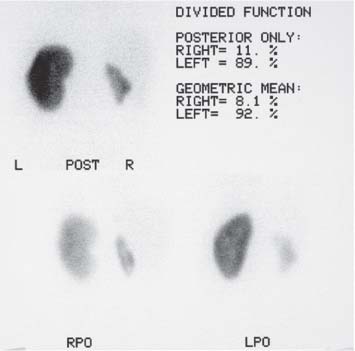
The commonest indication for a99mTc-dimercaptosuccinic acid (99mTc-DMSA) examination is to assess the kidneys for scarring, usually associated with reflux nephropathy in children, and to provide an accurate measure of divided renal function. While roughly 15% of the static renal imaging agent is excreted in the urine, the majority of99mTc-DMSA is fixed in the proximal tubules of functioning renal parenchyma. Images are acquired two to three hours following intravenous injection, maximizing the best target-to-background ratio, although delayed views may be necessary with reduced renal function. Anterior, posterior, and posterior–oblique views are routinely acquired, whilst pelvic views should be obtained if there is any possibility of an ectopic kidney. Focal cortical defects usually indicate the presence of renal scarring (Fig. 15.6). Divided renal function is calculated by measuring the number of “counts” within regions of interest (ROI) drawn around the kidneys on both anterior and posterior views to provide a geometric mean that is independent of renal depth.
Dynamic Renal Imaging
Both 99mTc-diethylene triamine pentaacetic acid (99mTc-DTPA) and 99mTc-mercaptoacetyltriglycine (99mTc-MAG3) can be used to perform dynamic isotope renography. Whilst99mTc-DTPA is solely filtered by the kidney, 99mTc-MAG3 has a more complex interaction but is predominantly excreted by tubular secretion. 99mTc-MAG3 may also be excreted by the hepatobiliary system in renal failure, with advantages over99mTcDTPA due to a lower radiation dose and better images both in children and in renal failure.
Fig. 15.5 A heavily T2-weighted coronal MR image, demonstrating a dilated renal pelvis and ureter to the level of an obstructing transitional cell carcinoma (TCC) of the distal ureter in a nonfunctioning left kidney (open arrow). A ureteric stent (solid arrow) is present in the left ureter
Staging of pelvic cancers |
Characterization of renal masses when CT and ultrasound are equivocal |
MR urography |
MR angiography |
Long-term follow-up of renal masses in von Hippel–Lindau and tuberous sclerosis |
Detection of undescended testes |
Urethral diverticulum |
Total renal function | GFR using 51Cr-EDTA |
Divided renal and intrarenal distribution of function | Static 99mTc-DMSA scan |
Perfusion, transit time, and outflow kinetics | Dynamic 99mTc-DTPA or 99mTc-MAC3 scan ± frusemide |
Renovascular hypertension | Dynamic 99mTc-DTPA or 99mTc-MAC3 scan before and after captopril |
VUR | Dynamic 99mTc-DTPA or 99mTc-MAC3 scan followed by indirect cystography or 99mTc-pertechnetate direct cystogram |
Bone imaging, e.g. for bone metastases | 99mTc-methylene diphosphonate |
Fig. 15.6 A99mTc-DMSA examination demonstrating a small, scarred right kidney and normal left kidney
The appropriate radioisotope is injected as an intravenous bolus and posterior images are acquired dynamically over 20–40 minutes (anterior images are obtained in renal transplant patients). An ROI is drawn around the renal outlines and after background correction, a time-activity curve or renogram is generated with “counts” in the ROI plotted against time. The reno-gram is divided into three phases: The first two to three minutes represent the vascular phase, followed by the secretory and excretory phases. By examining the different components of the curves, a picture of renal perfusion, excretion, and drainage may be obtained.
As renal uptake or function is proportional to renal blood flow, divided renal function may also be calculated by assessing the relative heights or rate of rise of the curves in the first two to three minutes before any radioisotope has left the kidneys. Individual relative function is expressed as a percentage.
Diuresis Renography
Urinary flow rate can influence the appearance of the renogram. If the kidney demonstrates pelvicalyceal dilatation, it may be necessary to challenge the kidney with significantly raised flow rates by administering a diuretic, such as frusemide (furosemide). The diuresis will promote washout of tracer in a nonobstructed system depicted by a fall in the time-activity curve. If the pelvicalyceal system is truly obstructed, diuresis will increase the dilatation with a continued rise in the slope of the time-activity curve. Different protocols for diuretic administration have been described with the original account specifying injection 20 minutes after the radioisotope to allow for assessment of the unmodified upper tracts and alleviate the need for diuretic administration if normal. If pelvicalyceal dilatation is present, maximal flow rates will be required and injection 15 minutes prior to radionuclide administration (F-15 protocol) is more appropriate, allowing dynamic imaging to commence at peak diuresis.
Captopril Renography
Following the initial description in 1983 by Majd et al., renograms performed before and after administration of 25–50 mg of captopril may be used to demonstrate RAS.1 This relies on the presence of high levels of angiotensin II in renovascular hypertension. Angiotensin-converting enzyme (ACE) inhibitors block the vasoconstricting effect that maintains GFR in RAS and the second time-activity curve will show a reduction in perfusion and a delay to peak. A change in split function can also be seen. Sensitivities and specificities vary according to the study population, but a prospective multi-center trial evaluated 380 hypertensive patients, who had a99mTc-DTPA examination before and after administration of captopril; 125 of the study group subsequently had a significant RAS at renal angiography.2Overall RAS of > 70% was detected with a sensitivity of 83% and specificity of 93% if renal function was normal. The performance of the test decreased in impaired renal function.
Direct and Indirect Cystography
As an adjunct to renography, a voiding study may subsequently be performed in toilet-trained children. The child sits with their back against the gamma camera head with the kidneys and bladder in the field of view and voids while fast frames are acquired dynamically. VUR can be seen as an increase in “counts” over the kidney or ureters (Fig. 15.7). Direct cystography is more invasive, requiring catheterization of the bladder and instillation of99mTc-pertechnetate with a further disadvantage of the lack of visualization of the posterior urethra in males. However, direct99mTc-pertechnetate cystography gives a lower radiation dose than a MCU and can be useful in the follow-up of children under age three.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree