CHAPTER 13
Noninflammatory Joint Disease

This chapter covers joint diseases that have predominantly noninflammatory features on radiographs.
 OSTEOARTHRITIS
OSTEOARTHRITIS
Osteoarthritis (degenerative joint disease) is a form of joint disease characterized by degenerative changes involving synovial joints. Osteoarthritis can be divided into primary and secondary types, but the division is artificial: The underlying cause is evident in secondary osteoarthritis but not in primary or idiopathic osteoarthritis. The distinction has some practical value in understanding the process and planning clinical management. Osteoarthritis is the most common form of arthritis. Its prevalence increases with age, so that osteoarthritis is nearly ubiquitous in patients older than 65 years of age. Up to 45 years of age, it is more prevalent in men; from 45 to 55 years of age, the prevalence is equal; and after 55 years of age, it is more prevalent in women. The most common presentation of osteoarthritis is joint pain and limitation of activity. Laboratory tests are used to eliminate other forms of arthritis as clinical possibilities.
Primary Osteoarthritis
The early morphologic abnormality in primary osteoarthritis is fibrillation of the articular cartilage. The surface develops fibril-like projections and becomes irregular. Underlying this morphologic change is disruption at the molecular level of the superficial armor-plate layer and collagen framework, resulting in progressive loss of proteoglycans from the ground substance and collagen from the framework. Chondrocytes increase protein synthesis, presumably in response to the continuing loss of structural components. Progressive erosion and formation of fissures in the surface eventually expose the subchondral bone. The initial event that incites fibrillation of the cartilage surface is unknown; some forms of primary osteoarthritis may result from an initial alteration in articular cartilage physiology.
Radiographic findings do not appear in osteoarthritis until articular cartilage loss results in secondary adaptive changes in bone. These findings include uneven loss of articular space, subchondral sclerosis, osteophytes, and subchondral cysts (Fig. 13.1); the absence of osteoporosis, ankylosis, and erosions is characteristic. Osteophytes tend to be largest in the plane of motion; therefore, osteophytes at the distal interphalangeal (DIP) and proximal interphalangeal (PIP) joints are best seen on the lateral view. In the hand and wrist, primary osteoarthritis typically affects the DIP and PIP joints and the basal joints of the thumb (Fig. 13.2). The basal joints of the thumb are composed of the first carpometacarpal (CMC) joint and the scaphoid-trapezium-trapezoid joints. Isolated degenerative involvement at this specific site is virtually diagnostic of primary osteoarthritis. The first metatarsophalangeal (MTP) joint, hips and knees, and the cervical and lumbar spine are also common sites of involvement. The metacarpophalangeal (MCP) joints, wrist, elbow, shoulder, and ankle are typically spared. The severity of radiographic changes does not necessarily correlate with the severity of symptoms.
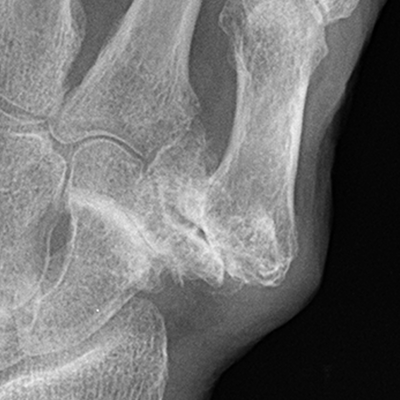
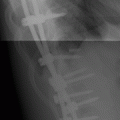
FIGURE 13.1. Osteoarthritis at the first CMC and scaphotrapezial joints with narrowing of the articular spaces, osteophyte formation, subchondral sclerosis, and subluxation.
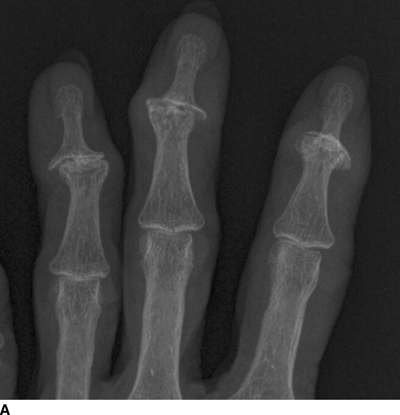
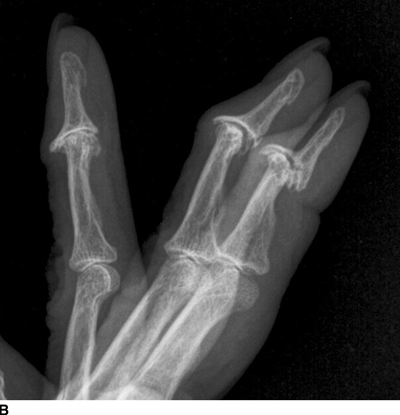
FIGURE 13.2. Osteoarthritis at the DIP joints of the fingers. A: PA radiograph. B: Lateral radiograph.
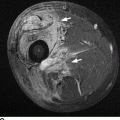
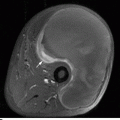
In the knee, the characteristic distribution of involvement is in the medial compartment and, to a less severe degree, the patellofemoral compartment. Joint space narrowing, subchondral sclerosis, osteophytes, and subchondral cysts are typical findings (Fig. 13.3). Occasionally, more severe involvement of the lateral or patellofemoral compartments occurs. Angular deformities and joint space narrowing are best demonstrated on standing views. Because the severity of involvement of the anterior and posterior portions of the femoral cartilage is typically uneven, the amount of joint space narrowing may vary between radiographs with the knee in extension and flexion. On MRI, early osteoarthritis is evident as abnormal high signal in articular cartilage on T2-weighted MRI. When isolated to the patella, this condition is called chondromalacia patellae (Fig. 13.4). Fibrillation of the cartilage surface, thinning of the cartilage, and frank loss of cartilage may be seen in progressively more severe cases. Subchondral bone edema at sites of cartilage loss, osteophyte formation, loose bodies, and effusions may be present in established osteoarthritis (Figs. 13.5–13.7).
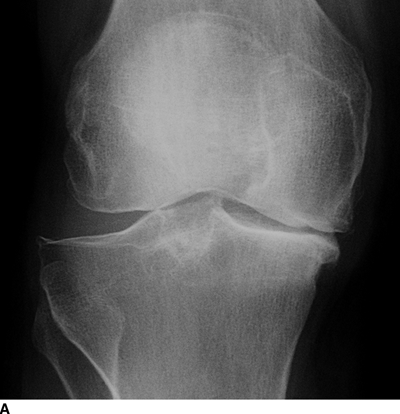
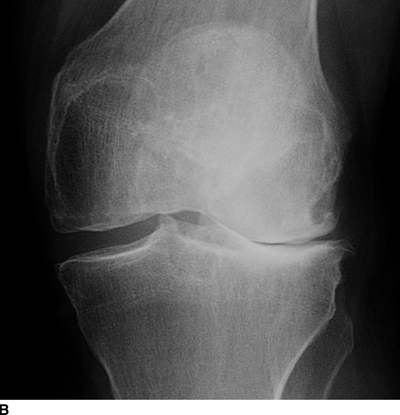
FIGURE 13.3. Osteoarthritis with asymmetric joint space narrowing, osteophytes, and subchondral sclerosis. A: Right knee. B: Left knee.
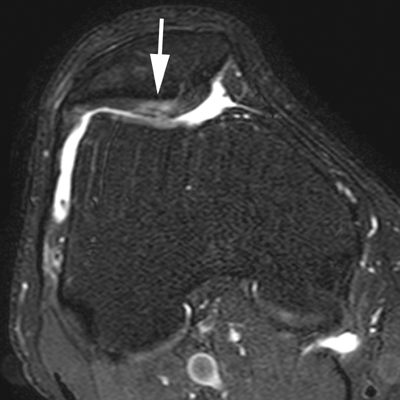
FIGURE 13.4. Chondromalacia patellae with effusion as well as fissured and fibrillated patellar cartilage (arrow) shown on axial T2-weighted, fat-saturated MRI.
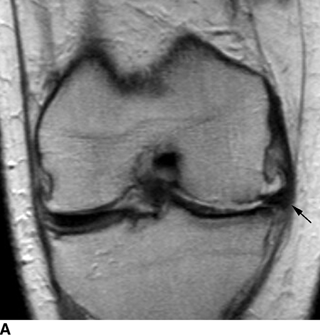
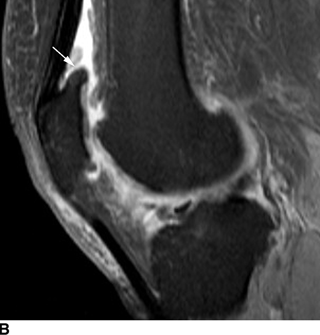
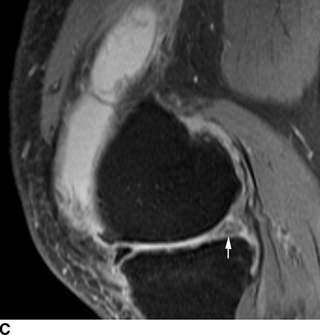
FIGURE 13.5. Osteoarthritis. A: Coronal T1-weighted MRI shows cartilage loss and meniscal extrusion (arrow) in the medial compartment. Medial and lateral compartments are involved by osteophytes. B: Sagittal T2-weighted fat suppressed MRI shows patellofemoral compartment osteophytes (arrow) and cartilage loss. C: Sagittal T2-weighted fat suppressed MRI through the medial compartment shows effusion, cartilage loss, osteophytes, and degenerative posterior horn medial meniscal tear (arrow).
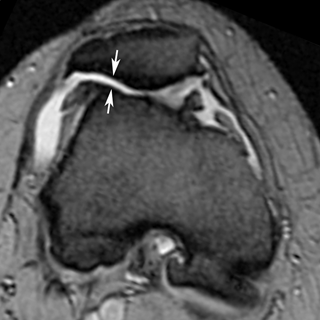
FIGURE 13.6. Patellofemoral osteoarthritis. Axial gradient echo MRI shows loss of cartilage from the lateral facet of the patella and the patellofemoral groove (arrows). Osteophytes are present on both sides of the patellofemoral joint and the posterior margin of the lateral condyle. An effusion is present.
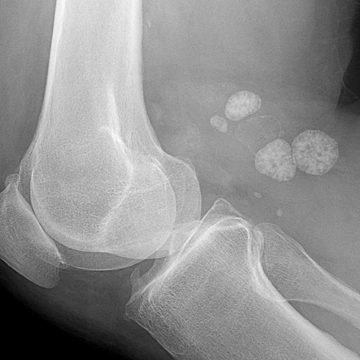
FIGURE 13.7. Osteoarthritis of the knee with large, calcified intra-articular bodies in a popliteal cyst.
In the hip, loss of articular space is usually found along the superior (horizontal) portion of the joint (Fig. 13.8). Less commonly, the medial joint space is narrowed. Osteophyte formation in the femoral head often forms a collar of bone around the femoral neck at the margin of the articular surface, usually seen best on frog lateral views. As with the knee, uneven involvement of the articular cartilage results in varying amounts of joint space narrowing from position to position. Mapping cartilage thickness with fluoroscopically positioned spot radiographs or cartilage-specific imaging parameters on MRI can be helpful in planning rotational osteotomies for treatment.
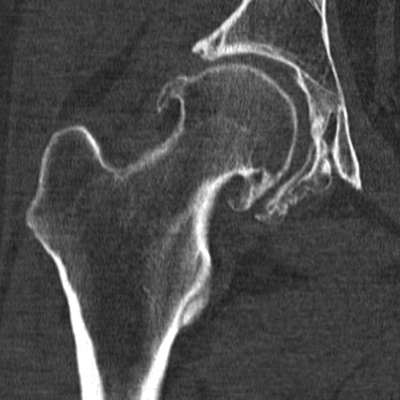
FIGURE 13.8. Osteoarthritis of the hip with advanced joint space asymmetric narrowing and prominent osteophytes.
Osteoarthritis of the synovial joints of the spine may be the predominant feature of degenerative spine disease or may occur in association with other features such as degenerative disk disease, previous trauma, scoliosis, kyphosis, or vertebral anomalies. The common sites of synovial joint osteoarthritis are the lower cervical and lower lumbar spine. The atlantoaxial joint is also synovial and may be affected. The pathologic process is identical to that of other synovial joints, leading to joint space narrowing, subchondral sclerosis, and osteophytes. Loss of articular cartilage may allow subluxation or excessive motion; bony hypertrophy may reduce motion. Osteophytes and ligamentous thickening may lead to nerve root involvement. These findings are best demonstrated by CT (Fig. 13.9).
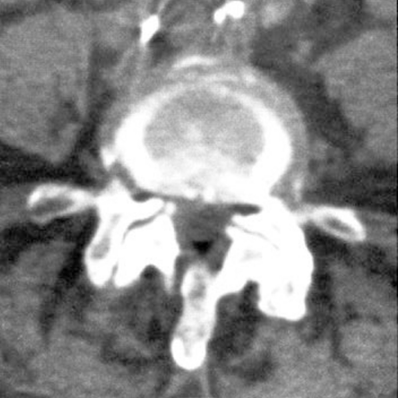
FIGURE 13.9. Lumbar facet osteoarthritis on CT scan. Osteophytes extend into the lateral recesses bilaterally, causing nerve root compression.
Osteoarthritis of the acromioclavicular joint is a common finding on shoulder imaging. On radiographs, osteophytes may be seen. On MRI, osteophytes and hypertrophy of the joint are typical, often with subchondral edema in the distal clavicle and acromion and fluid in the joint (Fig. 13.10).
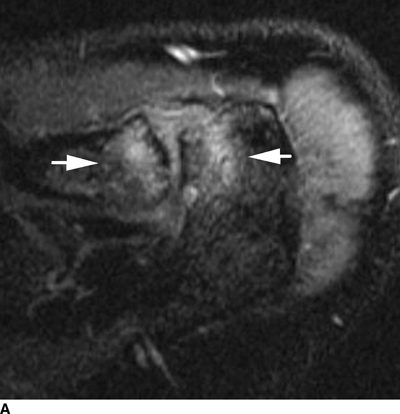
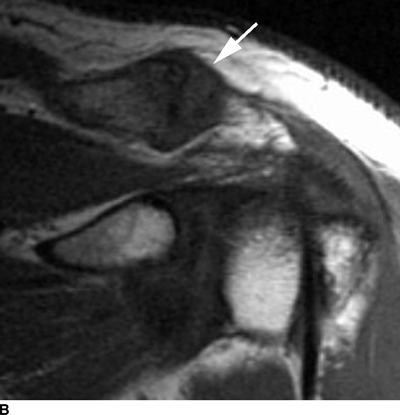
FIGURE 13.10. Osteoarthritis of the acromioclavicular joint. A: Axial T2-weighted fat suppressed MRI shows subchondral edema in the clavicle and acromion (arrows). B: Oblique coronal T1-weighted MRI shows hypertrophy of the acromioclavicular joint (arrow) with mass effect on the supraspinatus muscle.
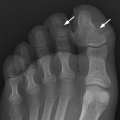
Inflammatory (erosive) osteoarthritis is a condition in which an acute synovitis accompanies primary osteoarthritis. Although joint degeneration always has some component of synovial inflammation because of the presence of joint debris and cartilage breakdown products, the inflammation dominates the clinical presentation in erosive osteoarthritis. Radiographs show the degenerative features and distribution of primary osteoarthritis, but the acute synovitis causes inflammatory erosions, uniform joint space narrowing, and sometimes ankylosis (Fig. 13.11). A characteristic “seagull” appearance may be seen on PA radiographs at the interphalangeal (IP) joints of the fingers, corresponding to central erosions and bony hypertrophy. The typical age at onset is in the fifth or sixth decade, and women are affected far more frequently than men. The inflammation usually subsides within a few months to a couple of years, leaving the degenerative changes. In the hand, erosive osteoarthritis characteristically affects the DIP and PIP joints and the basal joints of the thumb, as does nonerosive primary osteoarthritis.
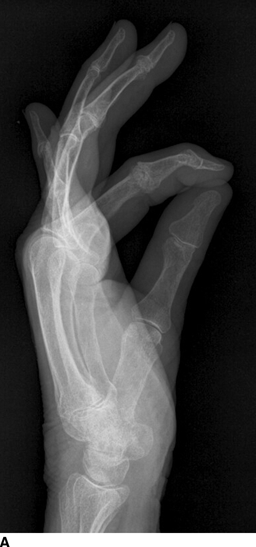
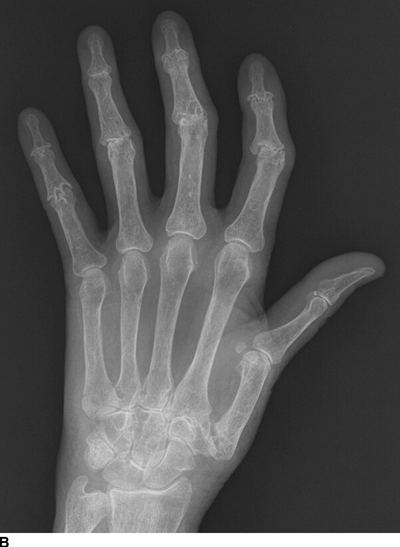
FIGURE 13.11. Inflammatory osteoarthritis with erosions and osteophytes at the PIP joints and the first CMC joint. A: Lateral radiograph. B: PA radiograph.
Secondary Osteoarthritis
Secondary degenerative changes in the joints result from three major factors: an abnormality of the articular cartilage, loss of subchondral bony support beneath normal articular cartilage, and abnormal alignment and mechanical stress. Any condition with one of these features may lead to permanent, progressive osteoarthritis. Secondary osteoarthritis may follow inflammatory arthritis if the inflammatory process has caused permanent cartilage damage and is quiescent long enough for the degenerative changes to develop. Mechanical trauma may injure the articular cartilage, which has a limited ability for repair. A fibrocartilage scar may replace damaged areas of hyaline cartilage. Joint debris, loose bodies, or displaced meniscal fragments within a joint may erode the articular cartilage. Osteochondral loose bodies derive nutrition from synovial fluid and may grow. Healthy cartilage wears prematurely when its underlying bony support is lost. For example, collapse of the subchondral bone of the femoral head after osteonecrosis leads rapidly to secondary degeneration. Less obvious changes in the subchondral bone due to repetitive subclinical trauma may also lead to osteoarthritis. An abnormally aligned joint or a joint that is subject to mechanical disadvantage or abnormal stresses may wear prematurely. Posttraumatic osteoarthritis may follow malunion of long-bone fractures, imperfectly reduced intra-articular fractures (Fig. 13.12), or posttraumatic joint instability. Many forms of developmental and acquired bone and joint dysplasia lead to early osteoarthritis, including developmental dysplasia of the hip (Fig. 13.13), Legg-Calvé-Perthes disease, and multiple epiphyseal dysplasia. The premature wear-out resulting from the abnormal joint geometry worsens in a vicious cycle of progressive malalignment, mechanical disadvantage, and abnormal stress.
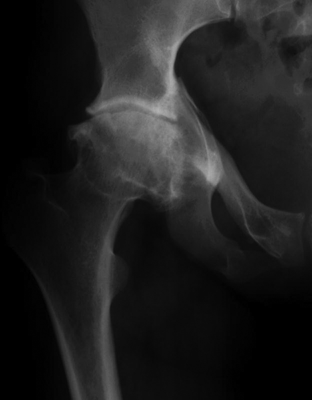
FIGURE 13.12. Posttraumatic osteoarthritis 12 years after acetabular fracture.
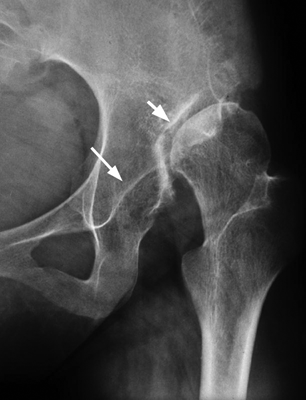
FIGURE 13.13. Secondary osteoarthritis in the left pseudoacetabulum (short arrow) in a 20-year-old woman with untreated DDH. The native acetabulum (long arrow) is empty.
Femoroacetabular Impingement
Femoroacetabular impingement designates a spectrum of developmental or acquired conditions of the hip in which a morphologic mismatch between the femoral head and the acetabulum hip may lead to mechanical impingement of the acetabular rim on the femoral neck at the extremes of motion, restricting range of motion, causing pain, and leading to acetabular labral pathology and degenerative changes. In the cam type of femoroacetabular impingement, the morphology of the femoral head and neck is abnormal as a result of conditions such as previous trauma, slipped capital femoral epiphysis, or Legg-Perthes disease. In the pincer type of femoroacetabular impingement, the morphology of the acetabulum is abnormal as a result of conditions such as acetabular retroversion or protrusio acetabuli. Patients who may be anatomically predisposed to femoroacetabular impingement may be asymptomatic if they do not engage in activities that require extreme ranges of motion, such as ballet or yoga. Radiologic features of femoroacetabular impingement include reactive changes in the femoral neck (Fig. 13.14) and evidence of abnormal femoral or acetabular morphology.
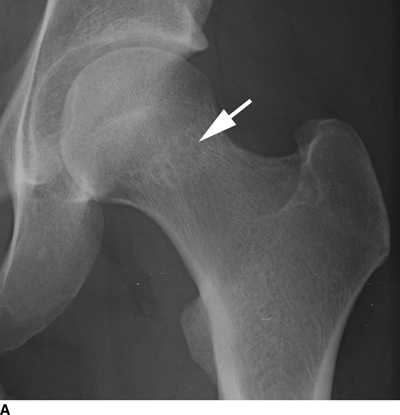
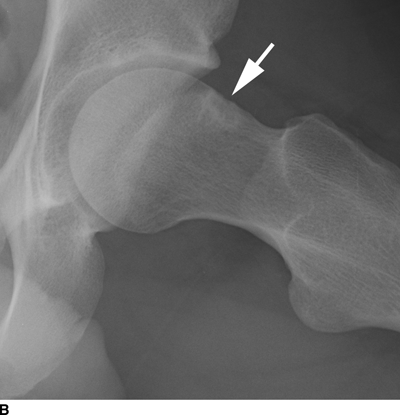
FIGURE 13.14. Femoroacetabular impingement with reactive bone changes at the femoral neck (arrows). A: AP radiograph. B: Frog lateral radiograph.
Subacromial Impingement
Subacromial impingement syndrome of the shoulder refers to trapping of the rotator cuff between the top of the humerus and the undersurface of the coracoacromial arch. The coracoid process, the coracoacromial ligament, and the acromion process form the coracoacromial arch. A downward-projecting bony excrescence or degenerative hypertrophy of the acromioclavicular joint is commonly associated with impingement syndrome (Fig. 13.15). However, impingement syndrome is considered to be a clinical rather than a radiologic diagnosis. Other conditions that may impinge on the rotator cuff include congenital, developmental, or acquired variations in the size and shape of structures around the coracoacromial arch, including the acromion and the coracoid. Other types of shoulder impingement include posterior superior glenoid impingement and subcoracoid impingement.
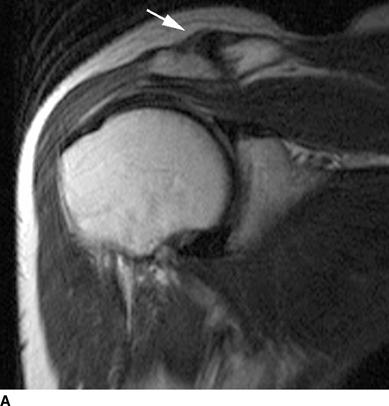
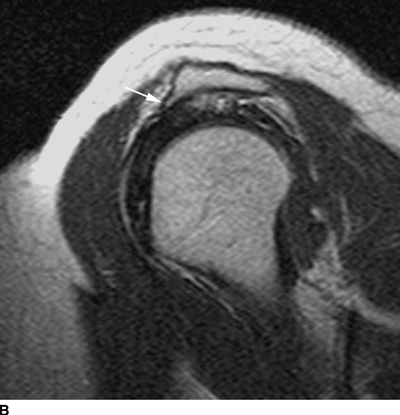
FIGURE 13.15. Rotator cuff impingement. A: Oblique coronal T1-weighted MRI shows osteoarthritis of the acromioclavicular joint with hypertrophy (arrow) and mass effect on the supraspinatus muscle. B: Oblique sagittal T1-weighted MRI shows anterior hooking (arrow) of the acromion process.
Neuropathic Osteoarthropathy
Neuropathic joints (Charcot joints) have lost proprioception and deep pain sensation. With continued use of the joint, relaxation and hypotonia of the supporting structures lead to malalignment and recurrent injury. Rapidly progressive erosion of the articular cartilage, reactive subchondral sclerosis, fractures, and fragmentation of the subchondral bone result in a disorganized joint. The presence of joint debris induces synovitis and chronic effusion. The damage and derangement may occur over a period of days to weeks with relatively little symptomatology. Both lower motor neuron (peripheral) and upper motor neuron (central) lesions may result in neuropathic osteoarthropathy. Diabetic neuropathy is the most common lower motor neuron lesion causing neuropathic osteoarthropathy; other causes include alcoholism, tuberculosis, amyloidosis, leprosy, peripheral nerve trauma, steroids, and congenital indifference to pain. Syringomyelia is the most common upper motor neuron lesion; other causes include meningomyelocele, trauma, multiple sclerosis, tabes dorsalis (syphilis), and cord compression.
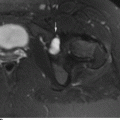
Neuropathic osteoarthropathy occurs in 0.1% of all diabetics and in 5.0% of those with diabetic neuropathy. Diabetic peripheral neuropathy causes loss of pain sensation and proprioception, leading to exceptional wear and tear without patient awareness of injury. The most frequent site of involvement is the foot (80%), especially the tarsometatarsal, intertarsal, and MTP joints; involvement may be unilateral or bilateral. Tarsal-metatarsal fracture dislocation (Lisfranc fracture dislocation) may occur spontaneously or with minimal trauma. Extensive sclerosis, osteophytosis, fractures, bony fragmentation, subluxation, dislocation, bony debris, effusion, and subchondral cysts are common findings. Chronic osteomyelitis is also relatively common in the diabetic foot, and the possible combination of neuropathic osteoarthropathy with infection can pose a diagnostic dilemma (Fig. 13.16). MRI with gadolinium enhancement may be helpful in this circumstance.
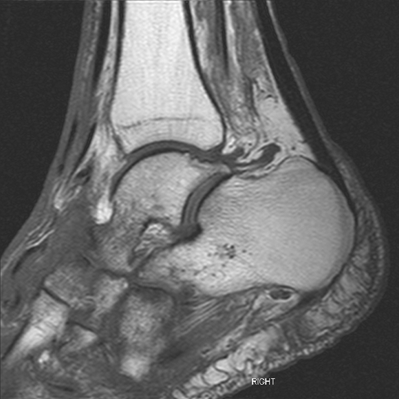
FIGURE 13.16. Neuropathic osteoarthropathy. Sagittal T1-weighted MRI of the foot shows swelling, disorganization, and edema of the midfoot.
Neuropathic osteoarthropathy occurs in 25% of patients with syringomyelia. The joint changes are usually in the upper extremity (80%), and they may be atrophic rather than proliferative. Acute resorption of the articulating ends of the bone without evidence of repair, gross soft-tissue swelling, and bony debris in the soft tissues are common findings. This process may mimic destruction from tumor or infection. The most commonly involved joint is the shoulder (Fig. 13.17).
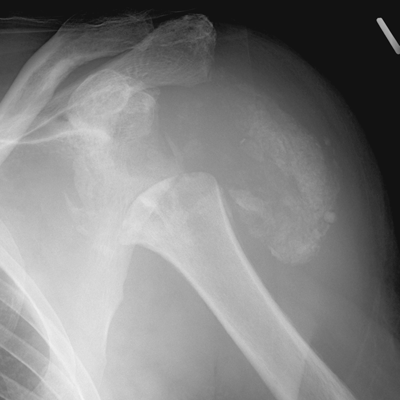
FIGURE 13.17. Neuropathic shoulder joint in a patient with syringomyelia.
 CRYSTAL-ASSOCIATED DISEASES
CRYSTAL-ASSOCIATED DISEASES
Crystal-associated joint diseases are pathologic conditions that occur in the presence of crystals. The crystals contribute to tissue damage, but the causal relationship between crystals and tissue damage is not well understood. Crystals precipitate from the extracellular fluid space into the articular tissues, where they accumulate. Deposits of crystals may then be shed episodically into the joint space. Clearance of crystals from the joint space and articular cartilage is poor because these structures are avascular, alymphatic, and largely devoid of scavenger cells. The presence of particles alters the mechanical properties of the tissues, tending to make them less elastic. Articular cartilage is particularly vulnerable to damage and ultimately undergoes degenerative changes. Large crystalline particles in the joint space can cause direct abrasive damage to the articular surfaces. Small particles can cause damage by biophysical and biochemical interactions with cell membranes and macromolecules and may also provoke an acute synovitis. Although the precise mechanisms mediating acute synovial inflammation are incompletely understood, different mechanisms appear to be activated by different crystals. These crystal-induced inflammatory reactions tend to have a sudden onset and a rapid, self-limited course. The sudden onset is probably related to the abrupt shedding of crystals into the joint space from a deposit in the articular tissues.
Crystal deposition diseases have three clinical presentations: (a) an asymptomatic state in which crystals can be detected, (b) an inflammatory arthritis, and (c) a chronic destructive arthropathy. The particular diseases are defined by the presence of characteristic crystals within affected joints. Aspiration of the joint during an acute inflammatory episode may yield material in which the associated crystal can be demonstrated. The three types of crystals that are commonly associated with joint diseases are calcium pyrophosphate dihydrate (CPPD), calcium hydroxyapatite, and monosodium urate monohydrate (Table 13.1).
TABLE 13.1 Crystal Deposition Diseases of Joints
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree