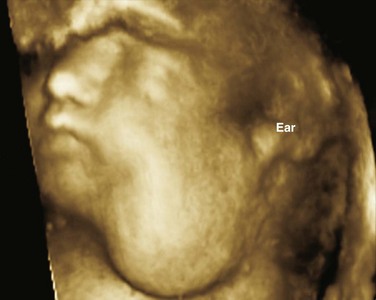
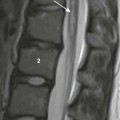
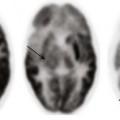
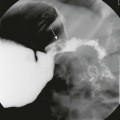
Chapter 14 Color Doppler provides valuable characterization of a lesion’s vascularity and blood supply, detects possible arteriovenous shunting, and depicts the major neck vessels. Three-dimensional reconstruction clearly depicts the relationship of cervical lesions with adjacent facial and skull base structures (e-Fig. 14-1). Potential shortcomings of ultrasonography involve cases with poor acoustic windows because of an anterior position of the placenta, maternal obesity or polyhydramnios, and abnormal fetal position with unusual flexion and extension of the fetal neck. Useful MRI sequences for fetal neck imaging include the following: 1. Ultrafast spin echo T2-weighted sequences are excellent for imaging of normal anatomical structures and depiction of cervical lesions. 2. Spoiled gradient echo T1-weighted imaging is helpful for imaging of the thyroid gland and blood flow within the major cervical vessels, as well as for depiction of intralesional blood products and calcifications. 3. Gradient echo planar imaging is helpful for detection of blood products, calcifications, and bony details. 4. Steady state or balanced gradient echo sequences are valuable in the prenatal assessment of preservation or impairment of swallowing in the fetus (Video 14-1). The details of fetal airway compromise secondary to the presence of a cervical mass should always be carefully examined. MRI sequences for imaging of neonatal neck lesions include the following: 1. High resolution 2- to 3-mm T1- and T2-weighted and postcontrast imaging with fat-suppression in orthogonal planes 2. Ultrafast spin echo imaging for optimal visualization of thin fluid–filled sinus tract or fistula 3. Diffusion-weighted imaging, which is particularly useful for identification of hypercellular tumors and infected cystic lesions with viscous, purulent content 4. Magnetic resonance angiography, which may provide crucial information regarding vascular anatomy On cross-sectional imaging, it appears as a lobulated mass with intense uniform enhancement, which usually demonstrates intralesional flow voids on MRI (Fig. 14-2, A). Proliferation may be biphasic. Careful assessment of upper airway status is important because the deep portions of a cervical hemangioma may enlarge sufficiently at this stage to impinge on the airway and cause obstruction. During the final involution phase, which may continue for up to 12 years, the rate of regression is variable. Involution is never complete, although the residual lesion may not be grossly visible; in the neck, it leaves persistent fibrofatty tissue. Infantile hemangiomas are positive for the glucose transporter 1 (GLUT-1) marker at all stages of lesion development. Figure 14-2 Infantile hemangiomas. Upper airway hemangiomas (see Fig. 14-2, B and C) become symptomatic in the proliferative phase, causing biphasic stridor, respiratory distress, and potentially even life-threatening airway obstruction, which may require emergent intubation. These hemangiomas are often multifocal and occur above and below the vocal cords. Close monitoring with cross-sectional imaging may be indicated because laryngoscopy may not accurately depict the degree of invasion. MRI is preferred in the setting of intubation or tracheostomy. In rare cases, cervicofacial infantile hemangioma may be associated with central nervous system hemangiomas in intracranial (see Fig. 14-2, D) or intraspinal locations. The reported cases of central nervous system hemangiomas indicate a specific pattern of distribution, with a predilection for the basal cisterns, ventricular system, and extradural spinal involvement.
Prenatal, Congenital, and Neonatal Abnormalities
Imaging Modalities
Ultrasonography
Magnetic Resonance Imaging
Vascular Tumors
Infantile Hemangioma
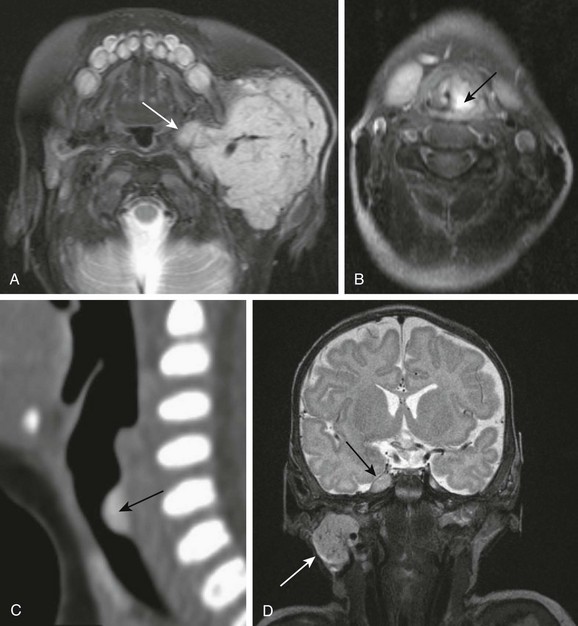
A, Parotid infantile hemangioma. A T2-weighted axial image with fat suppression shows a well-circumscribed lobular soft tissue mass in the left parotid with intrinsic flow voids and deep extension into the ipsilateral parapharyngeal space (arrow). The upper airways remain patent. B and C, Upper airways infantile hemangiomas. A T2-weighted axial image with fat suppression shows a small hyperintense hemangioma in the left vocal cord (black arrow), causing severe airway effacement (B). A sagittal reconstructed computed tomography image shows a small lobulated enhancing subglottic hemangioma (black arrow) (C). D, Multifocal infantile hemangiomas with intracranial lesions. A T2-weighted coronal image with fat suppression shows cervical (white arrow) and Meckel cave (black arrow) hemangiomas. (C, Courtesy Avrum N. Pollock, MD.)
Prenatal, Congenital, and Neonatal Abnormalities