Chapter 60 The pleura is a serous membrane consisting of two layers the visceral and the parietal pleura. The parietal pleura covers the inner aspect of the chest wall and the diaphragm. The visceral pleura is strongly adherent to the surface of the lungs and interlobar fissures. In healthy persons, the space between the visceral and parietal pleura is nothing more than two apposed pleural surfaces separated by a trace of glycoprotein-rich fluid. In normal conditions, no imaging study can visualize the pleural space because pleural membranes are only 0.2 to 0.4 mm thick, and the physiologic volume of pleural fluid forms a thin 5- to 10-micron layer. However, in children, various pathologic processes can occur within this space that require imaging evaluation. In this chapter, etiologies, imaging characteristics, treatment, and follow-up of pathologic processes involving the pleura are discussed.1–4 Etiology: The lymphatic system, which is part of the immune system and comprises a network of lymphatic vessels, plays an important role in the homeostasis of pleural fluid. Excess production or decreased absorption of lymphatic fluid can result in an increased volume of pleural fluid.1,5 Pleural effusions may be caused by systemic diseases or local infectious/inflammatory processes. Pleural effusions traditionally are classified into two types: transudates and exudates. Transudates usually result from underlying systemic diseases that alter the normal balance of formation and absorption of pleural fluid. Transudative pleural effusions commonly are seen in patients with nephrotic syndrome, heart failure, and cirrhosis. Exudates typically are caused by local infectious or inflammatory processes that result in the increased formation of pleural fluid. Exudative pleural effusions usually are seen in patients with pulmonary infections, neoplasms, hemothorax, and collagen vascular diseases.3–5 Causes of pleural effusion in pediatric patients differ substantially from those in adults. Whereas the most frequent cause of effusions in adults is congestive heart failure, pleural effusions in children are most commonly a result of underlying pleuropulmonary infection (Table 60-1).3 Although pediatric patients with small pleural effusions are usually asymptomatic, large pleural effusions can result in symptomatic respiratory distress or inspiratory pleuric chest pain as a result of stretching of the parietal pleura.3 When infection is the underlying cause, the predominant symptoms are cough, dyspnea, fever, and elevated white blood cell counts.1,3 Table 60-1 Imaging: The first-line imaging modality, chest radiography, plays an important role in the initial diagnosis of pleural effusions in children. The radiographic appearance of pleural effusions depends on the volume and consistency of the pleural fluid, the patient’s position, and the presence of septations and/or loculations. On chest radiographs, a free-flowing effusion forms an internally concave meniscus paralleling the chest wall that changes in position on lateral decubitus radiographs. On supine radiographs, a mobile fluid may layer over the hemithorax, resulting in a diffuse opacity. The presence of a lentiform-shaped effusion with internal convex margins is suggestive of a loculated pleural effusion.2,6 The most common indication of chest ultrasound in children is to characterize an opaque hemithorax seen on a chest radiograph (Fig. 60-1).6,7 Ultrasound is the best imaging modality in diagnosing and characterizing pleural fluid.7 Ultrasound is more sensitive than radiographs in detecting pleural fluid, particularly when the amount is small (~5 mL).2,8 Figure 60-1 Simple effusion in a 2-year-old boy. The internal echogenicity of pleural effusions on ultrasound can be categorized into four types (Table 60-2): (1) homogeneously anechoic (see Fig. 60-1, B); (2) nonseptated with internal low-level echoes (Fig. 60-2); (3) septated (Fig. 60-3, A); and (4) homogeneously echogenic (Fig. 60-4, A).7,9 Anechoic and free-flowing effusions are called simple pleural effusions, whereas the remainder are classified as complex pleural effusions. Figure 60-2 Complex effusion with low-level echoes in a 9-year-old girl with bacterial pneumonia. Figure 60-3 Complex septated effusion in a 6-year-old patient with bacterial pneumonia. Figure 60-4 Complex effusion in a 6-year-old patient with bacterial pneumonia. Computed tomography (CT) also is more sensitive than chest radiographs in detecting small pleural effusions. The advantage of CT is that it provides an unobstructed view of the entire underlying lung parenchyma, mediastinum, and chest wall; however, it is associated with ionizing radiation exposure. Intravenous administration of contrast material is necessary to optimally visualize and assess the pleura and lung parenchyma (Fig. 60-4, B). In comparison to ultrasound, CT is limited in characterizing pleural effusions and demonstrating internal septations (see Fig. 60-3).2 Treatment and Follow-up: The main treatment of choice for pleural effusions is thoracentesis, which can be helpful in evaluating the underlying cause of the pleural effusion and also in relieving symptoms. Indications for thoracentesis in children with pleural effusions are persistent fever, respiratory compromise, mediastinal shift, pleuritic pain, and underlying lung disease.3 The management of parapneumonic effusion is discussed in the following section. Etiology: A parapneumonic pleural effusion refers to a pleural fluid collection in association with an underlying pneumonia, whereas empyema is the presence of pus in the pleural space.1,10 Streptococcus pneumoniae is still the main etiological agent in parapneumonic effusions in children, although the number of cases caused by Staphylococcus aureus has increased.1,10 More recently, the Streptococcus milleri group, typically found in the oropharynx and upper respiratory and gastrointestinal tracts, has been recognized as an important pathogen causing purulent pleuropulmonary infections, especially in immunocompromised patients.11 Pleural infection can be classified into three phases based on the progression of infection: exudative, fibrinopurulent, and organizing (Box 60-1). During the exudative phase, the inflammatory process associated with the underlying pneumonia leads to the accumulation of free-flowing clear fluid within the pleural cavity. During the fibrinopurulent phase, deposition of fibrin in the pleural space results in septations and loculations, limiting the flow of pleural fluid. The fibrinopurulent phase may be divided into two stages: (1) early fibrinopurulent stage A and (2) late fibrinopurulent stage B. The early fibrinopurulent stage A is characterized by an increase in white blood cell accumulation, which causes thickening of the pleural fluid consistency. The late fibrinopurulent stage B is defined as the accumulation of pus in the pleural space. The organizing phase begins when fibroblasts infiltrate the pleural cavity, forming a thick rigid pleural rind. This pleural rind prevents lung reexpansion, impairs lung function, and creates a persistent pleural space with ongoing potential for infection. At this stage, spontaneous healing eventually may occur, or a chronic empyema may develop.1,6,12 Imaging: A chest radiograph is the initial imaging modality for assessing the presence of parapneumonic effusions. Ultrasound subsequently can be obtained for characterization of the parapneumonic effusion. In most cases, parapneumonic effusions are exudates and therefore are complex in appearance on ultrasound, although they may be simple in appearance during the early stage.9,13 As the underlying infection progresses, parapneumonic effusions may develop low-level internal echoes and floating debris (see Fig. 60-2) followed by septations and loculations (see Fig. 60-3, A), preventing free motion of the fluid. The internal echoes indicate increased underlying cellularity of fluid; these pleural effusions are usually exudates but not necessarily empyemas.1,6,7,9,13 The parapneumonic effusion eventually may become semi-solid in appearance on ultrasound (see Fig. 60-4, A). The development of pleural thickening indicates the presence of a fibrous capsule. The routine use of CT is not necessary to evaluate parapneumonic pleural effusions, particularly in pediatric patients, because it entails exposure to ionizing radiation.1,6,14 However, CT with intravenous contrast may aid in the evaluation of the lung parenchyma when a complication such as a bronchopleural fistula is suspected (Fig. 60-5) or in the differentiation of a parapneumonic effusion from a lung abscess.2
The Pleura
Pleural Effusion
Phase
Description
Exudative
Pleural inflammation leads to accumulation of free-flowing clear liquid
Fibrinopurulent
Deposition of fibrin in the pleural space results in septation/loculations limiting the flow of fluid; pus accumulates in the pleural cavity
Organizing phase
Fibroblasts infiltrate the pleura, forming a rigid pleura and preventing lung expansion
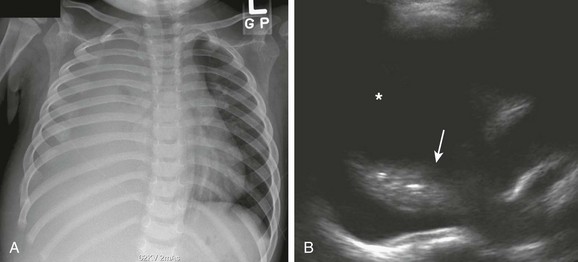
A, Complete opacification of the right hemithorax with obliteration of the right side of the mediastinum and hemidiaphragm. The cardiac and tracheal deviations to the left are imaging findings suggestive of a pleural effusion. B, Ultrasound of the right hemithorax confirms the presence of a large simple pleural effusion (asterisk). Consolidation (arrow) of the underlying lung also is seen.
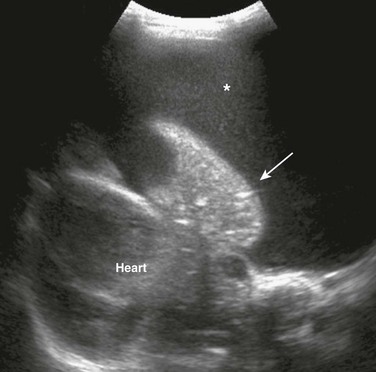
An ultrasound image of the left hemithorax reveals a large pleural effusion with floating low-level echoes indicating debris (asterisk) and the consolidated underlying lung (arrow).
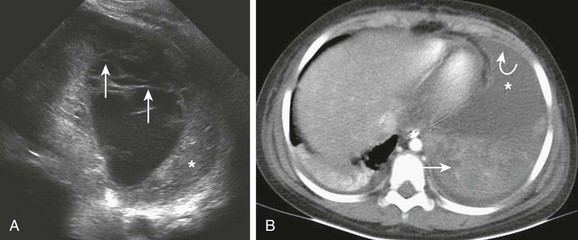
A, An ultrasound image of the left hemithorax shows a large parapneumonic effusion with multiple septations (arrows). Consolidation of the adjacent lung also is present (asterisk). B, An axial postcontrast computed tomography (CT) image shows the pleural effusion (asterisk) in the anterior aspect of the left base. The septations seen on the ultrasound are not clearly visualized on CT; however, pleural enhancement is present (curved arrow). The adjacent consolidated lung also is noted (straight arrow).
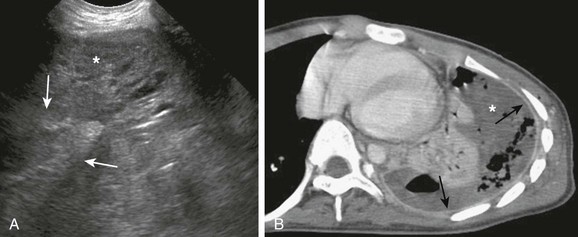
A, An ultrasound image shows a mostly solid-appearing parapneumonic effusion (arrows). The consolidated underlying lung (asterisk) also is seen. B, An axial contrast-enhanced computed tomography image shows the large pleural effusion (asterisk) with pleural enhancement (arrows).
Parapneumonic Effusion and Empyema
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree